Abstract
Climatic niches are key factors driving global and regional species distributions. The Atlantic Forest domain is considered one of the most threatened biomes in the world, and one of the main centres of plant diversity and endemism in the Neotropics. Of the over 13,000 species of vascular plants, nearly 15% are vascular epiphytes. Here we analysed for the first time how current epiphyte niches will be affected under future climate projections (SSP126 and SSP585) within 1.5 million km2 of Atlantic Forest in South America. Using the largest database of vascular epiphytes to date (n = 1521 species; n = 75,599 occurrence records) and ordination models, we found that the Atlantic Forest is expected to become warmer and drier and that up to 304 epiphyte species (20%) will have their average niche positions displaced outside the available climate space by the years 2040–2100. The findings from this study can help to inform ongoing legislative conservation efforts in one of the world’s most biodiverse regions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change is already causing species range shifts, local species loss and mass extinction events globally (IPCC 2022). Changes in temperature and precipitation has caused species to shift to areas of more suitable conditions, consequently altering their distributions, both locally and across continental scales (Bellard 2012; Pecl et al. 2017). However, species that have restricted distributions and/or greater sensitivity to environmental disturbances may not find new habitats that are suitable for them in the future, thus becoming more vulnerable and are at greater risk of extinction (Ruegg et al. 2021). In particular, stochastic and local disturbance events, such as droughts, disease outbreaks, and land use changes, may accelerate the extinction of species with smaller distribution ranges (Thuiller et al. 2005; Lawton 1995). In both cases – range shift and extinction – can lead to changes in the composition of local and regional communities, and might result in generalists and more tolerant species numbers to increase compared to more sensitive species (Baselga 2010; Spaak et al. 2017; Hidasi-neto et al. 2019; Mota et al. 2022). Climate change may thus impact not only individual species or groups of organisms, but also alter ecological functions with ecosystems (Galetti et al. 2013). Understanding species responses to changes in climate is thus crucial for building effective preventive measures and support conservation efforts (Stanton et al. 2014).
The use of climatic niche-based approaches has been shown to be a suitable strategy to assess species’ response to climate change (Pie et al. 2022). Climatic niches are the set of climatic conditions linked to the occurrence of a species. The focus on a subgroup of climatic variables, instead of assessing all biotic and abiotic dimensions, in an n-dimensional hypervolume of Hutchinson’s ecological space (Hutchinson 1957), makes it both a practical and heuristic approach (Pie et al. 2022). Climatic niche studies also provides an intersection of many diverse fields, including ecophysiology, invasion biology, biogeography, and macroecology (Petitpierre et al. 2012; Khaliq et al. 2015; Wasof et al. 2015; Atwater et al. 2018). Moreover, it is a key component of phylogenetic niche conservatism (Wiens and Donoghue 2004), which has been pivotal to understand the biogeographical distributions of many species, as well as the origin of latitudinal gradients of species diversity (e.g., Buckley et al. 2010; Kerkhoff et al. 2014).
The South American Atlantic Forest (AF) is considered one of the most endangered ecosystems in the world (Myers et al. 2000; Mittermeier et al. 2004); with nearly 95% of forest occurring in eastern Brazil and the remaining 5% occurring in Argentina and Paraguay (Marques et al. 2021). The AF covers an area of 1.5 million km2. However, only 12% of the original cover remains intact, with the remaining forest being fragmented (Ribeiro et al. 2009). Nevertheless, the mosaic of remaining native forest (incl. non-forest ecosystems) harbours over 16,000 species of land plants (Forzza et al. 2012). The biological diversity of this domain is believed to be driven by the regions geographic, geologic, and climatic heterogeneity; where it presents great latitudinal (3°S–30°S), altitudinal (0–2,800 m a.s.l.), and rainfall gradients. The latter is characterized by distinct seasonality in continental interior formations (~ 1,000 mm/year during the wet season), and higher precipitation in coastal forests (4,000 mm/year) (Câmara 2003). This heterogeneity has resulted in the establishment of both forests and open vegetation within the AF (Stehmann et al. 2009). In addition, approximately 47% of the plant species found in the AF are endemic to the domain (Forzza et al. 2012). The high level of endemism is associated with the isolation of wetter vegetations by dry open formations (e.g. Caatinga, Cerrado, and Chaco) from the Amazon, the Easter Andean forests and the Choco region in Colombia’s Pacific Coast (Zanella 2011), to the major humid forest formations in South America.
Vascular epiphytes, which are mechanically dependent plants that lack connection to the ground (Zotz 2016), and in some cases obtain their nutrients from atmospheric sources (Benzing 1990), are a conspicuous group of plants in the AF. Within the AF, vascular epiphytes account for over 2,000 species (Ramos et al. 2019), of which 78% are endemic to the domain (Freitas et al. 2016). Families such as Orchidaceae (79%), Gesneriaceae (89%), and Bromeliaceae (91%) show a particular high degree of endemism (Ramos et al. 2021). Because of their tight coupling to atmospheric conditions, epiphytes are believed to be particularly vulnerable to climate change (Zotz and Bader 2008). For example, it has been shown that sensitive epiphytes species in other parts of the world are more geographically restricted and their distribution is believed to be stronger related to changes in temperature (Hsu et al. 2012). It has also been suggested that epiphytes are more vulnerable than terrestrial species in the future (Reyes et al. 2021). Despite that, little is known about the effect of climate change on vascular epiphytes in the AF and across large geographical scales.
Therefore, this study aims a) to use high spatial resolution data of temperature and precipitation to describe the structure of the available climatic conditions across the AF; b) to determine the extent to which the available climatic space is occupied by different vascular epiphytes; c) to assess which regions of the climatic space will be most/least affected under different climate change scenarios; and d) to quantify which group of vascular epiphytes are under greater risk of niche displacement in the future. The findings of this work will support current legally binding conservation efforts (incl. the Brazilian National Atlantic Forest Law), to better understand current and future species distributions of one of the most diverse plant groups in the AF.
Materials and methods
Occurrence data
Vascular epiphyte occurrence data were extracted from Ramos et al. (2019) and de Araujo and Ramos (2021). These datasets comprised 2,095 species of structural dependent plants from 79,464 individual records (Fig. 1). The original data was compiled from published sources (comprising peer-reviewed articles, books, and theses), unpublished data, and herbarium records, spanning from 1815 to 2018. The geographical extent of the original data included countries such as the AF of Brazil, Argentina, and Paraguay. All non-vascular species were excluded. We used Flora do Brasil (Flora do Brasil 2020) and Zotz et al. (2021) to cross-reference epiphytic life-forms against our database. We retained any species classified as epiphyte in at least one of the reference databases.
Since many vascular epiphyte species are naturally rare, we also wanted to test if the displacement of a species was related to its number of records in the dataset, thus we generated three different datasets (e.g. C10, C5, and C3) in accordance to the minimum number of records per species (C10 = ten or more records; C5 five or more records; C3 three or more records). Following the removal of any species that did not match the occurrence criteria in each database, we retained 72,798 records from 1000 species for database C10, 74,882 records from 1312 species for database C5, and 75,599 records from 1,521 species for database C3.
Climatic variables
In order to build the environmental matrix, climate data was extracted from the 19 standard bioclimatic variables from WorldClim 2.1 database (Fick and Hijmans 2017) for the period between 1970 and 2000 at a spatial resolution of 30 s. Following Pie et al. (2022), a Principal Component Analysis was used, based on a covariance matrix of the 19 bioclimatic variables, which were scaled and centred prior to the analysis and retained for the first two axes. Our next step was to extract bioclimatic data from vascular epiphyte occurrence records using ‘raster’ 3.4–13 package (Hijmans 2021) and project them onto the first two axes of the principal components retained for the analysis. To investigate the extent of the occupancy of different species across the climate space, we compared the variance across ordination quadrants using Levene's test, incorporated into ‘car’ 3.0–11 (Fox and Weisberg 2019). By using the predictions in the Met Office Hadley Centre (UK) model–ukesm1-0-ll (2020), we explored how expected shifts due to climate change might affect the structure of the available climatic space. We used two SSP (e.g., Shared Socioeconomic Pathway) from the IPCC AR6 (e.g., 6th Intergovernmental Panel on Climate Change Assessment Report) climate change projections. SSP126, hereafter the optimistic scenario, forecasts global temperatures assuming atmospheric CO2 emissions will be cut to net zero in the year 2075, keeping global warming below 2 °C by 2100. SSP585, hereafter the pessimistic scenario, assumes that CO2 emissions will triple by 2075 and the increase in global temperature will be between 3.3 and 5.7 °C by 2100. We extracted the values of the above climate variables for each grid cell and designed them based on the PCA constructed with the current climate data. By estimating concave hulls, as implemented in ‘concaveman’ 1.1.0 (Gombin et al. 2020), we compared the extent of the corresponding climatic space. This approach allowed us to assess which regions of the climatic space harbour more species that are likely to have their climatic niche displaced in the future. All analyses were carried out in RStudio 2022.07.1 (R Core Team 2022) and maps were plotted in QGIS 3.16.14 (QGIS Development Team 2022).
To test whether the niche displacement of a species would be related to the number of records of that species in each dataset, we performed a logistic regression between the number of displaced species and its number of occurrences in the datasets using the glm function in R (R Core Team 2022; version 4.2.1). This was done separately for the C3, C5, and C10 datasets. The logistic regression analysis did not have its assumptions met by the data for the SSP126 scenario, probably due to the imbalance between the categories of the response variable (e.g. few entries of displaced species versus many entries of non-displaced species in this scenario). However, for SSP585 the logistic regression showed that there was no significant effect of the abundance of records of a species and the number of displaced species (Std. Error = 0.0008672; z value = -1.13; Pr( >|z|) = 0.26). This suggests that the lower number of records of some species did not necessarily imply that these species will be more likely among the species with their climatic niche displaced. Thus, any further analysis was performed on the C3 dataset only.
Finally, to test whether there was a difference between climate changes scenarios (SSP126 and SSP585) and the number of species found in different climate space regions as identified by the ordination analysis (e.g. warmer regions vs. cooler regions), a chi-square test was carried out using the chisq.test function in R (R Core Team 2022).
Results
The first two axes of the PCA explained 69% of the variance in climatic conditions across the AF. The first axis, which explains 49% of the data variance, was mainly related to mean air temperature, especially during colder months (e.g. June to August). The second axis, which accounted for 20% of the total variance, was mainly related to the precipitation, especially during the wet and warmer months (Table 1).
The broad distribution of species across most of the available climatic space indicates that vascular epiphytes tend to thrive in climatic conditions currently found throughout the AF (Fig. 2). The ordination climate space analysis suggests two distinct climate area across the AF; one with higher temperatures and lower precipitation (PC1-; PC2 +), hereafter referred to as the harsher zone, where the occurrence of epiphytes appears to be more restricted to species within Bromeliaceae, and a region of relatively lower temperature and higher precipitation (PC1 + , PC2-), here referred to as the milder zone, more occupied by other epiphyte families (Fig. 2). Among the 1,521 analysed species, 324 (21.3%) had their mean climatic occurrence within the harsher zone, 831 (54.6%) had their mean climatic occurrence within the milder zone, and 366 species (24.1%) had their mean climatic occurrence outside the harsher and milder zone (Fig. 2).
Principal component analysis of climatic conditions found in the AF. Gray circles show the climatic conditions. Occurrence of the different epiphyte species across the available climate space for the main epiphytic groups (e.g. Bromeliaceae, Orchidaceae, ferns and lycophytes and other angiosperm families) are shown in different colours. Closed circles represent species means and black open circles indicate individual records. The arrows describe the general climate space (see text for details). Red and blue rectangles show the harsher and the milder zone respectively. (Color figure online)
When projecting the PCA scores back cartographically (Fig. 3A, B), the lowest temperatures (Fig. 3A) were found in the southern part of the AF, especially in Argentina and Paraguay, as well as in the Brazilian southern states of Paraná, Santa Catarina, and Rio Grande do Sul. On the other hand, the central AF had warmer average temperatures. Lower temperatures were found in the higher regions of the Serra do Mar mountains, across the Brazilian states of Minas Gerais, São Paulo, Rio de Janeiro, and Espírito Santo. The purple band that extends across the interior of the Brazilian states of Minas Gerais, Bahia, and Piauí reflects the greater seasonality of temperature in the interior area of the Atlantic Forest, despite its relatively lower mean annual temperatures. Precipitation and seasonality patterns are notable from the purple regions across southern Brazilian states and on the south-eastern Brazil highland areas (Fig. 3B), indicating higher levels of precipitation. The purple hue that extends from southern Brazil to Minas Gerais state reflects local seasonality in precipitation (Fig. 3B).
Spatial distribution of component scores of the climatic conditions across the AF. To visualize the climate conditions associated with each grid cell, a principal component analysis was performed, and the results were mapped back to the corresponding cells. PC1 loadings mostly reflected variation in temperature (a), while PC2 described mostly precipitation (b). ARG Argentina, BRA Brazil, PAR Paraguay; Brazilian states: AL Alagoas, BA Bahia, CE = Ceará, ES Espírito Santo, GO Goiás, MG Minas Gerais, MS Mato Grosso do Sul, PB Paraíba, PE Pernambuco, PI Piauí, PR Paraná, RJ Rio de Janeiro, RN Rio Grande do Norte, RS Rio Grande do Sul, SC Santa Catarina, SE Sergipe, SP São Paulo
In quadrants with negative PC1 scores, species were less frequent and lied further to the boundaries of the available niche space. Variance was higher across the available niche space in cooler conditions (i.e., positive PC1 scores) than in warmer conditions (i.e., negative PC1 scores) (F = 6.28, p = 0.01229). It is important to note that this difference was not a by-product of the variances of the available climatic space when they compared in a similar manner (4.1 vs. 3.6, respectively). We found higher variance values when comparing regions with low annual precipitation (i.e., positive PC2 scores), with areas of high annual precipitation (i.e., negative PC2 scores) (F = 44.73, p = 3.168e-11). However, in this case, the difference could not simply be explained by differences in the available climatic niche space (11.3 vs. 6.8, respectively).
When comparing the entire available climatic space, there were significant differences among the studied groups (i.e., Bromeliaceae, Orchidaceae, ferns and lycophytes, and other angiosperm families) in their pattern of occupation (Table 2; Fig. 2), with ferns and lycophytes and orchids showing smaller variance in their climatic niches and Bromeliaceae presenting the larger variance in its occupation pattern (Table 2). Bromeliaceae largely occupies most of the available climatic space, but it is mostly associated with warmer regions (negative PC1). On the other hand, Orchidaceae, despite occupying most of the climatic space, have a higher number of occurrences in areas that represent positive PC1 and negative PC2 scores (i.e. milder temperatures + higher precipitation). Ferns and lycophytes were the groups with the most restricted occurrence across the climate space. Species of these groups are mostly related to milder temperature and humid areas (i.e. positive PC1 + negative PC2), many of which are close to the edge of the available climatic space.
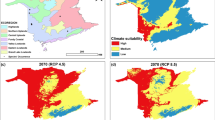
Climate projections for the years 2040 to 2100 for the optimistic (SSP126) and the pessimistic scenarios (SSP585), showed large climate niche shifts in the AF (Fig. 4). In general, the climate space will shift to hotter and drier conditions, with SSP585 projections showing the strongest shift (Fig. 5 and Fig. 6). Up to 20% of the analysed species will have their current average climatic niche outside future climatic spaces by 2100 (Table 3). This result is not due to a reduction in available climatic space, but due to a mismatch between present and future climatic spaces (Table 3 and Fig. 5). In general, the proportion of impacted species is larger for Orchidaceae, ferns and lycophytes compared to Bromeliaceae (Table 3). Under projections for the optimistic scenario, however, Bromeliaceae was the group with higher percentage of displaced species, while the pessimistic projections seem to affect ferns and lycophytes more. It is important to note that there were two species (i.e. Hymenophyllum crispum Kunth and H. magellanicum Willd.) that were indicated as outside the present climatic space. As suggested by Pie et al (2022), those were species living near the boundaries of the present climatic space but were categorized to be outside because of the way that concave hull was computed. They were left to ensure that all comparisons were carried out using the same context.
Principal component analysis of climatic conditions found in the AF, including climate space projections under SSP126 and SSP585 for the years 2040–2100. The different coloured lines represent different climate scenarios and years, whereas the different coloured open circles represent different epiphyte groups
Predicted consequences of climate space shifts according to different time frames and different climate change scenarios (a–h). The white area represents the current climate space, light green represents the future-predicted climate space, and dark green shows the overlap between current and predicted (see Table 4 for details)
Despite the harsher zone’s geographical location in the lowlands, which extend from Paraguay to Brazilian Northeast (Fig. 5) (307 m ± 141 m asl.; 22.46 °C ± 1.02 °C), all the potentially displaced species (234 m ± 108 m; 23.14 °C ± 0.69 °C) are found in the north-eastern coast, with most of them being found in Bahia state (Fig. 5). In the milder zone, which geographically corresponds to mid-elevation areas (694 m ± 227 m asl.; 18.5 °C ± 1.15 °C), most displaced species are found in higher altitudes (969 ± 218 m asl.; 17.06 °C ± 0.95 °C).
The χ-square test (χ2 = 48.46; p-value = 3.372e-12) revealed that species whose climatic mean is located in the harsher-zone are more sensitive to smaller shifts in their available climatic space compared to species on other zones (e.g. changes predicted for the optimistic scenario).
Discussion
Our analysis suggests that the climate space of the AF will get hotter and drier in the next 20–80 years. In addition, 10–35% of the currently climatic space will shift. These findings are in line with the latest IPCC assessment report for South America (IPCC 2022), which suggested that temperature in all South America subregions will most likely increase at rates greater than the global average. Precipitation, however, has been predicted to be more variable and regionally specific. While there is an expected decrease in precipitation in the harsher zone (i.e. the northern part of the AF), the milder zone (i.e. the southern part of the AF) will most likely experience an increase in precipitation according to IPCC predictions (IPCC 2022). In addition, the intensity and frequency of extreme precipitation and floods are projected to increase in the southern parts of the AF, whilst the northern parts will likely experience an increase in intensity of droughts (IPCC 2022).
We identified two prevalent climate spaces; a harsher (hot and dry) and milder (cold and humid) zone, which corresponds mostly to the northern and southern parts respectively of the AF. This north/south climate division has previously been reported in terms of differences in species diversity and composition for many organisms (Amorim and Pires 1996; Oliveira Filho and Fontes 2000; Pellegrino et al. 2005; Cabanne et al. 2007; Ledru et al. 2007; Thomé et al. 2010; DaSilva and Pinto-da-Rocha 2011; Dasilva et al. 2015), including epiphytes (Menini Neto et al. 2016). It has been suggested that the Doce River valley in northern Espírito Santo represents a dispersal barrier for many species between the south and the north of the AF.
We found that almost 300 (20%) epiphyte species may have their mean climatic niche displaced under future climate change projections, many of which seem to be more climate sensitive species or species that are already at their climate limit. For example, groups with the highest numbers of displaced species were bromeliads, ferns, and lycophytes. Interestingly, the lowland forests of the northern region of the AF, which is hot and dry, appeared to harbour a subset of species which are less tolerant to climate change; as it presents similar number of displaced species, both for the optimistic (n = 36; 11% of the species within the harsher zone) and the pessimistic scenario (n = 39; 12%). Concerning the southern region of the AF, which is cold and humid, our results suggests that it will be less affected under optimistic (SSP126) climate change predictions (n = 27; 3% of the species within the milder zone) compared to the harsher zone. However, under pessimistic predictions (SSP585), the shift in climate niche space within the milder zone leads to an increase of displaced species nearly eight times to that compared to the optimistic predictions for the same zone (n = 222; 27% of the species within the milder zone).
Although the majority of species within the harsher zone (n = 324) show adaptations to high temperatures (e.g. they have CAM metabolism and high leaf water retention capacity), there was a subset of species (n = 36; 11%) that still were displaced under more optimistic scenarios. It is likely that despite being adapted to areas with high temperature, low humidity and strong seasonality, the species that are predicted to be displaced may live close to their current limit of their climate niche, making them more susceptible. This suggests that even subtle changes in environmental conditions may have important ecological consequences for some species (Reyes et al. 2021). The harsher zone corresponds geographically to the lowlands (304 ± 141 m asl.), mostly represented by deciduous and semideciduous seasonal forest; all the displaced species were species found in the north-eastern coast such as the Bahia state. Many studies refer to a north/south division of the Atlantic Forest (Amorim and Pires 1996; Oliveira Filho and Fontes 2000; Pellegrino et al. 2005; Cabanne et al. 2007; Ledru et al. 2007; Thome et al. 2010; DaSilva and Pinto-da-Rocha 2011; DaSilva et al. 2015). The Doce River valley in northern Espirito Santo represents the barrier responsible for this disjunction, which separated the north-eastern region of AF from the south-eastern and southern regions. Stable Pleistocene forest refuges were formed even during the driest periods of the Quaternary (during the last glacial maximum, about 21 thousand years ago) in Bahia (in the Central Corridor of AF, north of the Rio Doce valley) and Pernambuco (further North) (Carnaval and Moritz 2008). Currently, the “Bahia refuge” (Carnaval and Moritz 2008; de Mello Martins 2011) corresponds to the “Bahia endemism area”, one of the five main endemism areas of the AF (Silva and Casteleti 2003). Nearly 80% of species displaced in the harsher zone are endemic to the Bahia region (Flora do Brasil 2020). For example, the genera Aechmea A. Juss. and Lymania R.W. Read had a particularly high number of endemic species that will be displaced (nine and six respectively), of which seven and five are endemic to the state.
The larger shift in the climate space predicted for the pessimistic scenario (up to 35%), is likely to affect species that occur in the milder zone. For example, we found that species in the milder zone occurred more frequently in higher elevation areas (693 m ± 227 m asl.) and areas with lower mean temperatures (18.5 °C ± 1.15 °C). Geographically, the milder zone corresponds to the mountain formations that spans from the southern Brazilian states to the south-eastern states of Espírito Santo and Minas Gerais, as well as the lower altitude areas in southern Brazil. This region encompasses a great variety of AF formations from submontana (50–500 m a.s.l.) to altomontana (> 1000 m a.s.l.) vegetation such as mixed ombrophilous forest, seasonal semideciduous forest and ombrophilous dense forest.
Ferns and lycophytes are likely to have the highest percentage of displaced species (e.g. n = 19–95; 8–42%). Other studies have found comparable results for epiphytic ferns. For example, Pie et al. (2022) found that 36–67 (28–53%) of epiphytic ferns and lycophytes in Honduras will be displaced by the year 2049–2099 under RCP2.6 and RCP8.5 projections respectively (IPCC 5th Assessment Report scenarios, 2014; RCP2.6 assumes that CO2 emissions start declining by 2020 and go to zero by 2100; RCP8.5 assumes that emissions continue to rise throughout the twenty-first century). Similarly, Reyes-Chávez et al. (2021) found that over 47% of ferns and lycophytes species along the highest mountain in Celaque National Park, Honduras will have to shift their range fully or partially above the maximum elevation of the mountain. We found that species within the genera Elaphoglossum and Hymenophyllum will be the most affected under the pessimistic scenario (SSP585) and that 19 and 13 species respectively, will be displaced by 2100. Within the AF, Elaphoglossum is especially diverse in mid-elevation montane forests, from 500 to 1500 m (Matos and Mickel 2014). Hymenophyllum on the other hand is highly hygrophilous, and is usually associated with microhabitats that are constantly humid (Kelly 1985; Hietz and Hietz-Seifert 1995), since their species lack cuticles (or have greatly reduced cuticles), differentiated epidermises, and stomata (Krömer et al. 2006). Particularly high diversity mountain regions (Suissa et al. 2021) that are occupied by species with narrow climate niches (e.g. many hygrophilous plants) (Pie et al. 2022), might be at greater risk. However, Kessler and Kluge (2022) recently suggested that the effect of climate change on ferns in mountain areas will be very species and context specific, as we currently still lack detailed data on how biotic and abiotic interacting effects might limit species responses to a changing climate.
For practical reasons of working with over 800 species in this study, we did not explicitly analyse intraspecific variation in climatic niches even though intraspecific variation in functional traits might play a relevant role in community dynamics (e.g., Lajoie and Vellend 2015; Niu et al. 2020; Kessler and Fluge 2022). Also, epiphytes are particularly sensitive to microclimatic changes (Hietz 1999), which are currently very difficult to account for in niche model simulations due to the lack of detailed spatial and temporal climate layers. One way this could be achieved is to use Climate Envelope Models (CEM) and parametrize them by using recent estimates of below canopy temperature and precipitation conditions, which account for the buffering potential of the above canopy (e.g. de Frenne et al. 2019; Zheng and Jia 2020). However, these models would be limited to individual species and would be computationally challenging to do across all AF epiphytes. Last, the epiphyte dataset we used may have collection biases, particularly for smaller, less-well surveyed epiphyte groups (see Araujo and Ramos 2021 for a detailed discussion). In addition, we calculated the average position that the climatic niche of each species occupies in relation to the predicted climatic space for the AF. Thus, it does not necessarily mean that species identified in our study to be displaced will disappear. However, it could increase the risk of extinction due to these species being closer to their climate niche limits, making them more vulnerable during perturbation events (e.g. droughts).
It is also important to highlight that, epiphytes are dependent on the host trees for their survival, thus any changes to the host tree cover or composition, due to climate and/or land-use change, could exacerbate our findings. For example, the AF has been significantly over-exploited in the past, including the cutting of brazilwood trees in the sixteenth century and later the removal of the forest cover for agricultural species such as sugarcane, coffee, cocoa, and foraging crops (Tabarelli 2005; Ribeiro et al. 2011). It is also home to the biggest cities in Brazil (São Paulo, Rio de Janeiro, and Porto Alegre) and evidence suggest that many potential host trees for epiphytes might disappear and/or change their community structure due to rapid changes in climate (Colombo and Joly 2010; Pinho et al. 2021).
Conclusion
In conclusion, we found that the AF climate space is likely to shift by 10–35% by the year 2100. Almost 20% of epiphyte species will have their mean climatic niche displaced, many of which are ferns and bromeliads. However, the severity of species displacement differed within a geographical context, while the harsher zone seems to be more impacted than the milder zone under SSP126 projections. Furthermore, species displacement was ten times higher under SSP585 projections for the milder zone compared to other zones. The findings from this work are important, as they can directly translate into current and future conservation forest management planning. Epiphytes have a close ecological relationship with many trees, thus any changes in the assemblages of epiphyte host trees (e.g. as a result of forest strategies such as logging or restoration) could exacerbate the loss of epiphytes in the future. We recommend that the species climate niche space in considered when planning for conservation.
Data availability
All raw data used in this study are openly avaliable to download from previouse publications. Please see methodology for relevant citations
References
Amorim DS, Pires MRS (1996) Neotropical biogeography and a method for maximum biodiversity estimation. In: Bicudo CEM, Menezes NA (eds) Biodiversity in Brazil: a first approach. CNPq, São Paulo, pp 183–219
Atwater DZ, Ervine C, Barney JN (2018) Climatic niche shifts are common in introduced plants. Nat Ecol Evol 2:34–43. https://doi.org/10.1038/s41559-017-0396-z
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Bellard C (2012) Impacts of climate change on the future of biodiversity. Ecol Lett 15:1–59. https://doi.org/10.1111/j.1461-0248.2011.01736.x
Benzing DH (1990) Vascular epiphytes. General biology and related biota. Cambridge University Press, Cambridge
Buckley LB, Davies JT, Ackerly DD, Kraft NJB, Harrison SP, Anacker BL et al (2010) Phylogeny, niche conservatism and the latitudinal diversity gradient in mammals. Proc R Soc B Biol Sci 277:2131–2138. https://doi.org/10.1098/rspb.2010.0179
Cabanne GS, Santos FR, Miyaki CY (2007) Phylogeography of Xiphorhynchus fuscus (Passeriformes, Dendrocolaptidae): Vicariance and recent demographic expansion in southern Atlantic forest. Biol J Linn Soc 91:73–84. https://doi.org/10.1111/j.1095-8312.2007.00775.x
Câmara IG (2003) Brief history of conservation in the Atlantic Forest. In: Galindo-Leal C, Câmara IG (eds) The Atlantic Forest of South America: biodiversity status, trends, and outlook. Cent Appl Biodivers Sci Island pp 31–42, Press, Washington, DC
Carnaval AC, Moritz C (2008) Historical climate modelling predicts patterns of current biodiversity in the Brazilian Atlantic forest. J Biogeogr 35:1187–1201
Colombo AF, Joly CA (2010) Brazilian Atlantic Forest lato sensu: the most ancient Brazilian forest, and a biodiversity hotspot, is highly threatened by climate change. Braz J Biol 70:697–708
Copertino MS, Garcia AM, Muelbert JH, Garcia CAE (2010) Introduction to the special issue on climate change and Brazilian coastal zone. Pan-Am J Aquat Sc 5:1–8
Dasilva MB, Pinto-da-Rocha R, Desouza AM (2015) A protocol for the delimitation of areas of endemism and the historical regionalization of the Brazilian Atlantic Rain Forest using harvestmen distribution data. Cladistics 31:692–705. https://doi.org/10.1111/cla.12121
DaSilva MB, Pinto-da-Rocha M (2011) História biogeográfica da Mata Atlântica: Opiliões (Arachnida) como modelo para sua inferência. In: Carvalho CJB, Almeida EAB (eds) Biogeografia da América do Sul—Padrões & Processos. Editora Roca, São Paulo, pp 41–51
de Mello MF (2011) Historical biogeography of the Brazilian Atlantic forest and the Carnaval-Moritz model of Pleistocene refugia: What do phylogeographical studies tell us? Biol J Linn Soc 104:499–509. https://doi.org/10.1111/j.1095-8312.2011.01745.x
de Araujo ML, Ramos FN (2021) Targeting the survey efforts: gaps and biases in epiphyte sampling at a biodiversity hotspot. For Ecol Manage. https://doi.org/10.1016/j.foreco.2021.119544
de Frenne P, Zellweger F, Rodríguez-Sánchez F et al (2019) Global buffering of temperatures under forest canopies. Nat Ecol Evol 3:744–749. https://doi.org/10.1038/s41559-019-0842-1
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Flora do Brasil (2020) Jardim Botânico do Rio de Janeiro. Accessed on: 17 Nov. 2022. https://floradobrasil.jbrj.gov.br/reflora/listaBrasil/PrincipalUC/PrincipalUC.do#CondicaoTaxonCP
Flora e Funga do Brasil. Jardim Botânico do Rio de Janeiro. Available at: < http://floradobrasil.jbrj.gov.br/ >. Accessed on: 17 Nov. 2022.
Forzza RC, Baumgratz JFA, Bicudo CEM, Canhos DAL, Carvalho AA, Coelho MAN et al (2012) New brazilian floristic list highlights conservation challenges. Bioscience 62:39–45. https://doi.org/10.1525/bio.2012.62.1.8
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd edn. Sage, Thousand Oaks
Freitas L, Salino A, Neto LM, Almeida TE, Mortara SR, Stehmann JR et al (2016) A comprehensive checklist of vascular epiphytes of the Atlantic Forest reveals outstanding endemic rates. PhytoKeys 58:65–79. https://doi.org/10.3897/phytokeys.58.5643
Galetti M, Guevara R, Côrtes MC, Fadini R, Von Matter S, Leite AB, Labecca F, Ribeiro T, Carvalho CS, Collevatti RG, Pires MM, Guimarães PR, Brancalion PH, Ribeiro MC et al (2013) Functional extinction of birds drives rapid evolutionary changes in seed size. Science 340(6136):1086–1090. https://doi.org/10.1126/science.1233774
Gombin J, Vaidyanathan R, Agafonkin V (2020) Concaveman: A very fast 2D concave hull algorithm. R package version 1.1.0. https:// CRAN.R- proje ct. org/package=concaveman
Hidasi-neto J, Joner DC, Resende F, Macedo Monteiro L, Valtuille F, Dias R et al (2019) Climate change will drive mammal species loss and biotic homogenization in the Cerrado Biodiversity Hotspot. Perspect Ecol Conserv 17:57–63. https://doi.org/10.1016/j.pecon.2019.02.001
Hietz P, Hietz-Seifert U (1995) Composition and ecology of vascular epiphyte communities along an altitudinal gradient in central Veracruz, Mexico. J Veg Sci 6:487–498
Hietz P (1999) Diversity and conservation of epiphytes in a changing environment. In: Proceedings of the International Conference on Biodiversity and Bioresources: Conservation and Utilization. Phuket, Thailand.
Hijmans RJ (2021) Raster: geographic data analysis and modelling. R package version 3.4–13. https://CRAN.R-project.org/package=raster.
Hsu RCC, Tamis WLM, Raes N, de Snoo GR, Wolf JHD, Oostermeijer G et al (2012) Simulating climate change impacts on forests and associated vascular epiphytes in a subtropical island of East Asia. Divers Distrib 18:334–347. https://doi.org/10.1111/j.1472-4642.2011.00819.x
Hutchinson GE (1957) Concluding remarks. Cold Spring Harbour Symp Quant Biol 22:415–427
IPCC (2022) Climate Change 2022: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Pörtner HO, Roberts DC, Tignor M, Poloczanska ES, Mintenbeck K, et al. (eds.)]. Cambridge University Press. Cambridge University Press, Cambridge pp. 3056,doi:https://doi.org/10.1017/9781009325844.
Kelly D (1985) Epiphytes and climbers of a Jamaican rain forest: vertical distribution, life forms and life histories. J Biogeogr 12(3):223–241
Kerkhoff AJ, Moriarty PE, Weiser MD (2014) The latitudinal species richness gradient in New World woody angiosperms is consistent with the tropical conservatism hypothesis. Proc Natl Acad Sci USA 111:8125–8130. https://doi.org/10.1073/pnas.1308932111
Kessler M, Kluge J (2022) Mountain ferns: what determines their elevational ranges and how will they respond to climate change? Am Fern J. https://doi.org/10.1640/0002-8444-112.4.285
Khaliq I, Fritz SA, Prinzinger R, Pfenninger M, Böhning-Gaese K, Hof C (2015) Global variation in thermal physiology of birds and mammals: evidence for phylogenetic niche conservatism only in the tropics. J Biogeogr 42:2187–2196. https://doi.org/10.1111/jbi.12573
Krömer T, Kessler M, Herzog SK (2006) Distribution and flowering ecology of bromeliads along two climatically contrasting elevational transects in the Bolivian Andes. Biotropica 38:183–195. https://doi.org/10.1111/j.1744-7429.2006.00124.x
Lajoie G, Vellend M (2015) Understanding context dependence in the contribution of intraspecific variation to community trait-environment matching. Ecol 96:2912–2922. https://doi.org/10.1890/15-0156.1
Lawton JH, May RM (1995) Extinction Rates. Oxford Univ. Press, Oxford
Ledru MP, Salatino MLF, Ceccantini G, Salatino A, Pinheiro F, Pintaud JC (2007) Regional assessment of the impact of climatic change on the distribution of a tropical conifer in the lowlands of South America. Divers Distrib 13:761–771. https://doi.org/10.1111/j.1472-4642.2007.00389.x
Marques MCM, Trindade W, Bohn A, Grelle CEV (2021) The Atlantic forest: an introduction to the Megadiverse forest of South America. In: Marques MCM, Grelle CEV (eds) The Atlantic Forest: History, Biodiversity, Threats and Opportunities of the Mega-Diverse Forest. Springer, Cham, pp 233–252
Matos FB, Mickel JT (2014) The Brazilian species of Elaphoglossum section Polytrichia (Dryopteridaceae). Brittonia 66:371–395. https://doi.org/10.1007/s12228-014-9352-2
Menini Neto L, Furtado SG, Zappi DC, de Oliveira Filho AT, Forzza RC (2016) Biogeography of epiphytic Angiosperms in the Brazilian Atlantic forest, a world biodiversity hotspot. Rev Bras Bot 39:261–273. https://doi.org/10.1007/s40415-015-0238-7
Met Office Hadley Centre (2020) WCRP CMIP6: Met Office Hadley Centre (MOHC) UKESM1–0-LL model output for the “histSST” experiment. Centre for Environmental Data Analysis, Accessed on: 17 Nov. 2022. https://catalogue.ceda.ac.uk/uuid/b4662472624a4e7aaeabf55122998721
Mittermeier RA, van Dijk PP, Rhodin AGJ, Nash SD (2004) Hotspots revisited: earth’s biologically richest and most endangered ecoregions, 2nd edn. University of Chicago Press, Chicago
Mota FMM, Heming NM, Morante-Filho JC, Talora DC (2022) Climate change is expected to restructure forest frugivorous bird communities in a biodiversity hot-point within the Atlantic Forest. Divers Distrib. https://doi.org/10.1111/ddi.13602
Myers N, Mittermeler RA, Mittermeler CG, Da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Niu K, Zhang S, Lechowicz MJ (2020) Harsh environmental regimes increase the functional significance of intraspecific variation in plant communities. Funct Ecol 34:1666–1677. https://doi.org/10.1111/1365-2435.13582
Oliveira Filho AT, Fontes MAL (2000) Patterns of floristic differentiation among Atlantic Forests in Southeastern Brazil and the influence of climate. Biotropica 32:793–810. https://doi.org/10.1111/j.1744-7429.2000.tb00619.x
Pecl GT, Araújo MB, Bell JD, Blanchard J, Bonebrake TC, Chen IC et al (2017) Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 355:6332. https://doi.org/10.1126/science.aai9214
Pellegrino KCM, Rodrigues MT, Waite AN, Morando M, Yassuda YY, Sites JW (2005) Phylogeography and species limits in the Gymnodactylus darwinii complex (Gekkonidae, Squamata): Genetic structure coincides with river systems in the Brazilian Atlantic Forest. Biol J Linn Soc 85:13–26. https://doi.org/10.1111/j.1095-8312.2005.00472.x
Petitpierre B, Kueffer C, Broennimann O, Randin C, Daehler C, Guisan A (2012) Climatic niche shifts are rare among terrestrial plant invaders. Science 335:1344–1348. https://doi.org/10.1126/science.1215933
Pie MR, Batke SP, Reyes-Chávez J, Dallimore T (2022) Fern and lycophyte niche displacement under predicted climate change in Honduras. Plant Ecol 223:613–625. https://doi.org/10.1007/s11258-022-01235-8
Pinho BX, Tabarelli M, ter Braak CJF et al (2021) Functional biogeography of neotropical moist forests: trait-climate relationships and assembly patterns of tree communities. Glob Ecol Biogeogr 30(7):1430–1446
QGIS Development Team (2022) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ramos FN, Mortara SR, Monalisa-Francisco N, Elias JPC, Neto LM, Freitas L, Kersten RA, Amorim AM, Matos FB, Nunes-Freitas AF et al (2019) Atlantic epiphytes: a data set of vascular and non-vascular epiphyte plants and lichens from the Atlantic Forest. Ecol 100(2):e02541. https://doi.org/10.1002/ecy.2541
Ramos FN, Alfenas UF, Mortara S, Alfenas UF (2021) The Atlantic Forest. Springer, Cham, pp 45–46. https://doi.org/10.1007/978-3-030-55322-7
Reyes-Chávez J, Quail M, Tarvin S, Kessler M, Batke SP (2021) Nowhere to escape - diversity and community composition of ferns and lycophytes on the highest mountain in Honduras. J Trop Eco 37:72–81. https://doi.org/10.1017/S0266467421000122
Ribeiro MC, Metzger JP, Martensen AC, Ponzoni FJ, Hirota MM (2009) The Brazilian Atlantic Forest: How much is left, and how is the remaining forest distributed? Implications for conservation. Biol Cons 142:1141–53. https://doi.org/10.1016/j.biocon.2009.01.033
Ribeiro MC, Martensen AC, Metzger JP, Tabarelli M, Scarano F, Fortin MJ (2011) The Brazilian Atlantic Forest: a shrinking biodiversity hotspot. In: Zachos FE, Habel JC (eds) Biodiversity hotspots. Springer, Berlin, pp 405–434
Ruegg K, Anderson EC, Somveille M, Bay RA, Whitfield M, Paxton EH et al (2021) Linking climate niches across seasons to assess population vulnerability in a migratory bird. Glob Chang Biol 27:3519–3531. https://doi.org/10.1111/gcb.15639
Silva JMC, Casteleti CH (2003) Status of the biodiversity of the Atlantic forest of Brazil. In: Galindo-Leal C, Câmara IG (eds) The Atlantic Forest of South America: biodiversity status, threats, and outlook. Island Press, Washington, pp 43–59
Spaak JW, Baert JM, Baird DJ, Eisenhauer N, Maltby L, Pomati F et al (2017) Shifts of community composition and population density substantially affect ecosystem function despite invariant richness. Ecol Lett 20:1315–1324. https://doi.org/10.1111/ele.12828
Stanton DE, Huallpa Chávez J, Villegas L, Villasante F, Armesto J, Hedin LO et al (2014) Epiphytes improve host plant water use by microenvironment modification. Funct Ecol 28:1274–1283. https://doi.org/10.1111/1365-2435.12249
Stehmann JR, Forzza RC, Salino A, Sobral M, Pinheiro D, Kamino HY, et al. (2009) Plantas da Floresta Atlântica. Instituto de Pesquisas Jardim Botânico do Rio de Janeiro Instituto de Pesquisas, Jardim Botânico do Rio de Janeiro, Rio de Janeiro.
Suissa JS, Sundue MA, Testo WL (2021) Mountains, climate and niche heterogeneity explain global patterns of fern diversity. J Biogeogr 48:1296–1308. https://doi.org/10.1111/jbi.14076
Tabarelli M, Pinto LP, Silva JM, Bede LC (2005) Challenges and opportunities for biodiversity conservation in the brazilian atlantic forest. Conserv Biol 19:695–700. https://doi.org/10.1111/j.1523-1739.2005.00694.x
Thomé MTC, Zamudio KR, Giovanelli JGR, Haddad CFB, Baldissera FA, Alexandrino J (2010) Phylogeography of endemic toads and post-pliocene persistence of the Brazilian Atlantic Forest. Mol Phylogenet Evol 55:1018–1031. https://doi.org/10.1016/j.ympev.2010.02.003
Thuiller W, Lavorel S, Araújo MB, Sykes MT, Prentice IC (2005) Climate change threats to plant diversity in Europe. Proc Natl Acad Sci USA 102:8245–8250. https://doi.org/10.1073/pnas.0409902102
Wasof S, Lenoir J, Aarrestad PA et al (2015) Disjunct populations of European vascular plant species keep the same climatic niches. Glob Ecol Biogeogr 24(12):1401–1412
Wiens JJ, Donoghue MJ (2004) Historical biogeography, ecology and species richness. Trends Ecol Evol 19:639–644. https://doi.org/10.1016/j.tree.2004.09.011
Zanella FCV (2011) Evolução da biota da diagonal de formações abertas secas da Ame´rica do Sul. In: Carvalho CJB, Almeida EAB (eds) Biogeografia da América do Sul-Padrões & Processos. Editora Roca, São Paulo, pp 198–220
Zheng CL, Jia L (2020) Global canopy rainfall interception loss derived from satellite earth observations. Ecohydro 13(2):e2186. https://doi.org/10.1002/eco.2186
Zotz G (2016) Plants on plants – the biology of vascular epiphytes. Springer, Switzerland
Zotz G, Bader MY (2008) Epiphytic plants in a changing world-global: change effects on vascular and non-vascular epiphytes. Prog Bot. https://doi.org/10.1007/978-3-540-68421-3_7
Zotz G, Weigelt P, Kessler M, Kreft H, Taylor A (2021) EpiList 1.0: a global checklist of vascular epiphytes. Ecol 102:3326. https://doi.org/10.1002/ecy.3326
Acknowledgements
This work was financially supported by Edge Hill University (GTA funding scheme). We thank all the researchers that contributed to the original database used in this study.
Funding
This work was supported by an Edge Hill University Ph.D. Teaching Graduate Scholarship awarded to MM.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data curation and analysis were performed by MM, with support from SB, MP and FR. The first draft of the manuscript was written by MM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Simon Pierce.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Murakami, M., Batke, S., Pie, M.R. et al. Climate change might lead to substantial niche displacement in one of the most biodiverse regions in the world. Plant Ecol 224, 403–415 (2023). https://doi.org/10.1007/s11258-023-01309-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-023-01309-1