Abstract
Plant invasions have a negative impact on biodiversity and lead to species loss. Although studies on traits promoting invasiveness have been largely developed, comparative studies on germination traits of closely related native versus alien invasive species, which also take into account threatened species, are lacking. We analysed the germination requirements of two congeneric Lindernia species inhabiting paddy field in Northern Italy. Germination tests were conducted exposing seeds of both species across a broad range of temperature and conditions (i.e., cold-wet stratification, dark condition, alternate temperature, gibberellic acid). The invasive L. dubia showed higher and faster germination, occurring over a broader range of temperatures compared to the native threatened L. procumbens, along with a lighter non-deep physiological dormancy. These results show a competitive advantage of the alien species already in the early stages of plant regeneration and provide a comprehensive overview of the germination requirements, necessary for the development of future conservation and management plans.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite efforts to halt invasions, invasive species affect negatively the richness, the abundance and the functional diversity of native communities (Vilà et al. 2011), increasing the extinction risk of native species. Traits promoting invasiveness include high growth rate and specific leaf area (Kaushik et al. 2022), long and early flowering periods (Pyšek and Richardson 2008; Colautti et al. 2006). In addition, seed traits may strongly contribute to invasion in the new region, with alien species showing high seed dispersal over a broader range of environmental conditions (Pyšek and Richardson 2008) and larger soil seedbanks compared to natives (Gioria and Pyšek 2016). Comparative germination studies of closely related native and invasive species contribute to identifying traits related with invasiveness since this approach minimizes biases associated with phylogenetic distance and habitat affinities (Gioria and Pyšek 2017). Nevertheless, when it counts for threatened species in view of plant conservation, a few studies investigated differences in germination traits, mostly reporting contrasting results (e.g., Paulů et al. 2017).
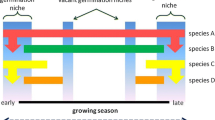
In our study we have investigated the germination requirement of the congeneric species Lindernia procumbens (Krock.) Philcox and L. dubia (L.) Pennell [Linderniaceae]. L. procumbens is native to the temperate to tropical zones of Eurasia, included in the Annex IV of the 92/43/EEC “Habitat” Directive and Annex I of Bern Convention. The species is classified as “Least Concern” (Bilz et al. 2011) on the European Red List but is locally threatened, being included in several national Red Lists due to its scattered distribution, the fragmented habitat and the intense agricultural activities that include the use of chemicals. Last but not least, the species suffer a competitive disadvantage with the invasive congeneric L. dubia (L.) Pennell, a North American species native to catchments of the Mississippi River (Šumberová et al. 2012), accidentally introduced in France in 1850 by ships on the Loire river banks and further spread through Europe (Simons and Jansen 2018).The life forms and ecological preferences of the two species are similar. They are short-lived annuals, reproduce exclusively by seeds and co-occur in periodically flooded habitats such as river floodplains, wet meadows and paddy fields. Previous studies stated that L. dubia displays a robust growth, being able to colonize a broader range of vegetation types and to reproduce over longer time during the growing season compared to L. procumbens (Šumberová et al. 2012). In our study we investigated the germination requirement of both species to deepen the information on 1) seed germination across a broad range of temperature and conditions (i.e., cold-wet stratification, dark condition, alternate temperature) 2) the presence of physiological dormancy. We hypothesised that seeds of L. dubia show higher and faster germination, wider temperature range and are less dormant compared to L. procumbens. By shading light on the regeneration niche of both species we hope to contribute to better-informed management decisions of the invasive species and to the development of conservation plan for the threatened species.
Material and methods
Germination tests
Considering the limited occurrence and scarce data availability of L. procumbens distribution, seeds of both species were collected in July 2019 in a paddy field (i.e., one population) where both species co-occur (Pavia Province, N-Italy). Seeds were manually cleaned and stored in the Seedbank of the University of Pavia under dry room conditions (15°CC and 15% RH) for 3 monthsmonths in darkness to preserve their viability until germination tests started (i.e., on 30th October 2019). Both fresh and cold-wet stratified seeds (at 0°CC—- dark condition for three months) were exposed to five constant temperatures (10, 15, 20, 25, 30°CC) with 12/12 dayday/night photoperiod; fresh seeds were also exposed to alternate temperature regime (i.e., 5/15 and 15/25°CC), with light provided at higher temperature. Germination tests were conducted sowing three replicates of 20 seeds each on 1% distilled water agar held in 60 mm diameter Petri dishes using temperature and light controlled condition (photosynthetically active radiation 40–50 μmolμmol mm − 2 ss − 1). Germination plates were checked for radicle emergence (> 1 mm) daily in the first week, then at weekly interval until germination stopped (i.e., no emergence after 6 weeks). Once cotyledon emergence was completed, seedlings were removed from the Petri dishes. At the end of the experiments, non-germinated seeds were cut-tested and mouldy and empty seeds were counted as inviable to calculate final germination percentage (FGP).
On 19th February 2020, further tests were conducted exposing seeds to gibberellic acid (250 mg/l, GA3) and at dark condition at the optimal germination temperature previously found (30 °C). Sowing method and photoperiod were the same as described above.
Data analysis
The effect of temperature and cold-wet stratification on seed germination was investigated using a generalised linear mixed model (glmer) with binomial error structure and logit link function where temperature (5 levels), species (2 levels), stratification (2 levels: i.e., presence/absence) and their interactions were tested against seed germination (model a, Table 1). Similarly, the effects of alternate vs. constant temperatures (2 levels) on germination was analysed at high (i.e., 15/25 vs 20 °C) and low (i.e., 5/15 vs 10 °C) temperature with species as a fixed factor (model b and c, respectively; Table 1). Replicates were considered as random factor. The effect of GA3 (2 levels, i.e., application/no application) was analysed using species as fixed factor (model d; Table 1). The effect of dark condition was not analysed since darkness inhibited seeds germination. In all models, the interaction and fixed factor combination was chosen taking into account the Akaike information criterion (AIC; Bozdogan 1987); models assumptions were tested with the Dharma package (Hartig and Lohse 2022). Finally, Mean Germination Time (hereafter, MGT), was calculated as follow:
where n is the number of seeds that germinated at time T, T are the days between the beginning of the test and the measurement and N is the total number of seeds that germinated. Differences in MGT between species were assessed for the germination experiments with a minimum FGP of 10%, where the variable species was considered as a fixed factor (model e; Table 1). Statistical analyses were performed with R 3.3.0 (R Core Team 2022).
Results
Both species germinated at the highest temperature (30 °C) without cold-wet stratification. L. dubia reported a significantly higher germination (81%) compared to L. procumbens (14%) at its optimum (Table 1). While seeds of L. procumbens germinated only at 30 °C, L. dubia germinated over a wider temperature range (from 15 to 30 °C; Fig. 1A).
Cold-wet stratification overall increased seed germination. However, it decreased seeds germination at the warmer temperatures (25, 30 °C), while increasing it on both species at cooler conditions (15, 20 °C) (Table 1a, Fig. 1B). Darkness completely inhibited seeds germination of both species. Alternate low temperature (5/15 °C) promoted germination, but only in L. dubia (38% FGP). L. dubia FGP was lower, but not significantly, at high alternate temperature (15/25 °C) than at constant temperature (20 °C; 27% vs 36%, respectively) while L. procumbens FGP was significantly lower (0 and 2% FGP both at constant and alternate temperature, respectively; Table 1b and Table 1c). Seeds germination was significantly improved by GA3, particularly in L. dubia (Table 1d, Fig. 1C). Because of the low germination of L. procumbens (< 10% in most experiments), differences in MGT were estimated at 30 °C. L. dubia germinated on average one day before L. procumbens, requiring 3 days, but this difference was only marginally significative (Table 1e).
Discussion
Germination behaviour of plants influences their fitness, persistence, evolutionary potential and the biotic environment, with differences between native and non-native congeners representing a mechanism that has contributed to the naturalization and invasiveness of many alien species (Gioria and Pyšek 2017). According with the hypothesis tested, the invasive L. dubia showed higher germination, occurring over a broader range of temperatures compared to the native threatened L. procumbens. These results echo previous studies investigating germination traits associated with invasiveness (e.g., Colautti et al. 2006; Pyšek and Richardson 2008; Gioria and Pyšek 2016; Gioria and Pyšek 2017).
Warm temperatures are capable of promoting the success of invasion by increasing the probability of germination of invasive species compared to native species (Trotta et al. 2023). However, the results observed in our experiment contrast with this general pattern, but echoes previously observed germination behaviours in Lindernia (see Hrivnák et al. 2016), with both species being defined as thermophilic (Nobis et al. 2010). Furthermore, our study confirmed the traits associated with the invasion success of L. dubia, displaying a more robust growth, ability to colonize a broader range of wetland vegetation types (i.e., the phytosociological classes of Littorelletea uniflorae, Phragmitetea, Bidentetea tripartitae and Isoëto-Nanojuncetea, with the latter shared with L. procumbens; Simons and Jansen 2018) and reproducing over longer time during the growing season compared to L. procumbens (Šumberová et al. 2012). These traits can also be explained by the different size and reproductive potential of the species, with L. dubia being usually bigger and producing more flowers and fruits than L. procumbens (Šumberová et al. 2012). Our results also highlight typical traits of invasiveness related to fast mean germination time, which facilitate access to resources and niches in the environment (Gioria and Pyšek 2017). Early germination is indeed a major factor in the success of several invasive species (Laube et al. 2015), demonstrating that competition mitigation or avoidance through the exploitation of vacant germination niches is a better strategy than superior competitive ability in the colonization of habitats where the species coexist (Keller and Shea 2021). At the same time, the delayed germination of L. procumbens, together with the limited germination cues, could be a strategy to reduce mortality risk associated with both unfavourable post-germination conditions and the effects of inter- and intra-specific competition among neighbouring species (Donohue et al. 2010).
Cold-wet stratification promoted the germination of both species over a wider temperature spectrum, albeit at lower germination percentage, while dark condition did not have any relevant effect on seeds germination Interestingly, while the warm alternate temperature (i.e. 25/15°C) does not affect the seed germination of the two species, the cold alternate temperature (15/5°C) improves germination compared to the constant associated regime (10°C) only for the invasive species. This result suggests that the germination of the invasive L. dubia may occur earlier in the vegetative season than that of native L. procumbens, thus (once again) providing a competitive advantage over space and nutrients resources. . Seeds germination was improved by GA3, suggesting a non-deep physiological dormancy in both species (Baskin and Baskin 2014). A certain proportion of fresh seeds may thus enter in primary or secondary dormancy to prevent the germination of all dispersed seeds, leading to the formation of a soil seed bank (Fenesi et al. 2014). The formation of a persistent seed bank is considered more important for the persistence of annual than perennial species (Gioria et al. 2020), with a short life cycle being often associated with naturalisation and invasiveness (Funk et al. 2016; Pyšek et al. 2017), as for L. dubia. Although L. procumbens have been demonstrated to display a large soil seed bank (Šumberová et al. 2012), its sensitivity to warm temperature outlines a fluctuation in the size of population strictly related with the climate. However, the increased temperature foreseen by climate change in the upcoming years, jointly with the increased frequency of heat waves, could determine an increase of the population size of the species (Nobis et al. 2010); further studies are necessary to better understand L. procumbens population dynamic.
By providing a comprehensive overview of the germination requirement of L. procumbens we hope to contribute to a conservation plan for this threatened species that is currently not available in Italy and also mandatory at EU level according to the Habitat Directive. Conservation activities for the species could be applied easily in agroecosystems (i.e., organic paddy fields), where the species is repeatedly found. However, specific measures and protocols must be developed considering both the short life-cycle and the size of the seeds, which are dust-like, that can hardly envisage the cultivation in nursery and the subsequent translocation through planned interventions.
Data availability
Data analyzed during this study are available from the corresponding author upon reasonable request.
References
Bilz M, Kell SP, Maxted N, Lansdown RV (2011) European Red List of Vascular Plants. Publications Office of the European Union, Luxembourg
Bozdogan H (1987) Model selection and Akaike’’s Information Criterion (AIC): The general theory and its analytical extensions. Psychometrika 52:345–370. https://doi.org/10.1007/BF02294361
Donohue K, Rubio de Casas R, Burghardt L, Kovach K, Willis CG (2010) Germination, post-germination adaptation, and species ecological ranges. Annu Rev Ecol Evol Syst 41:293–319. https://doi.org/10.1146/annurev-ecolsys-102209-144715
Fenesi A, Albert Á-J, Ruprecht E (2014) Fine-tuned ability to predict future competitive environment in Ambrosia artemisiifolia seeds. Weed Res 54:58–69. https://doi.org/10.1111/wre.12048
Funk JL, Standish RJ, Stock WD, Valladares F (2016) Plant functional traits of dominant native and invasive species in Mediterranean-climate ecosystems. Ecology 97:75–83. https://doi.org/10.1890/15-0974.1
Gioria M, Pyšek P (2016) The legacy of plant invasions: changes in the soil seed bank of invaded plant communities. Bioscience 66:40–53. https://doi.org/10.1093/biosci/biv165
Gioria M, Pyšek P (2017) Early bird catches the worm: germination as a critical step in plant invasion. Biol Invasions 19:1055–1080. https://doi.org/10.1007/s10530-016-1349-1
Gioria M, Pyšek P, Baskin CC, Carta A (2020) Phylogenetic relatedness mediates persistence and density of soil seed banks. J Ecol 108:2121–2131. https://doi.org/10.1111/1365-2745.13437
Hartig F, Lohse L 2022 DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models. https://CRAN.R-project.org/package=DHARMa Accessed 14 February 2024
Hrivnák R, Kochjarová J, Šumberová K, Schmotzer A (2016) Alien wetland annual Lindernia dubia (Scrophulariaceae): the first recently mentioned localities in Slovakia and their central European context. Biologia 71:281–286. https://doi.org/10.1515/biolog-2016-0039
Kaushik P, Kumar Pati P, Khan ML, Khare PK (2022) Plant functional traits best explain invasive species’’ performance within a dynamic ecosystem - A review. Trees for People 8:100260. https://doi.org/10.1016/j.tfp.2022.100260
Keller JA, Shea K (2021) Warming and shifting phenology accelerate an invasive plant life cycle. Ecology 102:e03219. https://doi.org/10.1002/ecy.3219
Laube J, Sparks TH, Bässler C, Menzel A (2015) Small differences in seasonal and thermal niches influence elevational limits of native and invasive Balsams. Biol Conserv 191:682–691. https://doi.org/10.1016/j.biocon.2015.08.019
Marushia RG, Cadotte MW, Holt JS (2010) Phenology as a basis for management of exotic annual plants in desert invasions. J Appl Ecol 47:1290–1299. https://doi.org/10.1111/j.1365-2664.2010.01881.x
Nobis A, Nobis M, Piotrowicz K, Kącki Z, Dajdok Z (2010) Lindernia procumbens in Poland: the relationship between weather conditions and the occurrence of the species. Biodiv Res Conserv 17:39–46. https://doi.org/10.2478/v10119-010-0005-3
Paulů A, Harčariková L, Münzbergov Z (2017) Are there systematic differences in germination between rare and common species? A case study from central European mountains. Flora 236–237:15–24. https://doi.org/10.1016/j.flora.2017.09.004
Pyšek P, Richardson DM (2008) Traits associated with invasiveness in alien plants: where do we stand? In: Nentwig W (ed) Biological invasions, Ecological Studies. Springer, Berlin, pp 97–125
Pyšek P, Pergl J, Essl F, Lenzner B, Dawson W, Kreft H, Weigelt P, Winter M, Kartesz J, Nishino M, Antonova LA, Barcelona JF, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castańo N, Chacón E, Chatelain C, Dullinger S, Ebel AL, Figueiredo E, Fuentes N, Genovesi P, Groom QJ, Henderson L, Inderjit, Kupriyanov A, Masciadri S, Maurel N, Meerman J, Morozova O, Moser D, Nickrent D, Nowak PM, Pagad S, Patzelt A, Pelser PB, Seebens H, Shu W, Thomas J, Velayos M, Weber E, Wieringa JJ, Baptiste MP, van Kleunen M (2017) Naturalized alien flora of the world: species diversity, taxonomic and phylogenetic patterns, geographic distribution and global hotspots of plant invasion. Preslia 89:203–274. https://doi.org/10.23855/preslia.2017.203
Pyšek P, Hulme PE, Simberloff D, Bacher S, Blackburn TM, Carlton JT, Dawson W, Essl F, Foxcroft LC, Genovesi P, Jeschke JM, Kühn I, Liebhold AM, Mandrak NE, Meyerson LA, Pauchard A, Pergl J, Roy HE, Seebens H, van Kleunen M, Vilà M, Wingfield MJ, Richardson DM (2020) Scientists’’ warning on invasive alien species. Biol Rev 95:1511–1534. https://doi.org/10.1111/brv.12627
R Core Team (2022) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria (https://www.R-project.org/)
Simons ELAN, Jansen MGM (2018) Ecology of naturalized invasive species Lindernia dubia (L.). Pennell in the Netherlands Gorteria 40:1–10
Šumberová K, Lososová Z, Ducháček M (2012) Distribution, habitat ecology, soil seed bank and seed dispersal of threatened Lindernia procumbens and alien Lindernia dubia (Antirrhinaceae) in the Czech Republic. Phyton 52:39–72
Trotta G, Vuerich M, Petrussa E, Hay FR, Assolari S, Boscutti F (2023) Germination performance of alien and native species could shape community assembly of temperate grasslands under different temperature scenarios. Plant Ecol 224:1097–1111. https://doi.org/10.1007/s11258-023-01365-7
Vilà M, Espinar JL, Hejda M, Hulme PE, Jarošik V, Maron JL, Pergl J, Schaffner U, Sun Y, Pyšek P (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14:702–708. https://doi.org/10.1111/j.1461-0248.2011.01628.x
Acknowledgements
We deeply thank M.Sc. students Tommaso de Pascale, Micol Orengo and Silvia Bonzani that participated in this study and collected germination data.
Funding
Open access funding provided by Università degli Studi di Pavia within the CRUI-CARE Agreement. The authors AC, SO and AM were funded by the “National Biodiversity Future Center—NBFC”—National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4—Call for tender No. 3138 of 16 December 2021, rectified by Decree n.3175 of 18 December 2021 of Italian Ministry of University and Research funded by the European Union—Next Generation EU, Project code CN_00000033.
Author information
Authors and Affiliations
Contributions
SO, FP, AM contributed to the study conception and design. FP and SL collected the data. AC and FP supervised and performed the statistical analysis. AC wrote the first draft of the manuscript. All authors contributed substantially to the revisions and approval of the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Jaime Moyano.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Corli, A., Orsenigo, S., Porro, F. et al. Germination niche of co-occurring threatened native and alien species: a case study in Lindernia procumbens and L. dubia. Plant Ecol 225, 725–729 (2024). https://doi.org/10.1007/s11258-024-01430-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-024-01430-9