Abstract
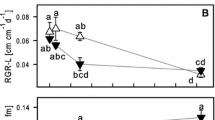
Laboratory experiments were carried out to determine the effects of exposure to different concentrations of As, Hg, Sb and Se on photosynthetic and respiratory rates and on photosynthetic efficiency in the aquatic bryophyte Fontinalis antipyretica Hedw. Specimens of the moss, collected from a clean site, were incubated in solutions of As, Hg, Sb and Se (at concentrations ranging from 0.1 μg l−1 to 10,000 μg l−1) for up to 22 days. The photosynthetic and respiratory rates were then determined by the light/dark bottle technique, and the photosynthetic efficiency was measured by the saturation pulse method. Although different responses were observed in relation to the concentration of the elements, clear responses in net photosynthesis and photosynthetic efficiency were generally only observed in the moss exposed to the highest concentrations of these elements in solution. Mercury was apparently the most toxic of the elements studied. Net photosynthesis and photosynthetic efficiency were also related to tissue concentrations of these elements in the moss. Despite the higher toxicity of Hg, this element can be accumulated at high concentrations in moss, probably at extracellular sites. For Sb, the same tissue concentration had very different physiological effects depending on the initial concentration to which the moss was exposed in solution. Temporal trends in chlorophyll fluorescence were more stable than trends in net photosynthesis. The respiratory rate was very variable and was not clearly related to the concentration of elements in solution or in moss tissues.






Similar content being viewed by others
References
Aronsson, K. A., & Ekelund, N. G. A. (2006). Effects on growth, photosynthesis and pigments of the freshwater moss Fontinalis antipyretica Hedw. after exposure to wood ash solution. Science of the Total Environment, 372, 236–246.
Babula, P., Adam, V., Opatrilova, R., Zehnalek, J., Havel, L., & Kizek, R. (2008). Uncommon heavy metals, metalloids and their plant toxicity: a review. Environmental Chemical Letters, 6, 189–213.
Bjorkman, O., & Demmig, B. (1987). Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. Planta, 170, 489–504.
Bolhàr-Nordenkampf, H. R., Long, S. P., Baker, N. R., Öquist, G., Schreiber, U., & Lechner, E. G. (1989). Chlorophyll fluorescence as a probe of the photosynthetic competence of leaves in the field: a review of current instrumentation. Functional Ecology, 3, 497–514.
Branquinho, C., Brown, D. H., & Catarino, F. (1997a). The cellular location of Cu in lichens and its effects on membrane integrity and chlorophyll fluorescence. Environmental and Experimental Botany, 38, 165–179.
Branquinho, C., Brown, D. H., Máguas, C., & Catarino, F. (1997b). Lead (Pb) uptake and its effects on membrane integrity and chlorophyll fluorescence in different lichen species. Environmental and Experimental Botany, 37, 95–105.
Brown, D. H., & Wells, J. M. (1990). Physiological effects of heavy metals on the moss Rhytidiadelphus squarrosus. Annals of Botany, 66, 641–647.
Brown, D. H., & Whitehead, A. (1986). The effect of mercury on the physiology of Rhytidiadelphus squarrosus (Hedw.) Warnst. Journal of Bryology, 14, 367–374.
Bruns, I., Friese, K., Markert, B., & Krauss, G.-J. (1997). The use of Fontinalis antipyretica L. ex Hedw. as a bioindicator for heavy metals. 2. Heavy metal accumulation and physiological reaction of Fontinalis antipyretica L. ex Hedw. in active biomonitoring in the River Elbe. Science of the Total Environment, 204, 161–176.
Butler, W., & Kitajima, M. (1975). Fluorescence quenching in photosystem II of chloroplasts. Biochimical & Biophysical Acta, 376, 116–125.
Carballeira, A., Díaz, S., Vázquez, M. D., & López, J. (1998). Inertia and resilience in the responses of the aquatic bryophyte Fontinalis antipyretica Hedw. to thermal stress. Archives of Environmental Contamination and Toxicology, 34, 343–349.
Carginale, V., Sorbo, S., Capasso, C., Trinchella, F., Cafiero, G., & Basile, A. (2004). Accumulation, localisation, and toxic effects of cadmium in the liverwort Lunularia cruciata. Protoplasma, 223, 53–61.
Carter, L. F., & Porter, S. D. (1997). Trace-element accumulation by Hygrohypnum ochraceum in the upper Rio Grande basin, Colorado and New Mexico, USA. Environmental Toxicology and Chemistry, 16(12), 2521–2528.
Cenci, R. M. (2000). The use of aquatic moss (Fontinalis antipyretica) as monitor of contamination in standing and running waters: limits and advantages. Journal of Limnology, 60(suppl. 1), 53–61.
Cesa, M., Bizzotto, A., Ferraro, C., Fumagalli, F., & Nimis, P. L. (2009). S.TR.E.A.M., system for trace element assessment with mosses. An equation to estimate mercury concentration in freshwaters. Chemosphere, 75, 858–865.
Cesa, M., Bizzoto, A., Ferraro, C., Fumagalli, F., & Nimis, P. L. (2010). Palladio, an index of trace element alteration for the River Bacchiglione based on Rhynchostegium riparioides. Water, Air, and Soil Pollution, 208, 59–77.
Connell, D. W., Lam, P., Richardson, B., & Wu, R. (1999). Introduction to ecotoxicology. Oxford: Blackwell Science.
Cruz de Carvalho, R., Branquinho, C., & Marques da Silva, J. (2011). Physiological consequences of desiccation in the aquatic bryophyte Fontinalis antipyretica. Planta, 234, 195–205.
Csintalan, Z., Proctor, M. C. F., & Tuba, Z. (1999). Chlorophyll fluorescence during drying and rehydration in the mosses Rhytidiadelphus loreus (Hedw.) Warnst., Anomodon viticulosus (Hedw.) Hook. & Tayl. and Grimmia pulvinata (Hedw.) Sm. Annals of Botanny, 84, 235–244.
Culioli, J.-L., Fouquoire, A., Calendini, S., Mori, C., & Orsini, A. (2009). Trophic transfer of arsenic and antimony in a freshwater ecosystem: a field study. Aquatic Toxicolgy, 94, 286–293.
Díaz, S., Villares, R., & Carballeira, A. (2012). Uptake Kinetics of As, Hg, Sb, and Se in the aquatic moss Fontinalis antipyretica Hedw. Water, Air, and Soil Pollution, 223, 3409–3423.
Drasch, G., Horvat, M., & Stoeppler, M. (2004). Mercury. In E. Merian, M. Anke, M. Ihnat, & M. Stoeppler (Eds.), Elements and their compounds in the environment (2nd ed., pp. 931–1005). Weinheim: Wiley-VCH.
Empain, A. (1973). Les bryophytes aquatiques utilisés comme traceurs de la contamination en métaux lourds des eaux douces. Memoires de la Societe Royale Botanique de Belgique, 7, 141–156.
Esteban, E., Moreno, E., Peñalosa, J., Cabrero, J., Millán, R., & Zornoza, P. (2008). Short and long-term uptake of Hg in white lupin plants: kinetics and stress indicators. Environmental and Experimental Botany, 62, 316–322.
Fernández, J. A., Vázquez, M. D., López, J., & Carballeira, A. (2006). Modelling the extra and intracellular uptake and discharge of heavy metals in Fontinalis antipyretica transplanted along a heavy metal and pH contamination gradient. Environmental Pollution, 139, 21–31.
Fracheboud, Y., & Leipner, J. (2003). The application of chlorophyll fluorescence to study light, temperature, and drought stress. In J. R. DeEll & P. M. A. Toivonen (Eds.), Practical Applications of Chlorophyll Fluorescence in Plant Biology (pp. 125–150). Dordrecht: Kluwer Academic Publishers.
Fu, Z., Wu, F., Amarasiriwardena, D., Mo, C., Liu, B., Zhu, J., et al. (2010). Antimony, arsenic and mercury in the aquatic environment and fish in a large antimony mining area in Hunan, China. Science of the Total Environment, 408, 3403–3410.
Gapeeva, M. V., Dolotov, A. V., & Chemeris, E. V. (2010). Prospects of using mosses (Fontinalis antipyretica Hedw. and Pylaisia polyantha (Hedw.) Bruch et al.) as indicators of environmental contamination with heavy metals. Russian Journal of Ecology, 41(1), 28–31.
Israr, M., Sahi, S., Datta, R., & Sarkar, D. (2006). Bioaccumulation and physiological effects of mercury in Sesbania drummondii. Chemosphere, 65, 591–598.
Kanamori, S., & Sugawara, K. (1965). Geochemical study of arsenic in natural waters. I. Arsenic in rain and snow. Journal of Earth Science, Nagoya University, 13, 23–35.
Koch, I., Feldmann, J., Wang, L., Andrewes, P., Reimer, K. J., & Cullen, W. R. (1999). Arsenic in the Meager Creek hot springs environment, British Columbia, Canada. Science of the Total Environment, 236, 101–117.
Krause, G. H., & Weis, E. (1991). Chlorophyll fluorescence and photosynthesis—the basics. Annual Review of Plant Physiology and Plant Molecular Biology, 42, 313–349.
Lichtenthaler, H. K., & Miehé, J. A. (1997). Fluorescence imaging as a diagnostic tool for plant stress. Trends in Plant Science, 2, 316–320.
López, J., & Carballeira, A. (1993). Interspecific differences in metal bioaccumulation and plant–water concentration ratios in five aquatic bryophytes. Hydrobiologia, 263, 95–107.
Martínez-Abaigar, J., Núñez-Olivera, E., & Sánchez-Díaz, M. (1993). Effects of organic pollution on transplanted aquatic bryophytes. Journal of Bryology, 17, 553–566.
Maxwell, K., & Johnson, G. N. (2000). Chlorophyll fluorescence: a practical guide. Journal of Experimental Botany, 51, 659–668.
Miyashita, S., Shimoya, M., Kamidate, Y., Kuroiwa, T., Shikino, O., Fujiwara, S., et al. (2009). Rapid determination of arsenic species in freshwater organisms from the arsenic-rich Hayakawa River in Japan using HPLC-ICP-MS. Chemosphere, 75, 1065–1073.
Moreno-Jiménez, E., Esteban, E., Carpena-Ruiz, R. O., & Peñalosa, J. M. (2009). Arsenic and mercury induced phytotoxicity in the Mediterranean shrubs Pistacia lentiscus and Tamarix gallica grown in hydroponic culture. Ecotoxicology and Environmental Safety, 72, 1781–1789.
Nieboer, E., & Richardson, H. S. (1980). The replacement of the nondescript term “heavy metals” by a biologically and chemically significant classification of metal ions. Environmental Pollution, 1, 3–26.
Nimis, P. L., Fumagalli, F., Bizzotto, A., Codogno, M., & Skert, N. (2002). Bryophytes as indicators of trace metals pollution in the River Brenta (NE Italy). Science of the Total Environment, 286, 233–242.
Pekka, L., Halmeenpaa, H., Ecke, F., Vuori, K.-M., Mokrotovarova, O., Ohlander, B., et al. (2008). Assessing pollution in the Kola River, northwestern Russia, using metal concentrations in water and bryophytes. Boreal Environmental Research, 13, 15–30.
Pérez-Llamazares, A., Fernández, J. A., Aboal, J. R., & Carballeira, A. (2009). A search for an extracellular extractant of Hg for use in the sequential elution technique with Pseudoscleropodium purum. Journal of Bryology, 31, 23–29.
Pérez-Llamazares, A., Aboal, J. R., Fernández, J. A., & Carballeira, A. (2011a). Sequential elution technique in moss Pseudoscleropodium purum: comparison between the commonly used extracellular extractant NiCl2 and other new extractants. Water, Air, and Soil Pollution, 215, 561–572.
Pérez-Llamazares, A., Fernández, J. A., Carballeira, A., & Aboal, J. R. (2011b). The sequential elution technique applied to cryptogams: a literature review. Journal of Bryology, 33, 267–278.
Rau, S., Miersch, J., Neumann, D., Weber, E., & Krauss, G.-J. (2007). Biochemical responses of the aquatic moss Fontinalis antipyretica to Cd, Cu Pb and Zn determined by chlorophyll fluorescence and protein levels. Environmental and Experimental Botany, 59, 299–306.
Rish, M. A. (2004). Antimony. In E. Merian, M. Anke, M. Ihnat, & M. Stoeppler (Eds.), Elements and their compounds in the environment (2nd ed., pp. 659–670). Weinheim: Wiley-VCH.
Roeck, U., Glasser, N., & Trémolières, M. (1995). Seasonal variations in mercury accumulation by the aquatic moss Fontinalis antipyretica Hedw. Acta Botanica Gallica, 142(6), 741–749.
Samecka-Cymerman, A., & Kempers, A. J. (1995). Preliminary investigations into the bioaccumulation of mercury by the liverwort Scapania undulata L. (Dum). Ecotoxicology and Environmental Safety, 31, 57–61.
Schrauzer, G. N. (2004). Selenium. In E. Merian, M. Anke, M. Ihnat, & M. Stoeppler (Eds.), Elements and their compounds in the environment (2nd ed., pp. 1365–1406). Weinheim: Wiley-VCH.
Schreiber, U., Bilger, W., & Neubauer, C. (1995). Chlorophyll fluorescence as a nonintrusive indicator for rapid assessment of in vivo photosynthesis. In E.-D. Schulze & M. M. Caldwell (Eds.), Ecophysiology of Photosynthesis (pp. 49–70). Berlin: Springer.
SEPA (Swedish Environmental Protection Agency). (2000). Environmental quality criteria. Lakes and watercourses. Report 5050. Swedish Environmental Protection Agency.
Sidhu, M., & Brown, D. H. (1996). A new laboratory technique for studying the effects of heavy metals on bryophyte growth. Annals of Botany, 78, 711–717.
Sommer, V. C., & Winkler, S. (1982). Reaktionen im gaswechsel von Fontinalis antipyretica Hedw. nach experimentellen belastungen mit schwermetallverbindungen. Archiv für Hydrobiologie, 4, 503–524.
Stoeppler, M. (2004). Arsenic. In E. Merian, M. Anke, M. Ihnat, & M. Stoeppler (Eds.), Elements and their compounds in the environment (2nd ed., pp. 1321–1364). Weinheim: Wiley-VCH.
Vázquez, M. D., López, J., & Carballeira, A. (1999). Uptake of heavy metals to the extracellular and intracellular compartments in three species of aquatic bryophyte. Ecotoxicology and Environmental Safety, 44, 12–24.
Vázquez, M. D., Villares, R., & Carballeira, A. (2013). Biomonitoring urban fluvial contamination on the basis of physiological stress induced in transplants of the aquatic moss Fontinalis antipyretica Hedw. Hydrobiologia, 707, 97–108.
Vieira, A. R., Gonzalez, C., Martins-Louçao, M. A., & Branquinho, C. (2009). Intracellular and extracellular ammonium (NH4 +) uptake and its toxic effects on the aquatic biomonitor Fontinalis antipyretica. Ecotoxicology, 18, 1087–1094.
Wehr, J. D., & Whitton, B. A. (1983). Accumulation of heavy metals by aquatic mosses. 2. Rhynchostegium riparioides. Hydrobiologia, 100, 261–284.
Wells, J. M., & Brown, D. H. (1990). Ionic control of intracellular and extracellular Cd uptake by the moss Rhytidiadelphus squarrosus (Hedw.) Warnst. New Phytologist, 116, 541–553.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Díaz, S., Villares, R., Vázquez, M.D. et al. Physiological Effects of Exposure to Arsenic, Mercury, Antimony and Selenium in the Aquatic Moss Fontinalis antipyretica Hedw.. Water Air Soil Pollut 224, 1659 (2013). https://doi.org/10.1007/s11270-013-1659-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-013-1659-1