Abstract
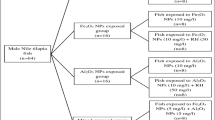
Metal oxide nanoparticles have wide applications, which have elevated serious alarms about their impacts on the environment. Therefore, we investigated the potential adsorptive capacity of rice husk toward Fe2O3 and Al2O3 nanoparticles to reduce their genotoxic effects. Fish were subjected to 10 mg/l of Fe2O3 and Al2O3 nanoparticles in single and combined doses with and without rice husk water treatment for 7 days. The genotoxic effects were evaluated using the micronucleus test in the peripheral blood and comet assay in liver tissues. Significant elevation of micronuclei induction in addition to eight nuclear and cytoplasmic abnormalities (P < 0.05) was observed in all fish groups compared to the control groups. Fish that exposed to Fe2O3 nanoparticle showed the maximum induction of all recorded anomalies. Moreover, two indices of DNA damage were evaluated by the comet assay (comet score and % tail DNA) in liver tissues. The scoring of comet cells indicated that the highest frequencies of stage 0 (undamaged DNA) were in control and Al2O3 exposed groups, while stage 4 (extensive DNA damage) was significantly elevated in Fe2O3 exposed fish. The % of DNA damage was maximized in the Fe2O3 nanoparticles exposed fish and minimized in Al2O3 nanoparticles exposed fish. Based on the frequencies of nuclear anomalies, degree, and percentage of DNA damage, all rice husk treated groups showed a marked reduction in the genotoxic damage compared with untreated groups. Finally, both nanoparticles showed genotoxic potential and the rice husk had an efficient absorptive capacity for both of them individually or combined.




Similar content being viewed by others
References
Abdel-Khalek, A. A. (2015). Antioxidant responses and nuclear deformations in freshwater fish, Oreochromis niloticus, facing degraded environmental conditions. Bulletin of Environmental Contamination and Toxicology, 94(6), 701–708.
Abdel-Khalek, A. A. (2016). Comparative evaluation of genotoxic effects induced by CuO bulk and nano-particles in Nile Tilapia, Oreochromis niloticus. Water, Air and Soil Pollution, 227, 35.
Abdel-Khalek, A. A., Badran, S. R., & Marie, M.-A. S. (2020). The efficient role of rice husk in reducing the toxicity of iron and aluminum oxides nanoparticles in Oreochromis niloticus: Hematological, bioaccumulation, and histological endpoints. Water, Air and Soil Pollution, 231, 53.
Abdel-Khalek, A. A., Hamed, A., & Marie, M.-A. S. (2016). The accumulation potency of bulk and nano zinc metal and their impacts on the hematological and histological perturbations of Oreochromis niloticus. Water, Air and Soil Pollution, 227, 206.
Acharya, J., Sahu, J., Mohanty, C., & Meikap, B. (2009). Removal of lead (II) from wastewater by activated carbon developed from tamarind wood by zinc chloride activation. Chemical Engineering Journal, 149, 249–262.
Aki, T., Nara, A., & Uemura, K. (2012). Cytoplasmic vacuolization during exposure to drugs and other substances. Cell Biology and Toxicology, 28, 125–131.
Canli, E. G., Dogan, A., & Canli, M. (2018). Serum biomarker levels alter following nanoparticle (Al2O3, CuO, TiO2) exposures in freshwater fish (Oreochromis niloticus). Environmental Toxicology and Pharmacology, 62, 181–187.
Chen, P. J., Tan, S. W., & Wu, W. L. (2012). Stabilization or oxidation of nanoscale zerovalent iron at environmentally relevant exposure changes bioavailability and toxicity in medaka fish. Environmental Science and Technology, 46(15), 8431–8439.
Cheng, C., Wang, J., Yang, X., Li, A., & Philippe, C. (2014). Adsorption of Ni (II) and cd (II) from water by novel chelating sponge and the effect of alkali-earth metal ions on the adsorption. Journal of Hazardous Materials, 264, 332–341.
Cherukuri, P., Glazer, E. S., & Curley, S. A. (2010). Targeted hyperthermia using metal nanoparticles. Advanced Drug Delivery Reviews, 62, 339–345.
Fenech, M., Chang, W. P., Kirsch-Volders, M., Holland, N., Bonassi, S., & Zeiger, E. (2003). HUMN project: Detailed description of the scoring criteria for the cytokinesis block micronucleus assay using isolated human lymphocyte cultures. Mutation Research, 534(1–2), 65–75.
Fenech, M., Kirsch-Volders, M., Natarajan, A. T., Surralles, J., Crott, J. W., Parry, J., Norppa, H., Eastmond, D. A., Tucker, J. D., & Thomas, P. (2011). Molecular mechanisms of micronucleus, nucleoplasmic bridge and nuclear bud formation in mammalian and human cells. Mutagenesis, 26(1), 125–132.
Heddle, J. A., Cimino, M. C., Hayashi, M., Romagna, F., Shelby, M. D., Tucker, J. D., Vanparys, P., & MacGregor, J. T. (1991). Micronuclei as an index of cytogenetic damage: Past, present, and future. Environmental and Molecular Mutagenesis, 18, 277–291.
Javed, M., Ahmad, I., Usmani, N., & Ahmad, M. (2016). Bioaccumulation, oxidative stress and genotoxicity in fish (Channa punctatus) exposed to a thermal power plant effluent. Ecotoxicology and Environmental Safety, 127, 163–169.
Jones, C. F., & Grainger, D. W. (2009). In vitro assessments of nanomaterial toxicity. Advanced Drug Delivery Reviews, 61, 438–456.
Kaygisiz, S. Y., & Ciğerci, İ. H. (2017). Genotoxic evaluation of different sizes of iron oxide nanoparticles and ionic form by SMART, Allium and comet assay. Toxicology and Industrial Health, 33(10), 802–809.
Kazimirova, A., Baranokova, M., Staruchova, M., Drlickova, M., Volkovova, K., & Dusinska, M. (2019). Titanium dioxide nanoparticles tested for genotoxicity with the comet and micronucleus assays in vitro, ex vivo and in vivo. Mutation Research, 843, 57–65.
Khan, M. S., Qureshi, N. A., & Jabeen, F. (2017). Assessment of toxicity in freshwater fish Labeo rohita treated with silver nanoparticles. Applied Nanoscience, 7, 167–179.
Krishnani, K. K., & Ayyappan, S. (2006). Heavy metals remediation of water using plants and lignocellulosic agrowastes. Reviews of Environmental Contamination and Toxicology, 188, 59–84.
Landsiedel, R., Kapp, M. D., Schulz, M., Wiench, K., & Oesch, F. (2009). Genotoxicity investigations on nanomaterials: Methods, preparation and characterization of test material, potential artifacts and limitations—Many questions, some answers. Mutation Research, 681, 241–258.
Lewis, W. K., Harruff, B. A., Gord, J. R., Rosenberger, A. T., Sexton, T. M., Guliants, E. A., & Bunker, C. E. (2010). Chemical dynamics of aluminum nanoparticles in ammonium nitrate and ammonium perchlorate matrices: Enhanced reactivity of organically capped aluminum. Journal of Physical Chemistry C, 115, 70–77.
Liao, W., McNutt, M. A., & Zhu, W. (2009). The comet assay: A sensitive method for detecting DNA damage in individual cells. Methods, 48, 46–53.
Mahboob, S., Al-Balwai, H. F. A., Al-Misned, F., & Ahmad, Z. (2014). Investigation on the genotoxicity of mercuric chloride to freshwater Clarias gariepinus. Pakistan Veterinary Journal, 34, 100–103.
Mottola, F., Iovine, C., Santonastaso, M., Romeo, M. L., Pacifico, S., Cobellis, L., & Rocco, L. (2019). NPs-TiO2 and lincomycin coexposure induces DNA damage in cultured human amniotic cells. Nanomaterials, 9(11), 1511.
Murali, M., Suganthi, P., Athif, P., Bukhari, A. S., Mohamed, S. H. E., Basu, H., & Singhal, R. K. (2017). Histological alterations in the hepatic tissues of Al2O3 nanoparticles exposed freshwater fish Oreochromis mossambicus. Journal of Trace Elements in Medicine and Biology, 44, 125–131.
Nabiev, I., Mitchell, S., Davies, A., Williams, Y., Kelleher, D., Moore, R., Gunko, Y. K., Byrne, S., Rakovish, Y. P., Donegan, J. G., Sukhanova, A., Conroy, J., Cottell, D., Gaponik, N., Rogach, A., & Volkov, Y. (2007). Non-functionalized nanocrystals can exploit a cell’s active transport machinery delivering them to specific nuclear and cytoplasmic compartments. Nano Letters, 7(11), 3452–3461.
Rajiv, S., Jerobin, J., Saranya, V., Nainawat, M., Sharma, A., Makwana, P., Gayathri, C., Bharath, L., Singh, M., Kumar, M., Mukherjee, A., & Chandrasekaran, N. (2016). Comparative cytotoxicity and genotoxicity of cobalt (II, III) oxide, iron (III) oxide, silicon dioxide, and aluminum oxide nanoparticles on human lymphocytes in vitro. Human and Experimental Toxicology, 35(2), 170–183.
Recio, L., Hobbs, C., Caspary, W., & Witt, K. L. (2010). Dose-response assessment of four genotoxic chemicals in a combined mouse and rat micronucleus (MN) and comet assay protocol. The Journal of Toxicological Sciences, 35(2), 149–162.
Rekulapally, R., Murthy Chavali, L. N., Idris, M. M., & Singh, S. (2019). Toxicity of TiO2, SiO2, ZnO, CuO, au and Ag engineered nanoparticles on hatching and early nauplii of Artemia sp. Peer Journal, 6, 1–16.
Sadiq, R., Khan, Q. M., Mobeen, A., & Hashmat, A. J. (2015). In vitro toxicological assessment of iron oxide, aluminum oxide and copper nanoparticles in prokaryotic and eukaryotic cell types. Drug and Chemical Toxicology, 38(2), 152–161.
Saravanan, M., Suganya, R., Ramesh, M., Poopal, R. K., Gopalan, N., & Ponpandian, N. (2015). Iron oxide nanoparticles induced alterations in haematological, biochemical and ionoregulatory responses of an Indian major carp Labeo rohita. Journal of Nanoparticle Research, 17, 274–285.
Sayed, A. E. D. H. (2016). Genotoxicity detection following exposure to silver nanoparticles in African catfish (Clarias gariepinus). International Journal of Nanoparticles, 9(1), 41–53.
Song, M. F., Li, Y. S., Kasai, H., & Kawai, K. (2012). Metal nanoparticle-induced micronuclei and oxidative DNA damage in mice. Journal of Clinical Biochemistry and Nutrition, 50(3), 211–216.
Srivastava, V. C., Mall, I. D., & Mishra, I. M. (2006). Characterization of mesoporous rice husk ash (RHA) and adsorption kinetics of metal ions from aqueous solution onto RHA. Journal of Hazardous Materials, 134(1–3), 257–267.
Thorek, D. L., Chen, A. K., Czupryna, J., & Tsourkas, A. (2006). Superparamagnetic iron oxide nanoparticle probes for molecular imaging. Annals of Biomedical Engineering, 34, 23–38.
Tice, R. R., Agurell, E., Anderson, D., Burlinson, B., Hartmann, A., Kobayashi, H., Miyamae, Y., Rojas, E., Ryu, J.-C., & Sasaki, Y. F. (2000). Single cell gel/comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environmental and Molecular Mutagenesis, 35, 206–221.
Zhu, B., He, W., Hu, S., Kong, R., & Yang, L. (2019). The fate and oxidative stress of different sized SiO2 nanoparticles in zebrafish (Danio rerio) larvae. Chemosphere, 225, 705–712.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Khalid University, Saudi Arabia for funding this work through research group project under grant number (R.G.P.1–56–40) and to the Faculty of Science, Cairo University, Egypt for supporting the current work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This manuscript complies with the ethical rules applicable for this journal.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abdel-Khalek, A.A., Dajem, S.B. & Morsy, K. The Potential Use of Rice Husk for Reducing the Genotoxic Effects of Iron and Aluminum Oxides Nanoparticles in Oreochromis niloticus. Water Air Soil Pollut 231, 139 (2020). https://doi.org/10.1007/s11270-020-04495-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-020-04495-0