Abstract
Water bodies can mitigate urban heat island effects and allow terrestrial animals to access water during periods of insufficient precipitation. Because precipitation is expected to become increasingly scarce in many global regions, urban riparian areas in drylands may yield important insight into the dynamic role of water in terrestrial animal communities. Here, we first investigated the role of spatiotemporal variability in water availability in insect biomass and biodiversity in an urban riparian area—particularly, whether seasonality modulates the spatial effects of water availability. High water availability was characterized spatially by proximity to a river, and temporally by high-precipitation seasons. Second, we investigated whether the relationship between biomass and biodiversity shifts due to variation in water availability. We determined the biomass and biodiversity of an insect community across a riparian zone in Stockton, CA, USA, an urban drylands area characterized by hot, dry summers and cool, wet winters. Time (seasonality exhibiting dramatic variation in precipitation) exerted strong effects on ecological indices and a biodiversity-biomass relationship. Space (proximity to a fixed water source) had more modest effects on the community, and there was some evidence that space and time modulated the effects of one another on the community. Urban animal community dynamics may be more sensitive to larger-scale climate patterns than to local, landscape-level factors, and covariation between biomass and biodiversity may be influenced by resource (water) availability. Studying insect communities in the riparian zones of urban drylands may be critical to understanding how animal communities respond to a warmer, drier climate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Temperatures are increasing worldwide, and urban ecosystems are warming faster than other ecosystems due to the urban heat island effect (Wilby 2008; IPCC 2021). Urban waterbodies (i.e., permanent sources of static or flowing water) can mitigate urban heat, and therefore serve as important cooling islands (Murakawa et al. 1991; Hathway and Sharples 2012; Sun et al. 2012; Xue et al. 2019). Urban waterbodies also support biodiversity and provide cultural ecosystems services (reviewed in Hassall 2014; Alikhani et al. 2021). Riparian areas, including those in urban drylands, are particularly important for preserving biodiversity (Naiman and Décamps 1997; Trammell et al. 2011; McCluney and Sabo 2014; Banville et al. 2017; García-Martínez et al. 2017; Ramey and Richardson 2017). In the absence of sufficient precipitation, urban rivers allow terrestrial organisms to access water, a vital resource that is becoming increasingly scarce in many regions due to climate change (Trenberth 2011; Sarhadi et al. 2018; Giorgi et al. 2019; IPCC 2021). Thus, proximity to a river can represent a gradient of resource availability for organisms. Although access to a water source offers advantages (Chamaillé-Jammes et al. 2007; Chen et al. 2015), such high-resource environments may have drawbacks, including increased competition (Kotowski et al. 2006; Pafilis et al. 2009; Laliberté et al. 2013) or predation risk (reviewed in Yuen and Dudgeon 2015).
Precipitation is a strong selective force on terrestrial plants and animals worldwide (Siepielski et al. 2017). It may also modulate the costs and benefits of permanent water sources to organisms whereby the relative value of a permanent water source is reduced by precipitation. Precipitation often varies seasonally, and seasonal climate patterns of precipitation and temperature can synergistically interact to modify the role of permanent water sources to organisms. For example, Mediterranean climates are found across several continents, and they exhibit an inverse relationship between temperature and precipitation (i.e., dry summers and wet winters: Kottek et al. 2006; Stahle et al. 2020). Here, close proximity to a river or pond may have a greater effect on community dynamics during a hot, dry summer than during a cool, wet winter. Yet, the interactive effects of water’s spatial and temporal availability (i.e., proximity to a permanent water source and seasonal precipitation patterns, respectively) on animal communities are not fully understood (but see Yuen and Dudgeon 2015; McCluney et al. 2018).
The biomass and biodiversity of communities are key ecological indicators, and they are related to one another in many communities. For example, the relationship between plant biomass and biodiversity may be positive (reviewed in Wu et al. 2015; Sanaei et al. 2018) or negative (e.g., Gough et al. 1994; Suo et al. 2008), potentially due to a unimodal pattern mediated by resource availability—the relationship between biomass and biodiversity is positive when resources are limited and becomes negative when resources are abundant (Rosenzweig 1995; Paquette and Messier 2011; but see Loreau et al. 2001). However, it is unclear if these patterns are also present in animal communities. Insects are sensitive to precipitation patterns (McCain and Colwell 2011) and are critical to ecosystem functioning (Cardoso et al. 2020), including in urban ecosystems (Hall et al. 2017). Therefore, the biomass-biodiversity relationship in urban insect communities may vary due to variation in resource (water) availability across space and time.
Thus, we addressed two aims by determining the biomass and biodiversity of an urban insect community across a riparian zone in seasons that varied dramatically in temperature and precipitation (Figs. 1 and S1). First, we investigated the role of spatiotemporal variability in water availability in insect biomass and biodiversity—particularly, whether seasonality modulates the spatial effects of water availability. Here, high water availability was characterized spatially by close proximity to a river, and it was characterized temporally by cooler, high-precipitation seasons (i.e., spring and winter: Fig. 1). Second, we investigated the relationship between insect biomass and biodiversity (e.g., positive or negative covariation), and whether the biomass-biodiversity relationship is affected by variation in water availability. Our study will clarify how urban animal communities are affected by variation in water availability, a key resource that is becoming increasingly limited in many geographic regions (Vicente-Serrano et al. 2014; Porkka et al. 2016; Sarhadi et al. 2018; IPCC 2021).
The effect of season on total precipitation and hourly ground temperature (mean ± s.e.m.) during sampling in a riparian zone of the Calaveras River in Stockton, CA, USA. Temperature data were collected by data loggers during sampling (see text), and precipitation data were collected from a weather station (KCASTOCK103) located 1 km from the field site. Letters above columns denote temperature differences across seasons
Materials and methods
Study site
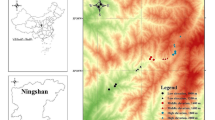
Pitfall traps were placed in the area surrounding the Calaveras River and Wood Memorial Bridge on the University of the Pacific campus (Fig. S1), which has been described previously (Stahlschmidt and Johnson 2018). The campus is located in central Stockton, California, an urban dryland area with a hot‐summer Mediterranean climate (Kottek et al. 2006) and a population of approximately 322,000 people (U.S. Census Bureau 2021).
Insect sampling
In each season (i.e., October 2018, and January, April, and July 2019), insects were sampled using pitfall traps arrays along 10 transects that were 50 m apart (Fig. S2). Each transect started at the edge of the river channel (i.e., 0 m from the river) and extended 20 – 50 m perpendicularly from the river depending on local habitat features. For example, transects spanning paved pedestrian paths were avoided thereby making some transects shorter than others (Fig. S2). Trap arrays along the transects were placed 5 m apart at 0 m to 20 m from the river, and generally 10 m apart at 20 m to 50 m from the river (Fig. S2).
Each trap array included three traps to allow for replication and to account for potential disturbance (e.g., human interference). Traps in each trap array were arranged 1 m from each other in a linear fashion parallel to the river. The pitfall traps were 150 mL (7 cm height and 6 cm diameter) plastic specimen containers buried flush with the ground surface ≥ 1 week prior to sampling. During sampling, each trap was half-filled with a 1:1 solution of water and a propylene glycol-based antifreeze (SIERRA® antifreeze-coolant) that served as a killing agent and preservative. When traps were opened and filled, a temperature data logger (HOBO Pendant, Onset Corp.) was placed on the ground at each trap array to record (and account for) hourly temperature during sampling. After 5 d of sampling, traps and data loggers were collected, and specimen containers were capped and stored until processing.
Insect processing
The solid contents of each trap were separated from the solution using a fine cheesecloth. Insects were then counted and identified to family using a dissecting microscope. Insect abundance and family richness were determined. Simpson’s index of diversity (SID) was also determined as an index of biodiversity because it is directly proportional to true diversity (Jost 2006). Insects were then dried to a constant mass at 50 °C before being weighed to determine the dry mass of insects in each trap.
Statistical analyses
Data were tested for normality, natural logarithm-transformed when necessary, and analyzed using SPSS (v.26 IBM Corp., Armonk, NY) or Past 4.10 (Hammer et al. 2001). Two-tailed significance was determined at α = 0.05. To examine the independent and interactive effects of distance from water and season, linear mixed model analyses were performed on insect abundance, dry mass, family richness, SID, and mean ground temperature at each trap array. For each mixed model, distance from water (covariate) and season (categorical factor) were included as main effects. For the abundance, dry mass, richness, and SID models, the number of traps at each trap array (n = 1–3, depending on disturbance) was included as a covariate to account for sampling effort. Mean ground temperature was also included as a covariate in these models because ground temperature typically positively covaried with distance from water thereby allowing us to account for temperature when determining the effects of water availability. In addition, a permutational multivariate analysis of variance (PERMANOVA) was used to determine the effects of distance from water and season on community composition. In the PERMANOVA, a Bray–Curtis dissimilarity index was used, and 9,999 permutations were performed.
Metrics of insect biomass (i.e., dry mass and abundance) were highly correlated with one another (R = 0.68; P < 0.001), as were metrics of insect biodiversity (i.e., family richness and SID: R = 0.81; P < 0.001). Therefore, dry mass and family richness were analyzed together to determine the effects of distance from water and season on the relationship between biomass and biodiversity. Here, a linear mixed model was performed on family richness at each trap array. Dry mass (covariate) was included as a fixed effect to determine the overall biomass-biodiversity relationship. Interactions between dry mass and distance from water, and between dry mass and season were also included to assess the role of spatial and temporal variation of water availability on the biomass-biodiversity relationship. Mean ground temperature was included as a covariate to account for temperature effects on community metrics. All linear mixed models included transect identity (n = 10) as a random effect. Pairwise post-hoc analyses used the Holm-Bonferroni method to control the Type I error rate associated with multiple comparisons.
Results
In total, 4506 individuals across 40 families were collected, and the dominant taxon often varied by season (Table 1; Fig. S3). Fall and winter collections were dominated by ground beetles (Carabidae) while ants (Formicidae) and bordered plant bugs (Largidae) were most common in spring and summer, respectively (Table 1). The similarity of community assemblages also varied by season—spring and summer were most similar, while summer and fall were least similar (F3,199 = 23, P = 0.0001) (Fig. S3). Community composition was also influenced by distance from water (F10,199 = 1.3, P = 0.017), but not by a distance × season interaction (F30,199 = 0.76, P = 0.57). Ground temperature was influenced by season (Fig. 1; F3,189 = 376, P < 0.001), and it was also influenced by distance from water (F1,196 = 35, P < 0.001) and by a distance × season interaction (F3,189 = 2.4, P < 0.001) where temperatures were higher farther from the water in each season other than spring.
Insect abundance was affected by season (spring > summer > fall = winter; F3,184 = 26, P < 0.001), the number of traps at each array (positive covariation: F1,196 = 17, P = 0.009), and ground temperature (positive covariation: F1,162 = 7.7, P = 0.006), but not by distance from water (F1,161 = 1.9, P = 0.17) or by a distance × season interaction (F3,190 = 1.8, P = 0.15) (Fig. 2a). Insect dry mass was also influenced by season (spring = summer = fall > winter; F3,182 = 11, P < 0.001), the number of traps at each array (positive covariation: F1,191 = 6.8, P = 0.010), and ground temperature (positive covariation: F1,179 = 8.5, P = 0.004), and it was also unaffected by distance from water (F1,178 = 0.088, P = 0.77) (Fig. 2b). However, it was influenced by an interaction between distance from water and season (highest at intermediate distances during wetter seasons: F3,187 = 3.5, P = 0.017) (Fig. 2b).
Effects of distance from water across seasons on a insect abundance and b insect biomass in a riparian zone along the Calaveras River in Stockton, CA. Values are displayed as mean ± s.e.m. for data from transects (n = 10) of pitfall trap arrays where the grayscale of symbols indicates seasonal climate patterns—from black (wet and cool) to white (dry and hot)
Insect family richness was affected by season (spring > summer > winter > fall; F3,181 = 37, P < 0.001), and it varied with distance from water (highest at intermediate distances: F1,147 = 4.8, P = 0.030), the number of traps at each array (positive covariation: F1,196 = 6.8, P = 0.010), and ground temperature (positive covariation: F1,150 = 7.1, P = 0.009) (Fig. 3a). The interaction between season and distance did not influence richness (F3,194 = 0.64, P = 0.59). Insect SID was influenced by season (spring = summer > winter > fall; F3,194 = 14, P < 0.001) and positively covaried with the number of traps at each array (F3,194 = 4.0, P = 0.048), but it was not affected by distance from water (F1,194 = 1.5, P = 0.22), a distance × season interaction (F3,194 = 1.4, P = 0.24), or ground temperature (F1,194 = 0.19, P = 0.66) (Fig. 3b).
Effects of distance from water across seasons on a insect family richness and b Simpson’s index of diversity for insects in a riparian zone along the Calaveras River in Stockton, CA. Values are displayed as mean ± s.e.m. for data from transects (n = 10) of pitfall trap arrays where the grayscale of symbols indicates seasonal climate patterns—from black (wet and cool) to white (dry and hot)
Overall, insect family richness positively covaried with insect dry mass (F1,197 = 77, P < 0.001), and this biomass-biodiversity relationship was influenced by season (significant effect of dry mass × season interaction on richness: F3,197 = 86, P < 0.001) where covariation was stronger during the drier seasons of fall and summer (R = 0.75 and R = 0.69, respectively) than during the wetter seasons of winter and spring (R = 0.56 and R = 0.51, respectively) (Figs. 1 and 4). In this model, family richness was not affected by ground temperature (F1,197 = 0.077, P = 0.78) or by a dry mass × distance interaction (F1,197 = 3.0, P = 0.087).
The strength of the relationship between metrics of insect biomass (dry mass) and biodiversity (family richness) varied across seasons in a riparian zone along the Calaveras River in Stockton, CA. Each point represents data from a single trap array, and lines of best fit are shown for each season. The grayscale of symbols indicates seasonal climate patterns—from black (wet and cool) to white (dry and hot). See text for further details
Discussion
Water constrains terrestrial life (Chapin III et al. 2002), and the dynamics by which space and time influence animals’ access to this vital resource are being altered by climate change (Giorgi et al. 2019; Trenberth 2011; Sarhadi et al. 2018; IPCC 2021). Here, we examined the effects of spatiotemporal availability of water on an urban insect community in a drylands riparian zone. Time (seasons varying dramatically in precipitation) strongly affected insect biodiversity, biomass, community composition, and abundance indices, and it also influenced the strength of relationships between some of these important ecological indicators. Conversely, space (proximity to a fixed water source) had more modest effects on the insect community, and there was some evidence that space and time modulated the effects of one another on the community. In sum, our results indicate that urban community dynamics may be more sensitive to larger-scale climate patterns than to local, landscape-level factors, and (to our knowledge) we are the first to report that the covariation between biomass and biodiversity in an animal community can be influenced by resource availability (i.e., seasonal variation in water availability).
Seasonality significantly affects animal communities, and seasonal effects often outweigh the impacts of other ecological drivers, such as invasive species or anthropogenic disturbance (Habel et al. 2018; Stahlschmidt et al. 2018; Escobedo-Kenefic et al. 2020). Like others (e.g., Bolger et al. 2000; Riedel et al. 2008; Banza et al. 2021), we show that metrics of insect biodiversity and abundance were greatest in spring, which was characterized by mild temperatures and relatively wet conditions due to heavy winter rainfall (Fig. 1). In stark contrast to summer and fall, spring was also marked by abundant herbaceous and shrub vegetation (Fig. S1). Such vegetation is the major source of primary productivity in many riparian zones (MacDonald et al. 2012; Parsons and Thoms 2013; Thapa et al. 2016; Cornejo-Denman et al. 2018)—therefore, seasonal precipitation may facilitate bottom-up effects on food availability to the insect community. Yet, the importance of vegetation as a habitat feature extends beyond its role as food. Vegetation creates vertical structure that can increase the abundance and variety of microhabitats, which can facilitate niche partitioning due to an increased availability of space, refuge, and thermal resources for animals, including those in urban riparian zones (Ober and Hayes 2008; Limb et al. 2009; Trammell et al. 2011; Schut et al. 2014; Štokmane and Spuņģis 2016; Banville et al. 2017; García-Martínez et al. 2017; Zaimes et al. 2019; Liu et al. 2021). Thus, determining the role of vegetation in urban insect communities in riparian areas may be critical to better understand the link between seasonal precipitation patterns and animal communities.
Seasonal precipitation patterns are the major determinant of some insect assemblages (reviewed in Grimbacher and Stork 2009). Yet, photoperiod, temperature, and precipitation can all vary seasonally and drive food availability, which, in turn, influences animal communities (Develey and Peres 2000; Boucher-Lalonde et al. 2013; Jones and Gilbert 2016; Humphries et al. 2017; Srinivasan et al. 2018; Escobedo-Kenefic et al. 2020). In tropical ecosystems, variation in temperature and photoperiod are often uncoupled from season, and seasonality is instead characterized largely by patterns of precipitation (Leigh et al. 2004). Although we could not disentangle photoperiod from season in our study, we did account for temperature to better focus on the precipitation-related effects of seasonality on a riparian insect community. Experimental water supplementation influences insect community assemblages independent of season in other temperate urban ecosystems (McCluney et al. 2018). Therefore, we encourage continued investigation into the links between water availability and insect assemblages in non-tropical ecosystems, given the important ecosystem services provided by insects worldwide (Ameixa et al. 2018).
The river tended to mitigate urban heat effects because ground temperatures were lower near the river in most seasons, similar to results in non-dryland urban areas (Murakawa et al. 1991; Hathway and Sharples 2012). However, in contrast to the large effects of season, the proximity to the river (i.e., spatial access to a water resource) only independently affected one metric of the urban insect community—more families were collected at intermediate distances (10–25 m) from the river (Fig. 3a). This “Goldilocks” effect may reflect a balance between the benefits and costs of resource accessibility (i.e., increased water availability vs. increased competition or predation risk, respectively) (Kotowski et al. 2006; Chamaillé-Jammes et al. 2007; Pafilis et al. 2009; Laliberté et al. 2013; Chen et al. 2015; Yuen and Dudgeon 2015). In our study, this effect was season-specific for several community metrics. For example, insect abundance, biomass, richness, and SID all tended to be greatest at intermediate distances in the wetter seasons of winter and spring (Figs. 2 and 3). This may indicate that the hydric and thermal benefits of river proximity were reduced during the spring, in particular, because it was characterized by wet conditions throughout the riparian zone (Figs. 1 and S1), as well as a lack of thermal buffering provided by the river (i.e., spring was the only season when temperatures were not lower near the river). Being close to water (0–5 m from river) may have been relatively costly to community metrics in the wettest season (winter) and most beneficial in the driest season (summer) (Figs. 2 and 3). Regarding our first aim (role of spatiotemporal variability in water availability in an insect community), our results indicate that seasonality can modulate the costs and benefits of proximity to water in an urban animal community.
Our second aim addressed the relationship between biomass and biodiversity in urban insects. This relationship is theorized to be negative in plants when resources are abundant due to increased competition, but the relationship becomes positive when resources are limited (Rosenzweig 1995; Paquette and Messier 2011; but see Loreau et al. 2001). Our study demonstrated a positive biomass-biodiversity relationship across all seasons (Fig. 4), indicating that our study site may have been resource-limited. Alternatively, the biomass-biodiversity relationship in animal communities may be controlled by different dynamics than it is in plant communities, which is feasible given animal and plant communities respond differently to other ecological drivers (e.g., climate: Schleuning et al. 2016; Bruckerhoff et al. 2020). Nonetheless, our results do suggest that resource (water) availability may play a role in this relationship for animals. The covariation of dry mass (biomass) and family richness (biodiversity) was stronger during the drier (lower-resource) seasons of fall and summer than during the wetter (higher-resource) seasons of winter and spring in a riparian insect community. However, future work in other ecosystems is required to better understand the factors driving the biomass-biodiversity relationship in animal communities. Further, examining more specific taxonomic information (e.g., genus- or species-level) may provide new insight into the dynamics underlying this relationship.
Climate change will continue to dramatically alter the availability of water in many geographic regions that include urban environments characterized by higher temperatures (Wilby 2008; Vicente-Serrano et al. 2014; Porkka et al. 2016; Sarhadi et al. 2018; IPCC 2021). Water limitation tends to reduce fitness-related traits in insects (e.g., Branson 2016; Padda and Stahlschmidt 2022), and insects are critical to terrestrial ecosystems (Cardoso et al. 2020). In our study, summer was characterized by an absence of precipitation in the riparian zone (Fig. 1), and by relatively high insect biodiversity and biomass metrics. Limited water access may have reduced competition and opened niches for certain taxa—twice as many unique insect families were collected in the hot, dry summer compared to the combined total for fall, winter, and spring (Table 1; Fig. S3). Despite its costs to individuals, water stress can facilitate insect biodiversity by weakening negative interspecific interactions or strengthening positive interactions (Staley et al. 2007; Pringle et al. 2013; but see McCluney and Sabo 2012). For example, insect species richness can be greater in hotter, drier areas of urban dryland environments (Adams et al. 2020). Clearly, drought and water limitation have key roles in shaping insect populations and communities. Therefore, studying insect communities in dryland riparian zones (e.g., McCluney and Sabo 2012; 2014), particularly in urban areas, may provide important insight into how insect communities (and the ecosystems they service) respond to the world’s future climate.
Data availability
Data will be uploaded to FigShare upon acceptance for publication.
Code availability
Not applicable.
References
Adams BJ, Li E, Bahlai CA, Meineke EK, McGlynn TP, Brown BV (2020) Local- and landscape-scale variables shape insect diversity in an urban biodiversity hot spot. Ecol Appl 30(4)
Alikhani S, Nummi P, Ojala A (2021) Urban wetlands: A review on ecological and cultural values. Water 13(22)
Ameixa OMCC, Soares AO, Soares AMVM, Lillebø AI (2018) Ecosystem Services Provided by the Little Things That Run the World. In: Selected Studies in Biodiversity (eds. B Şen, O Grillo). ISBN 978–1–78923–233–2
Banville MJ, Bateman HL, Earl SR, Warren PS (2017) Decadal declines in bird abundance and diversity in urban riparian zones. Landsc Urban Plann 159:48–61
Banza P, Evans DM, Medeiros R, Macgregor CJ, Belo ADF (2021) Short-term positive effects of wildfire on diurnal insects and pollen transport in a Mediterranean ecosystem. Ecol Entomol 46(6):1353–1363
Bolger DT, Suarez AV, Crooks KR, Morrison SA, Case TJ (2000) Arthropods in urban habitat fragments in southern California: Area, age, and edge effects. Ecol Appl 10(4):1230–1248
Boucher-Lalonde V, Kerr JT, Currie DJ (2013) Does climate limit species richness by limiting individual species' ranges? Proc R Soc B Biol Sci 281(1776)
Branson DH (2016) Drought impacts on competition in Phoetaliotes nebrascensis (Orthoptera Acrididae) in a northern mixed grassland. Environ Entomol 45(2):492–499
Bruckerhoff LA, Connell RK, Guinnip JP, Adhikari E, Godar A, Gido KB, Boyle AW, Hope AG, Joern A, Welti E (2020) Harmony on the prairie? Grassland plant and animal community responses to variation in climate across land-use gradients. Ecology 101(5):e02986
Cardoso P, Barton PS, Birkhofer K, Chichorro F, Deacon C, Fartmann T, Fukushima CS, Gaigher R, Habel JC, Hallmann CA, Hill MJ, Hochkirch A, Kwak ML, Mammola S, Ari Noriega J, Orfinger AB, Pedraza F, Pryke JS, Roque FO, Settele J, Simaika JP, Stork NE, Suhling F, Vorster C, Samways MJ (2020) Scientists' warning to humanity on insect extinctions. Biol Conserv 242
Chamaillé-Jammes S, Valeix M, Fritz H (2007) Managing heterogeneity in elephant distribution: Interactions between elephant population density and surface-water availability. J Appl Ecol 44(3):625–633
Chapin FS III, Matson PA, Mooney HA, Vitousek PM (2002) Principles of Terrestrial Ecosystem Ecology. Springer
Chen Y, Cao K, Schnitzer SA, Fan Z, Zhang J, Bongers F (2015) Water-use advantage for lianas over trees in tropical seasonal forests. New Phytol 205(1):128–136
Cornejo-Denman L, Romo-Leon JR, Castellanos AE, Diaz-Caravantes RE, Moreno-Vázquez JL, Mendez-Estrella R (2018) Assessing riparian vegetation condition and function in disturbed sites of the arid northwestern Mexico. Land 7(1)
Develey PF, Peres CA (2000) Resource seasonality and the structure of mixed species bird flocks in a coastal atlantic forest of southeastern brazil. J Trop Ecol 16(1):33–53
Escobedo-Kenefic N, Landaverde-González P, Theodorou P, Cardona E, Dardón MJ, Martínez O, Domínguez CA (2020) Disentangling the effects of local resources, landscape heterogeneity and climatic seasonality on bee diversity and plant-pollinator networks in tropical highlands. Oecologia 194(3):333–344
García-Martínez MÁ, Valenzuela-González JE, Escobar-Sarria F, López-Barrera F, Castaño-Meneses G (2017) The surrounding landscape influences the diversity of leaf-litter ants in riparian cloud forest remnants. PLoS ONE 12(2)
Giorgi F, Raffaele F, Coppola E (2019) The response of precipitation characteristics to global warming from climate projections. Earth Syst Dyn 10(1):73–89
Gough L, Grace JB, Taylor KL (1994) The relationship between species richness and community biomass: The importance of environmental variables. Oikos 70(2):271–279
Grimbacher PS, Stork NE (2009) Seasonality of a diverse beetle assemblage inhabiting lowland tropical rain forest in Australia. Biotropica 41(3):328–337
Habel JC, Seibold S, Ulrich W, Schmitt T (2018) Seasonality overrides differences in butterfly species composition between natural and anthropogenic forest habitats. Anim Conserv 21(5):405–413
Hall DM, Camilo GR, Tonietto RK, Ollerton J, Ahrné K, Arduser M et al (2017) The city as a refuge for insect pollinators. Conserv Biol 31(1):24–29
Hammer O, Harper D, Ryan P (2001) PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol Electron 4:1–9
Hassall C (2014) The ecology and biodiversity of urban ponds. Wiley Interdiscip Rev: Water 1(2):187–206
Hathway EA, Sharples S (2012) The interaction of rivers and urban form in mitigating the Urban Heat Island effect: A UK case study. Build Environ 58:14–22
Humphries MM, Studd EK, Menzies AK, Boutin S (2017) To everything there is a season: Summer-to-winter food webs and the functional traits of keystone species. Integr Comp Biol 57(5):961–976
IPCC (2021) Climate change 2021: The physical science basis. Contribution of working group I to the sixth assessment report of the intergovernmental panel on climate change [Masson-Delmotte V, Zhai P, Pirani A, Connors SL, Péan C, Berger S, Caud N, Chen Y, Goldfarb L, Gomis MI, Huang M, Leitzell K, Lonnoy E, Matthews JBR, Maycock TK, Waterfield T, Yelekçi O, Yu R, Zhou B (eds)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, In press
Jost L (2006) Entropy and diversity. Oikos 113(2):363–375
Jones NT, Gilbert B (2016) Changing climate cues differentially alter zooplankton dormancy dynamics across latitudes. J Anim Ecol 85(2):559–569
Kotowski W, Thörig W, Van Diggelen R, Wassen MJ (2006) Competition as a factor structuring species zonation in riparian fens - A transplantation experiment. Appl Veg Sci 9(2):231–240
Kottek M, Grieser J, Beck C, Rudolf B, Rubel F (2006) World map of the Köppen-Geiger climate classification updated. Meteorol Z 15(3):259–263
Laliberté E, Lambers H, Norton DA, Tylianakis JM, Huston MA (2013) A long-term experimental test of the dynamic equilibrium model of species diversity. Oecologia 171(2):439–448
Leigh EG Jr, Davidar P, Dick CW, Puyravaud J-P, Terborgh J, Ter Steege H, Wright SJ (2004) Why do some tropical forests have so many species of trees? Biotropica 36(4):447–473
Limb RF, Fuhlendorf SD, Townsend DE (2009) Heterogeneity of thermal extremes: Driven by disturbance or inherent in the landscape. Environ Manage 43(1):100–106
Liu WA, Li LF, Xue YH, Zeng C (2021) Butterfly perception of the environment: The impact of the vertical structure of coastal vegetation on butterfly diversity. J Environ Prot Ecol 22(3):1235–1245
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime JP, Hector A, Hooper DU, Huston MA, Raffaelli D, Schmid B, Tilman D, Wardle DA (2001) Ecology: Biodiversity and ecosystem functioning: Current knowledge and future challenges. Science 294(5543):804–808
MacDonald RL, Burke JM, Chen HYH, Prepas EE (2012) Relationship between aboveground biomass and percent cover of ground vegetation in Canadian boreal plain riparian forests. For Sci 58(1):47–53
McCain CM, Colwell RK (2011) Assessing the threat to montane biodiversity from discordant shifts in temperature and precipitation in a changing climate. Ecol Lett 14(12):1236–1245
McCluney KE, George T, Frank SD (2018) Water availability influences arthropod water demand, hydration and community composition on urban trees. J Urban Ecol 4(1)
McCluney KE, Sabo JL (2014) Sensitivity and tolerance of riparian arthropod communities to altered water resources along a drying river. PLoS ONE 9(10)
McCluney KE, Sabo JL (2012) River drying lowers the diversity and alters the composition of an assemblage of desert riparian arthropods. Freshw Biol 57(1):91–103
Murakawa S, Sekine T, Narita K, Nishina D (1991) Study of the effects of a river on the thermal environment in an urban area. Energy Build 16(3–4):993–1001
Naiman RJ, Décamps H (1997) The ecology of interfaces: Riparian zones. Ann Rev Ecol Syst 28:621–658
Ober HK, Hayes JP (2008) Influence of forest riparian vegetation on abundance and biomass of nocturnal flying insects. For Ecol Manage 256(5):1124–1132
Padda SS, Stahlschmidt ZR (2022) Evaluating the effects of water and food limitation on the life history of an insect using a multiple-stressor framework. In Press, Oecologia
Pafilis P, Meiri S, Foufopoulos J, Valakos E (2009) Intraspecific competition and high food availability are associated with insular gigantism in a lizard. Naturwissenschaften 96(9):1107–1113
Paquette A, Messier C (2011) The effect of biodiversity on tree productivity: From temperate to boreal forests. Global Ecol Biogeogr 20(1):170–180
Parsons M, Thoms MC (2013) Patterns of vegetation greenness during flood, rain and dry resource states in a large, unconfined floodplain landscape. J Arid Environ 88:24–38
Porkka M, Gerten D, Schaphoff S, Siebert S, Kummu M (2016) Causes and trends of water scarcity in food production. Environ Res Lett 11(1)
Pringle EG, Akçay E, Raab TK, Dirzo R, Gordon DM (2013) Water stress strengthens mutualism among ants, trees, and scale insects. PloS Biol 11(11)
Ramey TL, Richardson JS (2017) Terrestrial invertebrates in the riparian zone: Mechanisms underlying their unique diversity. Bioscience 67(9):808–819
Riedel R, Marinoni RC, Martins-Opolis N (2008) Spatio-temporal trends of insect communities in southern Brazil. J Entomol 5(6):369–380
Rosenzweig ML (1995) Species Diversity in Space and Time. Cambridge University Press, New York
Sanaei A, Ali A, Chahouki MAZ, Jafari M (2018) Plant coverage is a potential ecological indicator for species diversity and aboveground biomass in semi-steppe rangelands. Ecol Indic 93:256–266
Sarhadi A, Ausín MC, Wiper MP, Touma D, Diffenbaugh NS (2018) Multidimensional risk in a nonstationary climate: Joint probability of increasingly severe warm and dry conditions. Sci Adv 4(11)
Schleuning M, Fründ J, Schweiger O, Welk E, Albrecht J, Albrecht M, Beil M, Benadi G, Blüthgen N, Bruelheide H, Böhning-Gaese K, Dehling DM, Dormann CF, Exeler N, Farwig N, Harpke A, Hickler T, Kratochwil A, Kuhlmann M, Kühn I, Michez D, Mudri-Stojní S, Plein M, Rasmont P, Schwabe A, Settele J, Vujić A, Weiner CN, Wiemers M, Hof C. (2016) Ecological networks are more sensitive to plant than to animal extinction under climate change. Nat Commun 7
Schut AGT, Wardell-Johnson GW, Yates CJ, Keppel G, Baran I, Franklin SE, Hopper SD, Van Niel KP, Mucina L, Byrne M. (2014) Rapid characterisation of vegetation structure to predict refugia and climate change impacts across a global biodiversity hotspot. PLoS ONE 9(1)
Siepielski AM, Morrissey MB, Buoro M, Carlson SM, Caruso CM, Clegg SM, Coulson T, DiBattista J, Gotanda KM, Francis CD, Hereford J, Kingsolver JG, Augustine KE, Kruuk LEB, Martin RA, Sheldon BC, Sletvold N, Svensson EI, Wade MJ, MacColl ADC (2017) Precipitation drives global variation in natural selection. Science 355(6328):959–962
Srinivasan U, Elsen PR, Tingley MW, Wilcove DS (2018) Temperature and competition interact to structure Himalayan bird communities. Proc R Soc B Biol Sci 285(1874)
Stahle DW, Cook ER, Burnette DJ, Torbenson MCA, Howard IM, Griffin D, Villanueva Diaz J, Cook BI, Williams AP, Watson E, Sauchyn DJ, Pederson N, Woodhouse CA, Pederson GT, Meko D, Coulthard B, Crawford CJ (2020) Dynamics, variability, and change in seasonal precipitation reconstructions for North America. J Clim 33(8):3173–3195
Stahlschmidt ZR, Johnson D (2018) Moving targets: Determinants of nutritional preferences and habitat use in an urban ant community. Urban Ecosyst 21(6):1151–1158
Stahlschmidt ZR, Walman RM, Mills AM (2018) Red imported fire ants (Solenopsis invicta) and seasonality influence community refuge use. Biol Invasions 20(10):2849–2859
Staley JT, Mortimer SR, Morecroft MD, Brown VK, Masters GJ (2007) Summer drought alters plant-mediated competition between foliar- and root-feeding insects. Global Change Biol 13(4):866–877
Štokmane M, Spuņģis V (2016) The influence of vegetation structure on spider species richness, diversity and community organization in the Apšuciems calcareous fen. Latvia Anim Biodiversity Conserv 39(2):221–236
Sun Q, Wu Z, Tan J (2012) The relationship between land surface temperature and land use/land cover in Guangzhou, China. Environ Earth Sci 65(6):1687–1694. https://doi.org/10.1007/s12665-011-1145-2
Suo A-, Ju T-, Ge J- (2008) Relationship between species richness and biomass on environmental gradient in natural forest communities on Mt. Xiaolongshan, northwest China. For Stud China 10(4):212–9
Thapa R, Thoms MC, Parsons M (2016) The response of dryland floodplain vegetation productivity to flooding and drying. J Arid Environ 129:42–55
Trammell EJ, Weisberg PJ, Bassett S (2011) Avian response to urbanization in the arid riparian context of Reno, USA. Landsc Urban Plann 102(2):93–101
Trenberth KE (2011) Changes in precipitation with climate change. Clim Res 47(1–2):123–138
U.S. Census Bureau (2021) QuickFacts: Stockton city, California. Retrieved from https://www.census.gov/quickfacts/stocktoncitycalifornia
Vicente-Serrano SM, Lopez-Moreno J-, Beguería S, Lorenzo-Lacruz J, Sanchez-Lorenzo A, García-Ruiz JM, Azorin-Molina C, Morán-Tejeda E, Revuelto J, Trigo R, Coelho F, Espejo F (2014) Evidence of increasing drought severity caused by temperature rise in southern Europe. Environ Res Lett 9(4)
Wilby RL (2008) Constructing climate change scenarios of urban heat island intensity and air quality. Environ Plann B Plann Des 35(5):902–919
Wu X, Wang X, Tang Z, Shen Z, Zheng C, Xia X, Fang J (2015) The relationship between species richness and biomass changes from boreal to subtropical forests in China. Ecography 38(6):602–613
Xue Z, Hou G, Zhang Z, Lyu X, Jiang M, Zou Y et al (2019) Quantifying the cooling-effects of urban and peri-urban wetlands using remote sensing data: Case study of cities of Northeast China. Landsc Urban Plann 182:92–100
Yuen EYL, Dudgeon D (2015) Spatio-temporal variability in the distribution of ground-dwelling riparian spiders and their potential role in water-to-land energy transfer along Hong Kong forest streams. PeerJ 2015(7)
Zaimes GN, Loisios P, Fytopoulos P, Mersina C, Fyllas N, Iakovoglou V, Avtzis D (2019) Ground dwelling insects as environmental indicators of riparian habitats in agricultural Mediterranean landscapes. Environ Eng Manage J 18(9):1977–1986
Acknowledgements
We appreciate Louis Kim, Christine Le, and Jacob Whitlock for assisting with fieldwork.
Funding
Open access funding provided by SCELC, Statewide California Electronic Library Consortium. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
AB and ZRS conceived the ideas and designed methodology; all authors collected the data; ZRS analyzed the data; AB and ZRS led the writing of the manuscript; and all authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no relevant financial or non-financial interests to disclose.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bonfoey, A., Padda, S.S. & Stahlschmidt, Z. Spatiotemporal variation in water availability drives insect community dynamics in an urban riparian zone. Urban Ecosyst 26, 1309–1317 (2023). https://doi.org/10.1007/s11252-023-01375-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-023-01375-3