Abstract
Prometryn has been used in crop (e.g., corn and sorghum) field to prevent growth of annual grasses and broadleaf weeds for many years. As a moderately persistent herbicide in soil, prometryn may exert detrimental effects on environmental safety and crop production. The present study assessed the photodegradation of prometryn residues in soil by exploring a variety of factors such as soil moisture, temperature, and light exposure that potentially affect prometryn photodegradation. The dissipation rate of prometryn during a 14-day period of study was more than 90% under 15 (low pressure), 100, and 300 W (medium pressure) UV light exposure. The half-life of prometryn decay under UV light (53.5–116.4 h) was far less than that under xenon light (1131.6 h) and dark (3138.7 h) conditions. When the soil moisture (clay loam) was 60% of the field moisture capacity, it was most effective for prometryn photodegradation. The prometryn photodegradation on soil with 60% moisture level was increased with temperature and prometryn concentrations. The theoretical optimization scheme for eliminating prometryn in soil was recommended. The degraded products of prometryn under UV light and darkness were characterized using ultra high-performance liquid chromatography coupled to a linear ion trap-orbitrap hybrid mass spectrometer (UPLC-LTQ-orbitrap-MS/MS) and showed that prometryn decay in soil was through hydroxylation, dealkylation, and dethiomethylation pathways.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Application of herbicides in agriculture has greatly improved the crop production (Graebing et al. 2003). However, extensive use of herbicides in the current agronomic practice may burden soil because scavenging of soil herbicide residues is a time-consuming process. Furthermore, herbicides accumulated in soil have become contaminants and consequently risked the crop production, food safety, and human health (Seiber and Kleinschmidt 2011). Soil as a component of ecosystem has evolved elaborate mechanisms to scavenge toxicants such as herbicides. These mechanisms concern biological, chemical, and physical processes (Gu et al. 2012; Gao et al. 2013; Wang et al. 2014). Photodegradation is a remediation by the chemical reaction that is frequently used for cleanup of organic contaminants in soil and is affected by an array of environmental factors such as soil type, moisture, pH, humic substances, and temperature (Wang et al. 2014). For example, soil moisture promotes hydrolysis of pesticides by mobilizing chemicals in soil (Frank et al. 2002). The physical-mediated processes play important roles in degradation of organic toxicants (Graebing and Chib 2004; Si et al. 2004; Tajeddine et al. 2010; Sun et al. 2011; Khandelwal et al. 2014). Of these, photolysis is a significantly degradative pathway for removal of contaminants in soil (Lányi and Dinya 2003; Baranda et al. 2012). To date, numerous studies have been documented by applying UV radiation and photocatalysts as artificial situation to photodegradation (Quan et al. 2005; Wang et al. 2006; Zhang et al. 2008). The photodegradation was more effective under UV light than under sunlight (Wang et al. 2006; Das and Mukherjee 2011).
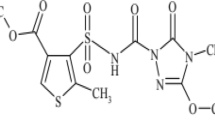
Prometryn is a selective herbicide of the s-triazine chemical family and has been widely used for pre- or post-emergence control of annual grasses and broadleaf weeds. Its environmental behavior in soils was investigated through soil property, solubility, Koc, Kow, stability, and soil adsorption (Cao et al. 2008). It was reported that the mean Koc value of prometryn was 400 (Wauchope et al. 1992). Prometryn was shown to have the relative low mobility in soil based on the classification scheme (Swann et al. 1983) and was stable in soil with the long half-life of approximately 60 days (Thurman et al. 2000). Over the last decades, prometryn residues have been detected in environment and breast milk (Balduini et al. 2003; Papadopoulou-Mourkidou et al. 2004). Meanwhile, prometryn was also found photocatalytically degraded in water system (Larson et al. 1991; Navarro et al. 2004; Vela et al. 2004; Kiss et al. 2007; Evgenidou et al. 2007; Wallace et al. 2010; Remucal 2014). However, less attention has been paid to the prometryn photodegradation in soil medium. In this study, we investigated the effects of soil moisture, temperature, the initial concentration of prometryn in soil, and different light exposures on photodegradation of prometryn in soil. The possible pathways of prometryn degradation under different conditions were presented. The objectives of the study were to evaluate the efficiency of prometryn degradation under the different conditions and to provide a potential way for decomposing prometryn in soil medium.
2 Materials and Methods
2.1 Soil Treatment
Prometryn (97.3% purity) was provided by Academy of Agricultural Science in Jiangsu, China. Pesticide-free soil (red earth) for experiment was collected from Yingtan, China (Dystric Regosols, N 28.23°; E 117.02°). The collected soil was air-dried, passed through a 0.85-mm sieve, and stored in darkness. The textural classes of the soil were detailed as follows: organic carbon, 0.54%; pH, 5.45; sand, 39.80%; silt, 28.20%; clay, 33.00%; and cation exchange capacity, 11 cmol kg−1. Soil was sterilized according to the method of Wang et al. The soil was autoclaved at 120 °C and 1.1 atm for 30 min. This procedure was repeated in triplicate to ensure complete sterilization of the soil. The unsterilized and sterilized soils were spiked with prometryn at 10 mg kg−1 as the initial testing concentration. After the spiked soil was stored under darkness for 24 h, the experiment started under light irradiation. The experiments of light exposures were ended when the concentration of prometryn was close to or less than 10% of the initial concentration in soil. Under light of xenon lamp and 15-, 100-, or 300-W mercury lamps, the length of exposure time was selected as 65 and 14 days, respectively.
2.2 Irradiation Treatment
The irradiation experiment was carried out in a climate chamber with a lighting device with a horizontal water-cooled quartz immersion (Nanjing Xujiang Machinery Factory, China). The lamps were set up with three mercury lamps (UV, 15, 100, and 300 W) and a xenon lamp of 350 W. The 15-W lamp was a low-pressure quartz mercury lamp. The 100- and 300-W lamps were undersized medium-pressure mercury in quartz lamps. The wavelengths and light intensities on the surface of soil samples are listed in Supplementary Table S1. The system incorporated a rotatable base plate below the light away from 15 cm, which was capable of regulating temperature by circulating water. The temperature 25 °C of climate chamber was maintained in the circulating water system. Experiments were performed as the following irradiation procedure: the soil was treated with different amounts of water uniformly to achieve different levels of soil humidity. The soil samples (2 g) were spread uniformly on quartz dishes (100 × 50 × 5 mm) with a 1.0 ± 0.1-mm thickness and homogeneously irradiated for 6 h daily. The quartz dishes containing the soil samples with the initial moisture level were weighed before the illumination experiment. During the experiment, the soil humidity was measured periodically (every 0.5 h) by weighing and the samples were sprayed with deionized water timely to maintain at the same level as initial moisture level (Gupta et al. 2008). The information of delivered radiation (J/cm2) is shown in Supplementary Table S1. The control soil with prometryn was incubated in darkness. Experiments were repeated in triplicate.
2.3 Soil Moisture
The treated soil (2 g) with 10 mg kg−1 prometryn was brought to the 0, 40, 60, and 90% field moisture capacity by adding 0, 0.2, 0.3, and 0.45 mL water and transferred to each plate, respectively. The concentration of 10 mg kg−1 prometryn was set based on the dry weight of soil. Before being treated under darkness and UV light (100 W) at 25 ± 1 °C, the soil samples were weighed. The sum of water loss in the soil samples was measured during all experiments. The moisture levels of the soil samples were maintained by compensation of lost water periodically. Soil samples in triplicate were drawn at different time intervals (0, 1, 3, 5, 7, 10, and 14 days). Prometryn residues in soil with different moisture treatments were quantified based on the dry weight.
The reflectance spectra of soil in the four moisture levels (0, 40, 60, and 90%) were recorded using a UV-2600 spectrophotometer (Shimadzu, Japan) equipped with diffuse reflectance accessory (Twomey et al. 1986). The reflectance of soil was measured between 200 and 800 nm with a 0.5-nm interval. The reflectance values were converted into absorbance.
2.4 Temperature Treatment
Soils with prometryn at 10 mg kg−1 were exposed to UV light (100 W) at 60% moisture content with three levels of temperature 15, 25, and 35 °C. The concentration of 10 mg kg−1 prometryn was based the on dry weight of soil. The intervals of sampling and analyses corresponded to those in the soil moisture experiment. Residues of prometryn in soil at different temperatures were quantified based on the dry weight.
2.5 Differently Fortified Concentrations of Prometryn
Soils were exposed to prometryn at 5, 10, and 15 mg kg−1 levels, respectively. The concentrations in the soil samples were close to the ordinary field dosage (800–1500 g a.i. ha−1) (Konstantinou et al. 2001). The samples were held at 60% moisture and illuminated under UV light (100 W) with a temperature of 25 ± 1 °C. Each concentration of prometryn in soil samples was analyzed at the time intervals as described earlier.
2.6 Determination of Prometryn and Degraded Products in Soil
Prometryn residues in soil were determined according to the method of Cao et al. (2008), with slight modification. The soil sample was extracted with the mixture solution of acetone and water (3:1, v/v) in an ultrasonic bath for three times, each time with 20 mL for 30 min. The extracted solution was centrifuged and filtrated. The filtrate was pooled and concentrated to remove acetone by vacuum evaporation at 40 °C. The remaining water was extracted with petroleum ether for three times, each time with 15 mL. The organic phase was concentrated to dryness by vacuum evaporation at 40 °C. The residue was dissolved in methanol (chromatographic grade) and detected by high-performance liquid chromatography (HPLC) (Waters 515; Water Technologies Co. Ltd., USA) with a 2487 Dual λ Absorbance Detector at 254 nm and a Hypersil ODS column (250 × 4.6 mm ID, 5 μm). The mobile phase was methanol/water (75:25, v/v) at a flow rate of 0.6 mL min−1 (Cao et al. 2008). The recoveries of prometryn from soils are listed in Supplementary Table S2.
To identify the degraded products of prometryn under different treatments, the soils were placed in darkness at 25 ± 1 °C for 65 days and exposed to UV light (100 W) for 3 days. The extraction and concentration process was the same as the analysis of prometryn residues in soil. The remaining water after extraction was added to C18 solid-phase extraction (SPE) column. Elute was discarded. The column was washed with 2 mL methanol. The washing solution was collected and analyzed using ultra high-performance liquid chromatography (UHPLC) (Thermo, USA) coupled to a linear ion trap-orbitrap hybrid mass spectrometer (LTQ Orbitrap XL) equipped with a heated-electrospray ionization probe. Separations were performed on a Hypersil Gold C18 column (100 mm × 2.10 mm, 3 μm, Thermo Fisher Scientific). Mobile phase was consisted of 0.1% formic acid in water (solvent A) and 0.1% formic acid in acetonitrile (solvent B). A linear elution gradient program was run for 36 min at a flow rate of 0.20 mL/min as follows: 5% (B) for 1 min, 1–15 min from 5 to 35% (B), 15–25 min from 35 to 95% (B), 95% (B) for 5 min, 30–31 min from 95 to 5% (B), and 5% (B) for 5 min. The temperatures of column oven and autosampler were 35 and 10 °C, respectively. The injection volume was 10 μL. The mass spectrometer was operated in a positive mode. The product ions were derived from the LTQ ion trap with normalized collision energy of 35% and q-activation of 0.25. The external mass calibration of orbitrap was performed once a week to ensure a working mass accuracy <5 ppm. The spectral data (accurate mass, mass spectra, and characteristic fragmentation) of prometryn and its degraded products were analyzed with the XCalibur software, version 2.1.
2.7 Data Analysis
Each experiment was performed in triplicate. Results in figures and tables were the mean of three replicated treatments and expressed as mean ± standard deviation. The data of prometryn degradation were subjected to regression analysis and fitted by the first-order kinetics (C t = C 0e−Kt). The half-life values of prometryn degradation were calculated by dissipation constant. The statistical significance of the data was determined by the variance post hoc test at the 95% confidence level (p < 0.05). The calculation formula (L = (C N − C I) / C 0 × 100%) was used to describe the proportion of prometryn degradation under UV light (Zhang et al. 2008), where L is a proportion of photodegradation at time t, C 0 is the initial concentration of prometryn in soil before incubating in darkness and light exposure, C I is the residual concentration of prometryn in soil under irradiation at time t, and C N is the residual concentration of prometryn in soil incubated in darkness at time t.
3 Results and Discussion
3.1 Effect of Soil Moisture on Photodegradation of Prometryn
Four moisture levels (0, 40, 60, and 90%) were set to investigate the effect of soil moisture on photodegradation of prometryn. Under darkness, the residual content of prometryn in wetted soil was lower than that in air-dried soil after 14-day incubation (Fig. 1a). The effect of different moisture levels (40, 60, and 90%) on prometryn degradation under darkness was insignificant. The proportion of photodegradation was calculated with the formula L = (C N − C I) / C 0 × 100%. When soil was exposed to the UV light irradiation at 100 W for 14 days, the photodegradation rate of prometryn reached 13.21, 73.97, 79.09, and 74.04% in treatments with 0, 40, 60, and 90% moisture, respectively (Fig. 1a). Compared to the dry soil, the moist soil was favorable to prometryn photodegradation, an observation in consistent with the previous report for atrazine photodegradation (Fan et al. 2005). The result indicated that water as a strong light absorber in soil accelerated the hydrolysis action in catabolic process of pesticide (Bogrekci and Lee 2006). As the metal and nitrate ions affect the hydrolysis of organic chemicals in soil, the hydrolysis of organic chemicals in soil occurs more readily than that in pure water (Feng and Nansheng 2000; Hultgren et al. 2002; Fisher et al. 2006; Tercero Espinoza et al. 2007; Du et al. 2014). However, the photodegradation rate of prometryn was not always enhanced with the increasing water content in soil. Photodegradation of prometryn in soil with 60% moisture was most effective for the four moisture levels (Fig. 1a). To further investigate the effect of soil moisture on prometryn photodegradation in soil, the reflectance spectra of soil with the four moisture levels were recorded using a spectrophotometer (Lobell and Asner 2002). While the dry soil samples had the low absorbance, the soil with 60% of field moisture capacity had the high absorbance (Fig. 1b). Notably, the prometryn photodegradation appeared to have a positive correlation with the ultraviolet absorbed intensity of soil in the 0–60% moisture. The absorbance of soil with 90% moisture in ultraviolet region (200–350 nm) was lower than the other wet soil samples. Previous studies showed that sufficient water in moist soil could seal up the crevices between particles; thus, the light was prevented to penetrate through the moist soil (Frank et al. 2002). The radar chart was used to estimate the beneficial moisture for the degradation of prometryn. The result showed that 60% moisture was optimum for prometryn degradation (Fig. 2a). These results indicated that the soil moisture could promote the absorbance of light and photodegradation of prometryn to some extent, but excessive soil moisture could block this action.
Effect of soil moisture on prometryn on soil surfaces under dark and UV irradiation (a) and the diffuse reflectance spectra of soils with different moisture levels in the UV–vis range (b). Soil containing prometryn at 10 mg kg−1 level was exposed for 6 h daily to UV light (100 W) at 25 ± 1 °C with 0, 40, 60, and 90% moisture, respectively. Prometryn residues in soils were quantified at the time intervals of 0, 1, 3, 5, 7, 10, and 14 days. Values are the means ± SD (n = 3)
3.2 Effect of Temperature on Photodegradation of Prometryn
Three levels of temperature (15, 25, and 35 °C) were used to test the photodegradation of prometryn. Following a 14-day period of irradiation, the soil residues of prometryn were only 3.16% at 35 °C, 11.29% at 25 °C, and 16.20% at 15 °C of the initial concentration, respectively (Fig. 3). The values of half-life increased with the decrease of temperature (Supplementary Table S3), indicating that the high temperature was effective to degrade prometryn. The best condition for prometryn degradation was 35 °C based on the theoretical optimization scheme (Fig. 2b).
Effect of temperature on prometryn on soil surfaces under dark and UV irradiation. Soil containing prometryn at 10 mg kg−1 level was exposed for 6 h daily to UV light (100 W) with 60% moisture at 15 ± 1, 25 ± 1, and 35 ± 1 °C, respectively. Prometryn residues in soils were quantified at the time intervals of 0, 1, 3, 5, 7, 10, and 14 days. Values are the means ± SD (n = 3)
3.3 Effect of Initial Concentration on Photodegradation of Prometryn
The photolysis kinetics of prometryn at different initial concentrations on soil surface is shown in Fig. 4. There were 82.13, 88.71, and 92.40% reductions of prometryn in soil with 14-day illumination at the initial levels of 5, 10, and 15 mg kg−1, respectively. The photodegradation of prometryn on soil surface under UV light followed the first-order kinetics. The correlation coefficient (R 2) value was higher than 0.95. The half-life of prometryn at the initial levels of 15 mg kg−1 was shortest among the three initial concentration levels (Supplementary Table S4). The increasing dosage of prometryn from 5 to 15 mg kg−1 had a modest effect on photodegradation, suggesting the faster transformation rate of prometryn in the high-dose treatments. The observation was very similar to pentachlorophenol photodegradation (Subramanian et al. 2007).
Degradation kinetics of prometryn on soil surfaces at different initial concentrations. Soils containing prometryn at 5, 10, and 15 mg kg−1 were exposed for 6 h daily to UV light (100 W) with 60% moisture at 25 ± 1 °C. Prometryn residues in soils were quantified at the time intervals of 0, 1, 3, 5, 7, 10, and 14 days. Values are the means ± SD (n = 3)
3.4 Effect of Different Light Exposures on Photodegradation of Prometryn
To evaluate the prometryn photolysis rates on soil surface under different light exposures, the soil samples exposed to 10 mg kg−1 prometryn at 60% moisture were irradiated by the light of xenon lamp for 65 days and ultraviolet light of 15-, 100-, and 300-W mercury lamps for 14 days, respectively. The prometryn photodegradation in soil under different light exposure followed the first-order kinetics (R 2 > 0.95) (Table 1). The faster degradation of prometryn was observed under the light exposure compared to the dark control. The dissipation of prometryn in xenon light irradiation was 65.40% of the initial concentration and more than twofold that under dark condition. The half-life values of prometryn were 1131.6 h in xenon light and 3138.7 h in dark control. The lost prometryn under UV light exposure for 14 days was close to 90%, and the half-life values were far less than those under xenon light and dark conditions (Table 1). These results indicated that the UV radiation facilitated the phototransformation of prometryn in soil. The photodegradation rate of prometryn increased with the UV intensity. The half-life of prometryn under 300 W UV was shortest (53.52 h) among the three UV light treatments. The half-life values of prometryn with 15 and 100 W UV were 116.4 and 109.44 h, respectively. The illumination intensity had a marked impact on the photolytic rate of prometryn. Lányi and Dinya (2003) reported that increasing degrading energy from 15 to 125 W UV improved the formation of single and less dangerous metabolites for some triazine-type herbicides. The degradation kinetic parameters of prometryn with the different light exposures were presented in radar chart (Fig. 2c). The comprehensive analysis of degradation kinetic parameters of prometryn displayed the best optimum scheme under 300 W UV, at 35 °C and with 60% soil moisture (Fig. 2d). To compare the energy consumption in the photodegradation process, the total delivered radiation on soil sample was investigated. The total radiation was calculated by the treatment time (s) and light intensity (μW cm−2) of the different light sources (Supplemental Table S1). The total delivered radiation at two wavelengths (254 and 365 nm) with 15-, 100-, and 300-W UV light for 14 days was 37.8, 517, and 699 J cm−2, respectively, for the prometryn photodegradation. The higher value of total delivered radiation pointed to the greater consumption of light energy in the photolytic process. The consumption of light energy in prometryn photodegradation was highest under 300 W UV, and the lowest delivered radiation with 15-W UV light was only 7% of that with 100 W UV. The half-life of prometryn photodegradation under 15 W (116.4 h) was slightly longer than that under 100-W UV light (109.44 h). Furthermore, the prometryn photodegradation was influenced by the wavelengths of emission spectra because the UV absorption occurred at the pesticide molecule (Zhang et al. 2008; Burrows et al. 2002). The 254-nm wavelength was in the range of the UV absorption of prometryn, whereas rare absorption of prometryn was found at 365 nm (Supplementary Fig. S1). Considering the energy conservation, 15-W UV light was more suitable to apply for the photodegradation of prometryn in soil.
3.5 Photodegradation of Prometryn on the Surface of Sterilized Soil
To investigate the effect of UV light on prometryn degradation on the soil surface, the degradation experiments of prometryn in sterilized soil were performed under darkness and UV light. The degradation rate of prometryn was modestly higher in unsterilized soil than in sterilized soil under darkness, but there was no significant difference (Supplementary Fig. 2S). It was suggested that soil microorganism could be destroyed under UV irradiation. The half-lives of prometryn degradation in unsterilized and sterilized soils were 2028.7 and 2188.8 h, respectively. After a 14-day period of UV irradiation, there was no significant difference of the prometryn photodegradation rates between the sterilized and unsterilized soil surfaces except for on the seventh and tenth days under the condition of 90% moisture and UV irradiation at 25 ± 1 °C (Supplementary Fig. 3S). Meanwhile, no significant difference of the prometryn photodegradation rates between the sterilized and unsterilized soil surfaces was observed at the time intervals of 0, 1, 5, 7, 10, and 14 days except for the third day under the condition of 60% moisture and UV irradiation at 35 ± 1 °C (Supplementary Fig. 4S). The half-life values of prometryn on sterilized and unsterilized soil under UV irradiation were 112.8 and 117.1 h (90% moisture, 25 ± 1 °C) and 73.4 and 72.2 h (60% moisture, 35 ± 1 °C), respectively (Supplementary Table 5S). These results suggested that the prometryn photodegradation rate and half-life on the sterilized and unsterilized soil surfaces were similar under UV irradiation, with a rare effect of soil microorganisms on the prometryn photodegradation on the soil surface. However, the significant difference of prometryn photodegradation rate on the seventh and tenth days (90% moisture) and on the third day (for 60% moisture) on the sterilized and unsterilized soil surfaces might result from the experiment error possibly from the uneven distribution of prometryn in soil, irradiation, or extraction.
3.6 Proposed Pathways of Prometryn Photodegradation in Soil
To investigate the proposed pathways of prometryn photodegradation in soil, the transformed products of prometryn under darkness and UV light (100 W) for 65 days and 3 days were characterized. Under this condition, nine transformed products of prometryn were identified by LC-MS (Table 2 and Fig. 5). Prometryn chromatogram showed a retention time of 20.35 min, and the structure was confirmed by the presence of [M+H]+ ions with m/z 242 (Table 2). The parent ion of prometryn was split into two major fragment ions at m/z 200 and m/z 158 (Fig. 5a). 6-Hydroxy-1,3,5-triazine-2,4-diamine (HDDPP) peaked at 1.62 min was confirmed by the fragment ion at m/z 86 (Table 2 and Fig. 5b), which was generated from the precursor ions at m/z 128 through a ring-opening rearrangement reaction and loss of NH2CN (Fig. 5b). The molecular parent ion of N-ethyl-N′-isopropyl-6-hydroxy-1,3,5-triazine-2,4-diamine (HDMP) at m/z 198 (8.30 min) was confirmed by the fragment ion peaks at m/z 180 and m/z 156 formed due to loss of a molecule of H2O and isopropyl, respectively (Table 2 and Fig. 5c). The fragment ion peak with m/z 128 of N-isopropyl-6-hydroxy-1,3,5-triazine-2,4-diamine (HDPP) at 10.39 min was generated by loss of an isopropenyl from HDPP (Table 2 and Fig. 5d). N-isopropyl-1,3,5-triazine-2,4-diamine (DSPP) at 12.05 min with fragment ion peak at m/z 112 was one of the dethiomethylated products (Table 2 and Fig. 5e). 6-(Methylsulfanyl)-6-hydroxy-1,3,5-triazine-2,4-diamine (DDPP) at 12.37 min was identified as the deisopropylated product of prometryn. In the MS2 of DDPP, the fragment ion peaks at m/z 110 and m/z 116 were formed through the loss of thiomethyl group and NH2CN, respectively (Table 2 and Fig. 5f). N,N′-diisopropyl-1,3,5-triazine-2,4-diamine (DSP) (m/z 196) peaked at 16.26 min was identified as dethiomethyl-prometryn by analyzing the fragment ion peaks at m/z 154 and m/z 112 formed by N-dealkylation (Table 2 and Fig. 5g). N-Isopropyl-6-(methylsulfanyl)-1,3,5-triazine-2,4-diamine (DPP) with the elimination of isopropyl from prometryn was identified according to the fragment ion peak at m/z 158 (Table 2 and Fig. 5h). N-ethyl-N′-isopropyl-6-(methylsulfanyl)-1,3,5-triazine-2,4-diamine (DMP) occurring at 17.60 min was confirmed by appearance of the fragment ion peaks at m/z 186 and m/z 138 (Table 2 and Fig. 5i). Hydroxyprometryn (HP) peaked at 17.95 min was derived from prometryn by substitution of a hydroxyl group for the thiomethyl. The fragment ion peaks at m/z 170 and m/z 128 were formed by N-dealkylation (Table 2 and Fig. 5j).
In the case of darkness, two major pathways were proposed including hydroxylation and dealkylation based on the detected transformed products of prometryn (Fig. 6). The representative hydrolytic product of PRO was N,N′-diisopropyl-6-hydroxy-1,3,5-triazine-2,4-diamine (HP). HDPP was formed by loss of isopropyl group from HP. HDDPP as a final product was substantially identified. Hydroxylation was the major way that attributed a lot to the microbial hydrolysis of prometryn (Meyer et al. 2009). In this study, the aromatic ring might be activated by the connection with H+ and be earlier attacked by nucleophile like H2O, which led to the elimination of CH3SH and formation of hydroxylation products in the acidic soil (pH 5.45) (Meyer et al. 2009). In the pathway of dealkylation, prometryn was cleaved into an isopropyl to produce DPP, which was also formed by the deethylation of DMP. Furthermore, DDPP, a lower-molecule derivative, was observed in the dealkylation process. Its formation could be derived from oxidative dealkylation (Erickson 1989; Ellis et al. 2006).
For the irradiation, the special pathway of dethiomethylation was presented (Fig. 6). DSP and DSPP were found in the process of dethiomethylation. Studies by Kiss et al (2007) showed that the aqueous photodegradation of prometryn by UV irradiation underwent two parallel ways of degradation through loss of thiomethyl and alkyl groups. Two major degraded products, DSP (loss of thiomethyl group) and DPP (loss of alkyl group), were further degraded to DSPP by loss of the isopropyl groups and thiomethyl groups, respectively. Interestingly, both DSP and DSPP were formed under UV condition. Generation of the two products was attributed to homeolysis of the C–S bond at the 2-position of triazine ring of excited prometryn, leading to the triazinyl radical. Subsequently, the resulting triazinyl radical obtained an H atom from H2O (Azenha et al. 2003; Filipiak et al. 2005; Gu et al. 2012). This was a direct photodegradation process. In the pathway of hydroxylation, the substitution of methylthio group to hydroxy group was the dominant process in the photodegradation of some triazine-type herbicides (Lányi and Dinya 2003), where HP, HDPP, and HDMP were found. The degraded products by the hydroxylation pathway were consistent with the previous results (Lányi and Dinya 2003; Canle et al. 2005; Remucal 2014). The generation of hydroxylation product (HP) might be the result of the addition of H2O or OH− at the 2-position of excited prometryn, followed by the elimination of a methylthio group. However, this pathway might not be prominent due to the slow rate of direct photodegradation for prometryn (Konstantinou et al. 2001; Navarro et al. 2004). More likely, the removal of prometryn occurred via an indirect photochemical degradation, especially reacted with reactive intermediates. The similar results were reported in aqueous media (McMartin et al. 2003; Wallace et al. 2010). Although several low molecular weight degradation products such as DDPP and HDDPP were not identified in soil under UV light, some other products such as HP, DMP, DPP, DSP, DSPP, HDPP, and HDDPP identified here were reported to relate directly photodegradation and photocatalytic degradation of prometryn in aqueous solutions or natural water (Konstantinou et al. 2001; Evgenidou and Fytianos 2002; Canle et al. 2005; Devipriya and Yesodharan 2005; Kiss et al. 2007). In this study, HDMP and DDPP were identified as the photodegradation products of prometryn in soil for the first time.
4 Conclusions
Dissipation studies on prometryn under the dark, xenon lamp, and UV radiation conditions revealed that prometryn was sensitive to light, especially to UV radiation. The photodegradation rate of prometryn was affected by environmental factors. Prometryn dissipation in 60% moisture soil was faster than that of prometryn in soil with any other moisture levels under UV radiation exposure. The increasing temperature and dosage of prometryn could facilitate the dissipation of prometryn in soil. UV light promoted the photodegradation of prometryn on soil and decreased the content of prometryn in soil. The rates of prometryn transformation under UV irradiation were much higher than those under darkness. Prometryn could be degraded through multi-pathways of dealkylation and hydroxylation under irradiation and darkness. Moreover, prometryn could be transformed by dethiomethylation under UV irradiation. The new photodegraded products HDMP and DDPP were identified.
References
Azenha, M. E. D. G., Burrows, H. D., Canle, L., Coimbra, R., Fernández, M. I., García, M. V., Peiteado, M. A., & Santaballa, J. A. (2003). Kinetic and mechanistic aspects of the direct photodegradation of atrazine, atraton, ametryn and 2-hydroxyatrazine by 254 nm light in aqueous solution. Journal of Physical Organic Chemistry, 16(8), 498–503.
Balduini, L., Matoga, M., Cavalli, E., Seilles, E., Riethmuller, D., Thomassin, M., & Guillaume, Y. C. (2003). Triazinic herbicide determination by gas chromatography–mass spectrometry in breast milk. Journal of Chromatography B, 794(2), 389–395.
Baranda, A. B., Barranco, A., & de Marañón, I. M. (2012). Fast atrazine photodegradation in water by pulsed light technology. Water Research, 46(3), 669–678.
Bogrekci, I., & Lee, W. S. (2006). Effects of soil moisture content on absorbance spectra of sandy soils in sensing phosphorus concentrations using UV-VIS-NIR spectroscopy. Transactions of ASABE, 49(4), 1175–1180.
Burrows, H. D., Canle, L. M., Santaballa, J. A., & Steenken, S. (2002). Reaction pathways and mechanisms of photodegradation of pesticides. Journal of Photochemistry and Photobiology B, 67(2), 71–108.
Canle, L. M., Fernandez, M. I., & Santaballa, J. A. (2005). Developments in the mechanism of photodegradation of triazine-based pesticides. Journal of Physical Organic Chemistry, 18(2), 148–155.
Cao, J., Guo, H., Zhu, H. M., Jiang, L., & Yang, H. (2008). Effects of SOM, surfactant and pH on the sorption–desorption and mobility of prometryn in soils. Chemosphere, 70(11), 2127–2134.
Das, S. K., & Mukherjee, I. (2011). Effect of light and pH on persistence of flubendiamide. Bulletin of Environmental Contamination and Toxicol, 87(3), 292–296.
Devipriya, S., & Yesodharan, S. (2005). Photocatalytic degradation of pesticide contaminants in water. Solar Energy Material and Solar Cells, 86(3), 309–348.
Du, Y. X., Chen, H., Zhang, Y., & Chang, Y. G. (2014). Photodegradation of gallic acid under UV irradiation: insights regarding the pH effect on direct photolysis and the ROS oxidation-sensitized process of DOM. Chemosphere, 99, 254–260.
Ellis, L. B. M., Roe, D., & Wackett, L. P. (2006). The University of Minnesota biocatalysis/biodegradation database: the first decade. Nucleic Acids Research, 34(suppl 1), D517–D521.
Erickson, L. E. (1989). Degradation of atrazine and related s-triazines. Critical Reviews in Environmental Control, 19(1), 1–14.
Evgenidou, E., & Fytianos, K. J. (2002). Photodegradation of triazine herbicides in aqueous solutions and natural waters. Journal of Agriculture and Food Chemistry, 50(22), 6423–6427.
Evgenidou, E., Bizani, E., Christophoridis, C., & Fytianos, K. (2007). Heterogeneous photocatalytic degradation of prometryn in aqueous solutions under UV–Vis irradiation. Chemosphere, 68(10), 1877–1882.
Fan, X. Z., Lu, B., Gong, A. J. (2005). Dynamics of solar light photodegradation behavior of atrazine on soil surface. Journal of Hazardous Materials, 117(1), 75–79.
Feng, W., Nansheng, D. (2000). Photochemistry of hydrolytic iron (III) species and photoinduced degradation of organic compounds. A minireview. Chemosphere, 41(8),1137–1147
Filipiak, P., Hug, G. L., Bobrowski, K., & Marciniak, B. (2005). Photochemistry of 4-(methylthio) phenylacetic acid: steady-state and laser flash photolysis studies. Journal of Photochemistry and Photobiology A, 172(3), 322–330.
Fisher, J. M., Reese, J. G., Pellechia, P. J., Moeller, P. L., & Ferry, J. L. (2006). Role of Fe (III), phosphate, dissolved organic matter, and nitrate during the photodegradation of domoic acid in the marine environment. Environmental Science and Technology, 40(7), 2200–2205.
Frank, M. P., Graebing, P., & Chib, J. S. (2002). Effect of soil moisture and sample depth on pesticide photolysis. Journal of Agricultural and Food Chemistry, 50(9), 2607–2614.
Hultgren, R. P., Hudson, R. J. M., Sims, G. K. (2002). Effects of soil pH and soil water content on prosulfuron dissipation. Journal of Agricultural and Food Chemistry, 50(11), 3236–3243.
Gao, Y. F., Yang, H., Zhan, X. H., & Zhou, L. X. (2013). Scavenging of HCHs and DDTs from soil by thermal desorption and solvent washing. Environmental Science and Pollution Research, 20(3), 1482–1492.
Graebing, P., & Chib, J. S. (2004). Soil photolysis in a moisture-and temperature-controlled environment. 2. Insecticides. Journal of Agricultural and Food Chemistry, 52(9), 2606–2614.
Graebing, P., Frank, M. P., & Chib, J. S. (2003). Soil photolysis of herbicides in a moisture-and temperature-controlled environment. Journal of Agricultural and Food Chemistry, 51(15), 4331–4337.
Gu, J. L., Dong, D. B., Kong, L. X., Zheng, Y., & Li, X. J. (2012). Photocatalytic degradation of phenanthrene on soil surfaces in the presence of nanometer anatase TiO2 under UV-light. Journal of Environmental Sciences (China), 24(12), 2122–2126.
Gupta, S., Gajbhiye, V. T., Gupta, R. K. (2008). Effect of light on the degradation of two neonicotinoids viz acetamiprid and thiacloprid in soil. Bulletin of Environmental Contamination and Toxicol, 81(2),185–189.
Khandelwal, A., Gupta, S., Gajbhiye, V. T., Varghese, E. (2014). Degradation of kresoxim-methyl in soil: Impact of varying moisture, organic matter, soil sterilization, soil type, light and atmospheric CO2 level. Chemosphere. 111, 209–217
Kiss, A., Rapi, S., & Csutorás, C. (2007). GC/MS studies on revealing products and reaction mechanism of photodegradation of pesticides. Microchemical Journal, 85(1), 13–20.
Konstantinou, I. K., Zarkadis, A. K., & Albanis, T. A. (2001). Photodegradation of selected herbicides in various natural waters and soils under environmental conditions. Journal of Environmental Quality, 30(1), 121–130.
Lányi, K., & Dinya, Z. (2003). Photodegradation study of some triazine-type herbicides. Microchemical Journal, 75(1), 1–13.
Larson, R. A., Schlauch, M. B., & Marley, K. A. (1991). Ferric ion promoted photodecomposition of triazines. Journal of Agricultural and Food Chemistry, 39(11), 2057–2062.
Lobell, D. B., & Asner, G. P. (2002). Moisture effects on soil reflectance. Soil Science Society of America Journal, 66(3), 722–727.
McMartin, D. W., Headley, J. V., Wood, B. P., & Gilies, J. A. (2003). Photolysis of atrazine and ametryne herbicides in Barbados sugar cane plantation soils and water. Journal of Environmental Science and Health, Part B, 38(3), 293–303.
Meyer, A. H., Penning, H., & Elsner, M. (2009). C and N isotope fractionation suggests similar mechanisms of microbial atrazine transformation despite involvement of different enzymes (AtzA and TrzN). Environmental Science and Technology, 43(21), 8079–8085.
Navarro, S., Vela, N., Giménez, M. J., & Navarro, G. (2004). Persistence of four s-triazine herbicides in river, sea and groundwater samples exposed to sunlight and darkness under laboratory conditions. Science of the Total Environment, 329(1), 87–97.
Papadopoulou-Mourkidou, E., Karpouzas, D. G., Patsias, J., Kotopoulou, A., Milothridou, A., Kintzikoglou, K., & Vlachou, P. (2004). The potential of pesticides to contaminate the groundwater resources of the Axios river basin. Part II. Monitoring study in the south part of the basin. Science of the Total Environment, 321(1), 147–164.
Quan, X., Zhao, X., Chen, S., Zhao, H. M., Chen, J. W., & Zhao, Y. Z. (2005). Enhancement of p,p′-DDT photodegradation on soil surfaces using TiO2 induced by UV-light. Chemosphere, 60(2), 266–273.
Remucal, C. K. (2014). The role of indirect photochemical degradation in the environmental fate of pesticides: a review. Environmental Science: Processes and Impacts, 16(4), 628–653.
Seiber, J. N., & Kleinschmidt, L. A. (2011). Contributions of pesticide residue chemistry to improving food and environmental safety: past and present accomplishments and future challenges. Journal of Agricultural and Food Chemistry, 59(14), 7536–7543.
Si, Y., Zhou, J., Chen, H., Zhou, D. M., & Yue, Y. D. (2004). Effects of humic substances on photodegradation of bensulfuron-methyl on dry soil surfaces. Chemosphere, 56(10), 967–972.
Subramanian, B., Yang, Q. L., Yang, Q. J., Khodadoust, A. P., & Dionysiou, D. D. (2007). Photodegradation of pentachlorophenol in room temperature ionic liquids. Journal of Photochemistry and Photobiology, A, 192(2), 114–121.
Sun, X., Liu, H., Zhang, Y., Zhao, Y. Z., & Quan, X. (2011). Effects of Cu (II) and humic acid on atrazine photodegradation. Journal of Environmental Sciences (China), 23(5), 773–777.
Swann, R. L., Laskowski, D. A., McCall, P. J., & Kuy, K. V. (1983). A rapid method for the estimation of the environmental parameters of octanol/water partition coefficient, soil sorption constant, water to air ratio, and water solubility. Research Reviews, 85, 17–28.
Tajeddine, L., Nemmaoui, M., Mountacer, H., Dahchour, A., & Sarakha, M. (2010). Photodegradation of fenamiphos on the surface of clays and soils. Environmental Chemistry Letters, 8(2), 123–128.
Tercero Espinoza, L. A., Neamţu, M., & Frimmel, F. H. (2007). The effect of nitrate, Fe (III) and bicarbonate on the degradation of bisphenol A by simulated solar UV-irradiation. Water Research, 41(19), 4479–4487.
Thurman, E. M., Bastian, K. C., & Mollhagen, T. (2000). Occurrence of cotton herbicides and insecticides in playa lakes of the High Plains of West Texas. Science of the Total Environment, 248, 189–200.
Twomey, S. A., Bohren, C. F., & Mergenthaler, J. L. (1986). Reflectance and albedo differences between wet and dry surfaces. Applied Optics, 25(3), 431–437.
Vela, N., Navarro, G., Giménez, M. J., & Navarro, S. (2004). Gradual fall of s-triazine herbicides in drinking and wastewater samples as influenced by light and temperature. Water, Air, and Soil Pollution, 158(1), 3–19.
Wallace, D. F., Hand, L. H., & Oliver, R. G. (2010). The role of indirect photolysis in limiting the persistence of crop protection products in surface waters. Environmental Toxicology and Chemistry, 29(3), 575–581.
Wang, J., Chen, S., Quan, X., Zhao, H. M., & Zhao, Y. Z. (2006). Enhanced photodegradation of phenolic compounds by adding TiO2 to soil in a rotary reactor. Journal of Environmental Sciences (China), 18(6), 1107–1112.
Wang, H. Z., Zuo, H. G., Ding, Y. J., Miao, S. S., Jiang, C., & Yang, H. (2014). Biotic and abiotic degradation of pesticide Dufulin in soils. Environmental Science and Pollution Research, 21(6), 4331–4342.
Wauchope, R. D., Buttler, T. M., Hornsby, A. G., Augustijn-Beckers, P. M. W., & Burt, J. P. (1992). The SCS/ARS/CES pesticide properties database for environmental decision-making. Reviews of Environmental Contamination and Toxicology, 123, 1–164.
Zhang, L., Li, P., Gong, Z. Q., & Li, X. M. (2008). Photocatalytic degradation of polycyclic aromatic hydrocarbons on soil surfaces using TiO2 under UV light. Journal of Hazardous Materials, 158(2), 478–484.
Acknowledgements
The authors acknowledge the financial supports of the National Key Research and Development Project of China (no. 2016YFD0200201) and the Jiangsu Innovation Program for Graduate Education (KYLX15_0616).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
This information is available free of charge via the Internet at http://www.sciencedirect.com.
ESM 1
(DOC 761 kb)
Rights and permissions
About this article
Cite this article
Jiang, C., Li, X.J., Wang, Y.R. et al. Assessment of Photodegradation of Herbicide Prometryn in Soil. Water Air Soil Pollut 228, 135 (2017). https://doi.org/10.1007/s11270-017-3307-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3307-7