Abstract
Purpose
As an indicator of nutrition and immunity, the prognostic value of the prognostic nutritional index (PNI) has been confirmed in various diseases. However, the relationship between PNI and the incidence of pneumonia in peritoneal dialysis (PD) patients remains unknown. The purpose of this study was to investigate the relationship between PNI and new-onset pneumonia in patients undergoing PD.
Methods
Thousand two hundred and nighty eight patients were enrolled in this multicenter retrospective study from February 1, 2010, to February 28, 2020. A total of 899 patients were included in the final statistical analysis. The patients were stratified into two groups by PNI quartiles. The primary endpoint was a new-onset pneumonia event. Cox regression model analysis was used to explore the association between PNI and the first occurrence of pneumonia.
Results
During a mean follow-up of 41.43 months, 147 patients developed new-onset pneumonia. Kaplan–Meier survival curves showed a significant difference in the incidence of the first presentation of pneumonia between the two groups, that patients in the low PNI group had a higher risk of pneumonia (P = 0.016). By adjusting for demographic parameters, comorbidities, and laboratory indicators, the Cox regression model showed that the high PNI group had less risk compared to the low PNI group (HR 0.479 95% CI 0.297–0.772, P = 0.003). There were no interactions in the subgroups as follows: diabetes, hypertension, age, and sex.
Conclusions
Low PNI levels were independently associated with the first occurrence of pneumonia in PD patients. PNI was an independent predictor of new-onset pneumonia in PD patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patients with chronic kidney disease were more susceptible to infections [1,2,3]. Infections were the second most common cause of hospitalization and death, and about one in a quarter of infection-related deaths were due to pulmonary disease in the National Kidney Registry study [4]. Some studies showed the cumulative probability of pneumonia hospitalization was 36% at 5 years in dialysis patients. The mortality after pulmonary infectious was 14–16-fold higher in dialysis patients compared with the normal population [5, 6]. Long-term respiratory tract infection is prone to cardiovascular events, such as myocardial infarction, which poses a huge burden on the health care system [7].
In patients with hemodialysis, good nutritional status had a protective effect against pulmonary infection and was associated with improved survival [8]. On the contrary, there is a close relationship between malnutrition and inflammation, chronic malnutrition leads to compromised immune function and infections [9]. Protein-energy depletion (PEW), characterized by significantly low serum albumin levels, inflammation, and oxidative stress, is common in patients with chronic kidney disease [10]. PEW is associated with significantly higher mortality and lower quality of life [11]. Prognostic nutritional index (PNI), combined with serum albumin and lymphocyte count, may be an excellent PEW biomarker [12]. The low PNI was associated with CVD mortality in PD patients [13]. It is not clear whether PNI is associated with the occurrence of pneumonia in patients with PD.
Methods
Participants
The cohort included 1298 patients who underwent peritoneal dialysis between February 1, 2010, and February 28, 2020, from 3 peritoneal dialysis centers, and followed until June 1, 2020. Patients aged < 18 years or >80 years and who lacked albumin or lymphocyte count data were excluded. The study proposal was conducted in accordance with the ethical principles of the Declaration of Helsinki and approved by the Institutional Review Board of the Sixth Hospital of Sun Yat-sen University (No. 2021SLYEC-177). Written informed consent was not required for this study because of the retrospectively collected medical records available in the hospitals.
Data collection
All data were collected within 3 months after starting peritoneal dialysis. Demographic data included age, gender, body mass index (BMI), smoking and drinking history, underlying diseases, such as diabetes, hypertension, and cardiovascular disease. PNI was calculated using the formula PNI = (10 × Alb g/dL) + (0.005 × total lymphocyte counts/mm3) [13]. BMI was calculated as body weight in kilograms divided by height in meters squared. The weight and height determined at admission were used to calculate BMI. The diagnosis of a history of diabetes was based on the diagnostic criteria of the American Diabetes Association [14]. Hypertension was recorded if the patient was taking antihypertensive medication or had two blood pressure measurements ≥ 140/90 mmHg [15]. Other histories of diseases were recorded according to the first page of the patient's medical record. The definition of pneumonia mainly depends on discharge diagnosis. The diagnosis of pneumonia is defined by a professional respiratory physician based on the patient's symptoms, such as cough, dyspnea, and fever, the results of laboratory tests, and the presence of a new infiltrate on the chest X-ray or CT [16].
Outcomes
The primary outcome of this study was the first occurrence of pneumonia. The follow-up endpoint was any of the following: death, transfer to hemodialysis, transfer to another center, transfer to kidney transplant, or censoring on June 1, 2020.
Statistical analysis
Patients were divided into two groups based on the quartiles of PNI. The first quartile PNI is low PNI (PNI ≤ 36.1), and the other three quartiles PNI are high PNI (PNI > 36.1) [13]. Continuous variables conforming to a normal distribution were expressed as mean ± standard deviation, and continuous variables conforming to a skewed distribution were expressed as median (25th–75th percentile). Categorical variables were expressed in terms of numbers (percentiles). The difference between the PNI groups was tested by t test, Chi-squared, or the nonparametric Mann–Whitney test. Univariate Cox regression explored possible factors associated with the onset of first pneumonia. Cox proportional risk regression model was used to test the relationship between PNI and new pneumonia: model 1: BMI and age, model 2: model 1 plus comorbid conditions and history of smoking and drinking, model 3: model 2 plus laboratory parameters. The Kaplan–Meier curve was used to examine the difference in the risk of new-onset pneumonia between the two PNI groups during follow-up. The log-rank test was used to evaluate the differences. Spearman correlation analysis was used to explore the correlation between PNI and other nutrition-related indicators and other indicators. The forest map was used to show subgroup analysis, comparing whether there are interactions. The statistical analysis was done by SPSS25.0 and GraphPad Prism (version 8.0.2). P value < 0.05 was considered statistically significant.
Results
Participants
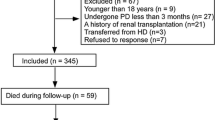
A total of 1298 patients were collected in three centers, of which 399 were excluded for the following reasons: significant data were absent from the study (n = 377), age younger than 18 years or older than 80 years (n = 22). 899 patients were ultimately included in the study. 147 patients developed pneumonia. (Fig. 1). Baseline demographic data and laboratory indicators of patients are demonstrated in Table 1. Of the 899 patients, the median age was 59 (47–67) years; 516 (57.4%) patients were men; 259 (28.8%) had diabetes, and 615 (68.4%) had hypertension. The median PNI level at baseline was 40.6 (36.8–43.6) for all patients. The median follow-up time was 41.13 months and the maximum follow-up time was 122 months. Patients in the high PNI group had a lower incidence of diabetes. There was no significant difference in the incidence of previous cardiovascular disease and hypertension. The levels of white blood cells, triglycerides, hemoglobin, serum calcium, phosphorus, and potassium in patients with low PNI were lower than those in patients with high PNI. The levels of platelets and HDL were higher in the low PNI group (Table 1).
Correlations between baseline PNI and clinical parameters
The Spearman correlation coefficients demonstrated that PNI values were significantly positively correlated with BMI (r = 0.077), WBC (r = 0.111), RBC (r = 0.190), hemoglobin (r = 0.208) and triglyceride (r = 0.207). Meanwhile, PNI was negatively correlated with age (r = −0.204) and HDL (r = −0.091). The correlation between PNI and cholesterol was not statistically significant (Table 2).
Baseline PNI and new-onset pneumonia
Table 3 shows that patients with higher BMI, diabetes and older age were more likely to suffer from pneumonia after adjustment for covariates (P < 0.05). After adjustment for age, BMI, complications, and laboratory measures, low PNI was an independent risk factor for pneumonia. Table 4 shows that the high PNI may reduce the risk of pneumonia in PD patients compared with the low PNI group (HR 0.479, 95% CI 0.297–0.772) by the adjusted Cox regression model.
Kaplan–Meier survival curves and log-rank tests showed a statistically significant difference in the first occurrence of pneumonia between the two groups (log-rank test, P = 0.016) (Fig. 2). We also investigated the association between different subgroups (including men or women, whether diabetes mellitus, hypertension, over 60 years < 60 years) and the incidence of first pneumonia. The forest map in Fig. 3 showed that there is no interaction in those subgroups.
Discussion
This retrospective cohort study explored that the low PNI was a risk factor for pneumonia in patients with PD. To our knowledge, this is the first report on the effects of PNI on the occurrence of pneumonia in patients with PD.
Previous studies have shown that PNI was related to the occurrence of pneumonia in non-dialysis patients. A retrospective study showed that preoperative PNI was closely related to postoperative pneumonia in patients undergoing curative lung cancer resection [17]. Another study showed that PNI was independently correlated with the severity of coronavirus disease in 2019 [18]. No research has focused on the relationship between PNI and pneumonia in PD patients. The research on the relationship between prognosis and PNI in PD patients mainly focuses on all-cause mortality, cardiovascular events, and cardiovascular mortality [13, 19]. Yang reported that PNI had better predictive value in all-cause mortality and cardiovascular events in patients with PD [19].
Inflammation, immunity, and nutrition are closely related to each other in patients [20]. Long-term chronic inflammation can cause immune depression and malnutrition. At the same time, malnutrition can lead to immune deficit and acute infection. PEW is common in patients with chronic kidney disease, especially in patients undergoing peritoneal dialysis, and is associated with mortality and hospitalization rates for infections [21]. Inadequate food intake and changes caused by uremia, such as increased energy consumption, persistent inflammation, acidosis, and a variety of endocrine disorders are the main causes of PEW [22]. Many studies have shown that hypoalbuminemia was a strong indicator for PEW [23]. Serum albumin had a protective effect against endothelial dysfunction that related to inflammation and oxidative stress [24]. Serum albumin levels below 30 g/L are associated with a twofold increased risk of peritonitis [25]. However, serum albumin is unreliable in the diagnosis of PEW, because it is affected by many factors, such as inflammation, liver disease, diabetes. PNI is not only a nutritional index but also an immunological index. Low PNI means low albumin and/or decreased lymphocyte counts, which was closely associated with the occurrence of pneumonia in PD patients.
In this study, PNI was positively correlated with nutritional indicators, such as hemoglobin, triglyceride, and BMI. This proved that PNI can well reflect the nutritional status of patients. The results of our study showed that PNI was negatively correlated with HDL. Sangmee has shown that patients with low baseline HDL had a significantly increased risk of future pneumonia hospitalization over a median follow-up of more than 20 years [26], this result is in accordance with our results. Previous studies also showed old age, low BMI, and diabetes were all risk factors for pneumonia [4, 27]. Spearman correlation analysis showed that PNI was negatively correlated with age in our study. The low levels of PNI represent the worse nutritional status of elderly patients. Although BMI is an immune and nutritional index, the standard of BMI is different in different races. This limits the use of BMI in comparing the nutritional and immune status of different races. While the correlation coefficient between PNI and anemia, protein metabolism and MBD was not high. The possible reasons are as follows: first, anemia is more related to erythropoietin and iron metabolism [28], but has a lower correlation coefficient with PNI which represents inflammation and nutrition. Second, the average PD effluent protein loss was 6.41 ± 2.16 g/day [29]. The loss of protein will seriously affect the protein metabolism indicators, such as serum creatinine, BUN, and BMI, which is the reason why the correlation coefficient between PNI and the above-mentioned indexes is not high. Third, the MBD index is more affected by parathyroid function and dialysis adequacy [30], so the correlation coefficient with PNI is not high. Although the correlation coefficient is low, it may be suggestive that PNI may integrate and represent the prognostic value of all these factors.
In previous studies, the cutoff value of PNI ranged from 40 to 50 [31, 32]. At the moment, no large-scale study has given the exact cutoff value of PNI in predicting death, cardiovascular events, and inflammation. The cutoff value of PNI in our study is lower than that in other reports, possibly because the selected patients are different. The enrolled patients in our study were undergoing PD, and those patients are more likely to develop symptoms of protein malnutrition [33]. This study has some limitations that need to be addressed. First, although this was a multicenter retrospective study and important confounding factors were adjusted, there may still be some potential bias and limits the causality that can be inferred from observed associations. Second, baseline data were used in this study, but the follow-up laboratory data were lacking. Third, as the database has been established for a long time, we lack data on CRP and dietary. Fourth, the etiological data were not available for all patients with new-onset pneumonia, so the type of pneumonia in the patients could not be distinguished. Finally, our study did not determine the accurate cutoff value of PNI.
To prevent pneumonia in patients with peritoneal dialysis, the albumin and lymphocyte counts of patients should be monitored regularly, and the malnutrition and immune status of patients should be roughly assessed. Early and timely intervention can prevent the occurrence of pneumonia and reduce the incidence of complications caused by pneumonia.
Conclusions
PNI is a promising indicator for predicting inflammation. Low PNI is an independent risk factor for new-onset pneumonia in PD patients. Patients undergoing PD with the low level of PNI should improve nutritional status to avoid the occurrence of pneumonia.
References
McDonald HI et al (2015) CKD and the risk of acute, community-acquired infections among older people with diabetes mellitus: a retrospective cohort study using electronic health records. Am J Kidney Dis 66(1):60–68
Foley RN (2006) Infections and cardiovascular disease in patients with chronic kidney disease. Adv Chronic Kidney Dis 13(3):205–208
Naqvi SB, Collins AJ (2006) Infectious complications in chronic kidney disease. Adv Chronic Kidney Dis 13(3):199–204
Guo H et al (2008) Pneumonia in incident dialysis patients–the United States Renal Data System. Nephrol Dial Transplant 23(2):680–686
Slinin Y, Foley RN, Collins AJ (2006) Clinical epidemiology of pneumonia in hemodialysis patients: the USRDS waves 1, 3, and 4 study. Kidney Int 70(6):1135–1141
Sarnak MJ, Jaber BL (2001) Pulmonary infectious mortality among patients with end-stage renal disease. Chest 120(6):1883–1887
Dalrymple LS et al (2011) Risk of cardiovascular events after infection-related hospitalizations in older patients on dialysis. Clin J Am Soc Nephrol 6(7):1708–1713
Liu J et al (2018) Clinical characteristics of pneumonia in chinese hemodialysis patients. Chin Med J (Engl) 131:498–501
Merker M et al (2020) Association of baseline inflammation with effectiveness of nutritional support among patients with disease-related malnutrition: a secondary analysis of a randomized clinical trial. JAMA Netw Open 3(3):e200663
Han S, Han D (2012) Nutrition in patients on peritoneal dialysis. Nat Rev Nephrol 8(3):163–175
Lodebo BT, Shah A, Kopple JD (2018) Is it important to prevent and treat protein-energy wasting in chronic kidney disease and chronic dialysis patients? J Renal Nutr 28(6):369–379
Onodera T, Goseki N, Kosaki G (1984) [Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients]. Nihon Geka Gakkai Zasshi 85:1001–1005
Peng F et al (2017) Low prognostic nutritional index associated with cardiovascular disease mortality in incident peritoneal dialysis patients. Int Urol Nephrol 49(6):1095–1101
Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (2003) Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 26:S5-20
Georgianos PI, Agarwal R (2016) Epidemiology, diagnosis and management of hypertension among patients on chronic dialysis. Nat Rev Nephrol 12(10):636–647
Viasus D et al (2011) Epidemiology, clinical features and outcomes of pneumonia in patients with chronic kidney disease. Nephrol Dial Transplant 26(9):2899–2906
Park S et al (2020) The prognostic nutritional index and postoperative complications after curative lung cancer resection: a retrospective cohort study. J Thorac Cardiovasc Surg 160(1):276-285.e1
Hu X et al (2021) Predictive value of the prognostic nutritional index for the severity of coronavirus disease 2019. Nutrition 84:111123
Yang Y et al (2020) Evaluation of objective nutritional indexes as predictors of worse outcomes in peritoneal dialysis patients. Nutrition 79–80:110963
Wu D et al (2018) Nutritional modulation of immune function: analysis of evidence, mechanisms, and clinical relevance. Front Immunol 9:3160
Ikizler TA et al (2013) Prevention and treatment of protein energy wasting in chronic kidney disease patients: a consensus statement by the International Society of Renal Nutrition and Metabolism. Kidney Int 84(6):1096–1107
Carrero JJ et al (2013) Etiology of the protein-energy wasting syndrome in chronic kidney disease: a consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J Renal Nutr 23(2):77–90
Fouque D et al (2008) A proposed nomenclature and diagnostic criteria for protein-energy wasting in acute and chronic kidney disease. Kidney Int 73:391–398
Kremer H et al (2011) Human serum albumin improves endothelial dysfunction and survival during experimental endotoxemia: concentration-dependent properties. Crit Care Med 39(6):1414–1422
Cho Y, Johnson DW (2014) Peritoneal dialysis-related peritonitis: towards improving evidence, practices, and outcomes. Am J Kidney Dis 64(2):278–289
Bae SS et al (2021) Major lipids and future risk of pneumonia: 20-year observation of the atherosclerosis risk in communities (ARIC) Study Cohort. Am J Med 134(2):243-251.e2
Chebib N et al (2021) Pneumonia prevention in the elderly patients: the other sides. Aging Clin Exp Res 33(4):1091–1100
Babitt JL et al (2021) Controversies in optimal anemia management: conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int 96:1280–1295
Lu W et al (2019) Peritoneal protein clearance predicts mortality in peritoneal dialysis patients. Clin Exp Nephrol 23(4):551–560
Ketteler M et al (2018) Diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder: synopsis of the kidney disease: Improving Global Outcomes 2017 Clinical Practice Guideline Update. Ann Intern Med 168(6):422–430
Tokunaga R et al (2015) Prognostic nutritional index predicts severe complications, recurrence, and poor prognosis in patients with colorectal cancer undergoing primary tumor resection. Dis Colon Rectum 58(11):1048–1057
Migita K et al (2013) The prognostic nutritional index predicts long-term outcomes of gastric cancer patients independent of tumor stage. Ann Surg Oncol 20(8):2647–2654
Zha Y, Qian Q (2017) Protein nutrition and malnutrition in CKD and ESRD. Nutrients 9(3):208
Acknowledgements
We thank all study participants and staff in our study.
Funding
This research was supported by The Key Project of Social Science and Technology of Dongguan (No. 201950715046061).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the patients had a potential conflict of interest.
Ethics approval and consent to participate
The study proposal was conducted in accordance with the ethical principles of the Declaration of Helsinki and approved by the Institutional Review Board of the Sixth Hospital of Sun Yat-sen University (No. 2021SLYEC-177).
Consent for publication
All authors are uniformly published.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shang, S., Huang, Y., Zhan, X. et al. The relationship between the prognostic nutritional index and new-onset pneumonia in peritoneal dialysis patients. Int Urol Nephrol 54, 3017–3024 (2022). https://doi.org/10.1007/s11255-022-03233-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-022-03233-1