Abstract
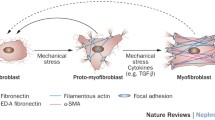
Most inflammatory kidney diseases have the final outcome of fibrosis with the loss of kidney architecture and progressive loss of kidney function. Excess matrix deposition is observed, which may be an inadequate attempt to limit organ damage. The primary sources of matrix synthesis are resident cells that may acquire different activated phenotypes and likely orchestrate matrix deposition. Over the last decades, intense efforts were undertaken to define the origin of myofibroblasts, resulting in four different controversially discussed hypotheses: bone marrow recruitment, vascular pericyte-derived myofibroblasts, epithelial-to-mesenchymal transition (EMT), and endothelial-to-mesenchymal transition (EndMT). In a recent article, LeBleu et al. (Nat Med 19(8):1047–1053, 2013) address this issue and come to the conclusion that most of the different hypotheses are likely true, however to different extents. To arrive at this conclusion, the authors have performed genetic cell tracking and quantification by cell labeling in newly generated knockout mouse models. Quantitative analyses have been made and yield the following estimates: 50 % of the myofibroblasts are derived through proliferation from resident fibroblasts, 35 % differentiate from bone marrow-derived cells, 10 % arise from EndMT, and 5 % through EMT.

Similar content being viewed by others
References
Zhang C et al (2004) Role of connective tissue growth factor in renal tubular epithelial-myofibroblast transdifferentiation and extracellular matrix accumulation in vitro. Life Sci 75(3):367–379
Micallef L et al (2012) The myofibroblast, multiple origins for major roles in normal and pathological tissue repair. Fibrogenesis Tissue Repair 5(Suppl 1):S5
Kalluri R (2009) EMT: when epithelial cells decide to become mesenchymal-like cells. J Clin Invest 119(6):1417–1419
Zeisberg M, Kalluri R (2004) The role of epithelial-to-mesenchymal transition in renal fibrosis. J Mol Med (Berl) 82(3):175–181
Kalluri R, Neilson EG (2003) Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest 112(12):1776–1784
Iwano M et al (2002) Evidence that fibroblasts derive from epithelium during tissue fibrosis. J Clin Invest 110(3):341–350
Picard N et al (2008) Origin of renal myofibroblasts in the model of unilateral ureter obstruction in the rat. Histochem Cell Biol 130(1):141–155
Liu Y (2010) New insights into epithelial-mesenchymal transition in kidney fibrosis. J Am Soc Nephrol 21(2):212–222
Humphreys BD et al (2010) Fate tracing reveals the pericyte and not epithelial origin of myofibroblasts in kidney fibrosis. Am J Pathol 176(1):85–97
Huang F, Chen YG (2012) Regulation of TGF-beta receptor activity. Cell Biosci 2:9
Akhmetshina A et al (2012) Activation of canonical Wnt signalling is required for TGF-beta-mediated fibrosis. Nat Commun 3:735
Rojas A et al (2012) The role played by perivascular cells in kidney interstitial injury. Clin Nephrol 77(5):400–408
Lin SL et al (2008) Pericytes and perivascular fibroblasts are the primary source of collagen-producing cells in obstructive fibrosis of the kidney. Am J Pathol 173(6):1617–1627
Kostadinova-Kunovska S et al (2007) Morphological changes in the tubulointerstitial compartment in primary glomerulopathies. Prilozi 28(2):61–74
Rastaldi MP et al (2002) Epithelial-mesenchymal transition of tubular epithelial cells in human renal biopsies. Kidney Int 62(1):137–146
Zhu C, Mertens PR (2010) Epithelial-mesenchymal transition to be or not to be? Is the answer yes and no at the same time? Int Urol Nephrol 42(3):843–846
Li J, Qu X, Bertram JF (2009) Endothelial-myofibroblast transition contributes to the early development of diabetic renal interstitial fibrosis in streptozotocin-induced diabetic mice. Am J Pathol 175(4):1380–1388
Zhao J, Tramontano A, Makker SP (2007) Albumin-stimulated TGFbeta-1 in renal tubular cells is associated with activation of MAP kinase. Int Urol Nephrol 39(4):1265–1271
Chuang PY, Menon MC, He JC (2013) Molecular targets for treatment of kidney fibrosis. J Mol Med (Berl) 91(5):549–559
LeBleu VS et al (2013) Origin and function of myofibroblasts in kidney fibrosis. Nat Med 19(8):1047–1053
Acknowledgments
P.R.M. is funded by DFG Grants Me1365/7-1 and SFB854 TP1.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ballhause, T.M., Soldati, R. & Mertens, P.R. Sources of myofibroblasts in kidney fibrosis: All answers are correct, however to different extent!. Int Urol Nephrol 46, 659–664 (2014). https://doi.org/10.1007/s11255-013-0626-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-013-0626-5