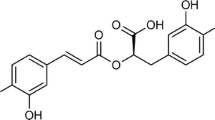
Abstract
Coffea spp. are remarkable sources of phytochemicals, but the lack of a well-defined culture medium aimed at the induction of non-embryogenic and friable callus hampers the establishment of plant cell suspension cultures for large-scale production of valuable compounds. In this paper, we describe a one-medium protocol suitable to obtain both callus and cell suspension cultures from leaves of two elite cultivars of C. arabica. The protocol was developed through an iterative process involving the determination of the best concentration of auxin and cytokinin, their optimal ratio, as well as the most effective molecule of either hormone class. Young leaves were found to be a good and easy-to-use explant source for callus induction and proliferation, provided that a cytokinin was present in association with a chlorinated auxin in a full strength, semi-solid MS medium. The best results were obtained by hormone concentration and combination of 1 mg/L of both kinetin and 2,4,5-trichlorophenoxyacetic acid. The same ratio of these growth regulators was conveniently used for the development and stabilization of cell suspension cultures in liquid MS medium. When grown in darkness, stabilized suspension cultures showed a fine and homogeneous texture, with a 10-fold biomass increase within 25 days and a cell viability > 90%. In addition, the phytochemical profile revealed the presence of the most widely studied coffee compounds. The protocol can be applied to obtain adequate amounts of cell biomass for use in physiological studies concerning the production of secondary metabolites.
Key messages
A protocol for callus induction and undifferentiated cell suspensions establishment in Coffea arabica was developed. This technique represents a useful tool for in vitro production of remarkable phytochemicals from coffee.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coffee is one of the most economically important crops, with Coffea arabica and C. canephora representing the two main cultivated species (Wintgens 2012). Its significance goes beyond the popular beverage, as it is a major source of phytochemicals including not only caffeine and chlorogenic acids, but also diterpenes, trigonelline, proanthocyanidins, ferulic-, caffeic- and p-coumaric acids (Moeenfard et al. 2016; Munyendo et al. 2021). Among these secondary metabolites (SMs), some are typical of the coffee plant and, according to the evidence collected so far, they could be applied in human therapy against cancer, diabetes, cardiovascular diseases, and obesity (Munyendo et al. 2021).
As it occurs for other medicinal plants, large-scale production of SMs through extraction and purification from field harvested material can represent a challenge due to a number of concurring reasons, such as the low concentration in the collected organ(s), the presence of chemically similar compounds, or seasonal variability caused by biotic and/or abiotic stresses. To circumvent difficulties in the effective use of wild stands or crops, chemical synthesis has often been suggested, but in many instances such practice, although technologically possible, appeared costlier as compared to extraction and purification from natural sources. Actually, many SMs are the outcome of complex enzymatic pathways that are difficult to reproduce in vitro.
Cell suspension cultures (CSCs) provide a further technological option for large-scale production of high quality and standard SMs of plant origin in World Health Organization list (2023) and references therein. This system, potentially coupled with strategies such as elicitation by exogenously applied substances or metabolic engineering, has already increased the productivity of some SMs exploitable for the treatment of life-threatening diseases. CSCs of Gymnema sylvestre were successfully used to increase the synthesis of the anti-diabetic agent gymnemic acid through the modification of macro-elements in the liquid medium (Praveen et al. 2011). Similarly, hormones such as picloram, IBA and BAP strongly enhanced the production of anti-HIV dipyranocoumarins in Calophyllum inophyllum CSCs (Pawar et al. 2011). Finally, the addition of methyl jasmonate to media adjusted for CSCs of Taxus canadensis (Linden and Phisalaphong 2000) and T. cuspidata (Mirjalili and Linden 1996) significantly enhanced the industrial production of taxol, one of the most successful anticancer drugs.
To establish a stabilized cell line of a plant, the initial step typically involves callus induction from tissues. In Coffea spp., the main explant sources used for such purpose are orthotropic internodes (Frischknecht and Baumann 1980; Baumann and Röhrig 1989), seeds (Furuya et al. 1990; Kurata et al. 1991) and leaves (Herman and Haas 1975; Ogita et al. 2004). Friable calluses were inoculated in liquid media to perform short-term experiments or long-term maintenance. In the former case, a pre-cultivation period was performed to split callus masses into small aggregates, which were eventually collected to obtain the main culture targeted to provide material for SMs analysis (Kurata et al. 1991, 1998; Sartor and Mazzafera 2000; Gomes-Junior et al. 2006, 2007). Conversely, fine cell suspensions are critical for the development and maintenance of productive cell lines in molecular farming. Indeed, long-term CSCs were established by Baumann and Röhrig (1989), Frischknecht and Baumann (1980) and Townsley (1974), but the available information in respect to critical factors (e.g., basal medium, hormone type and concentration, aggregates presence and size) is largely incomplete. In particular, no systematic study was conducted to investigate the effect of different concentrations and types of auxins and cytokinins on friable callus induction. In addition, the transition from solid to liquid culture was accompanied by a change in PGRs (Plant Growth Regulators), without clear explanatory reasons.
The results presented in this paper could be applied to overcome the existing technological constraints in friable, non-embryogenic callus induction and CSCs development, thus simplifying functional studies on coffee SMs and their molecular farming.
Materials and methods
Chemicals
2,4,5-trichlorophenoxyacetic acid (2,4,5-T), 2,4-dichlorophenoxyacetic acid (2,4-D), indole-3-acetic acid (IAA), indol-3-butyric acid (IBA), α-naphthaleneacetic acid (NAA), β-naphthoxyacetic acid (βNOA), p-clorophenoxyacetic acid (4-CPA), dicamba (Di), 6-benzylaminopurine (BAP), kinetin (KT), thidiazuron (TDZ) and myo-inositol were purchased from Duchefa Biochemie (Harleem, Netherlands); 1,3-diphenylurea (DPHU), 2-isopentenyladenine (2-iP), N-benzyl-9-(tetrahydropyranyl)-adenine (BPA), zeatin (ZT), dihydrozeatin (DHZ), adenine hemisulfate (AHS) and sucrose were purchased from Sigma-Aldrich Chemie (Steinheim, Germany). Bacto agar was obtained from Difco Laboratories (Michigan, United States). Tween 80 was purchased from Labelians (Nemours, France). Acetonitrile, methanol, formic acid (HPLC analytical grade), trigonelline, theobromine and theophylline were purchased from Sigma Aldrich (Saint Louis, MO, USA), chlorogenic acid (trans-5-caffeoylquinic acid) was purchased from PhytoLab (Vestenbergsgreuth, Germany). Water was obtained from a milliQ system (Merck Millipore, Molsheim, France).
Plant material
Young, fully expanded leaves selected from the terminal node of orthotropic branches of greenhouse-grown Red Bourbon and Castillo plants were used as explant sources for in vitro callogenesis. Immediately after harvest, leaves were washed briefly in running tap water and cleaned with a commercial detergent. Surface sterilization was carried out under a laminar flow hood by soaking leaves for 30 s in 95% (v/v) EtOH and 5 min in a 0.2% (w/v) NaOCl solution with Tween 80 (5 drops in 500 mL), finally the leaves were rinsed three times with sterile distilled water. Treated leaves were blotted between layers of 150-mm filter paper (Whatman Grade 1) and punched amidst the secondary veins with a sterile cork borer (Ø 5 mm) to detach explants of uniform shape.
Culture media for callus induction
Leaf discs were placed on a full-strength MS medium (Murashige and Skoog 1962) enriched with sucrose (30 g/L) and myo-inositol (1 g/L), and solidified with Bacto agar (8 g/L). The pH of the medium was adjusted to 5.8 before autoclaving at 121 °C for 20 min. To prevent inactivation, PGRs were added by sterile filtration after autoclaving, when the temperature of the medium was around 50 °C. To investigate both the optimal concentration and type of PGRs for friable callus induction, trials were arranged in three steps: (1) a factorial experiment was carried out in order to determine the optimal concentration and ratio between 2,4-D and KT, selected as the most cited PGRs for callus induction in C. arabica. Leaf explants were placed onto 15 culture media resulting from the combination of 3 concentrations of 2,4-D (1, 2, and 3 mg/L) with 5 concentrations of KT (0, 0.5, 1, 2, and 4 mg/L); (2) in order to determine the optimal cytokinin for callus induction, culture media were supplemented with 1 mg/L 2,4-D and 1 mg/L of a cytokinin chosen from the following: KT, AHS, BPA, ZT, DHZ, 2-iP, TDZ, BAP, and DPHU; (3) based on the results of previous trials, the optimal auxin for callus induction was evaluated in culture media supplemented with 1 mg/L KT and 1 mg/L of an auxin selected from the following: 2,4-D, NAA, IBA, IAA, 4-CPA, βNOA, Di, and 2,4,5-T.
All experiments were conducted in three replicates, with one replicate consisting of 15 leaf discs for both Red Bourbon and Castillo. Cultures were maintained in the dark at 28 °C for 42 days, and on the 21st day, explants were transferred onto a fresh medium with the same composition.
At the end of the experiments, callus induction was evaluated by weighing the explant and the callus together (fresh weight, FW) with a high precision balance (Gibertini elettronica, Milan, Italy) under aseptic conditions. Four classes were established to define callus texture: watery, friable, compact and mixed (friable and compact callus jointly present).
Cell suspension cultures
Red Bourbon and Castillo cell suspension cultures were grown in the presence of combinations of 2,4,5-T (1 mg/L) or 2,4-D (1 mg/L) with 0.5 mg/L or 1 mg/L KT. CSCs were started by transferring 2 g FW of 21-day old calluses into 250 mL Erlenmeyer baffled flasks (Corning, New York, USA) with vent cap, each containing 50 mL MS medium supplemented with the aforementioned PGRs, 30 g/L sucrose and 1 mg/L myo-inositol. CSCs were agitated on a rotary shaker (110 rpm, 26 ± 1 °C, in the dark) (MaxQ600, Thermo Fisher Scientific, Ohio, United States) and subcultured every 10–12 days by transferring 10 mL of suspension to 40 mL of fresh medium. Three replicates, each represented by an independent flask, were designed for all treatments. At the end of the third subculture, the highest performing treatment was evaluated as cell fresh weight (FW) and consequently selected to assess the time course of CSCs. Both FW per mL CSC and cell viability were evaluated in triplicate, at the inoculation time and at subsequent intervals. 1 g FW of cells was used as inoculum. Fluorescein diacetate (FDA) staining (McCabe and Leaver 2000) was selected as viability probe. A 0.5% stock solution of FDA was prepared in acetone and stored at -20 °C until use. Before testing, the solution was diluted 20-fold with phosphate-buffered saline. Two µL of the final solution were added to 0.1 mL CSC, and the samples were gently mixed by inversion for 5 min at room temperature in the dark. Using a fluorescent microscope (Zeiss Axio Observer Z1 with 25 HE filter, Oberkochen, Germany), the percentage of viable cells was calculated by dividing the FDA-stained cells by the total cell count, which was assessed under visible light.
Phytochemical profiles
A major compounds characterization was carried out for CSCs of both cvs., grown in darkness and under a 12/12 photoperiod. Light was provided by fluorescent lamps (Osram Fluora) with a photon flux density of 36 µmol m− 2 s− 1. For each growth condition, 5 independent replicates were analyzed. Extraction of phytochemicals was performed as described in Monteiro et al. (2020), with slight modifications: 100 mg of freeze-dried cells was sonicated for 30 min at room temperature (35 kHz, Bandelin sonorex, Berlin, Germany) in 1 mL of MeOH/H2O (70:30, v/v). After overnight extraction under agitation, the samples were centrifuged for 10 min at 10,000 rpm (Allegra X-30 centrifuge, Beckman Coulter, Indianapolis, IN, USA). The supernatant was collected and kept at 4 °C before analysis. Trigonelline, theobromine, caffeine, and chlorogenic acids (3-CQA, 5-CQA, 4-CQA, 3,4-DiCQA, 3,5-DiCQA, 4,5-DiCQA) were successfully separated and identified by UHPLC-DAD: 1290 UHPLC system (Agilent Technologies, Santa Clara, CA, USA) equipped with a degasser, quaternary pump, column thermostat and diode array detector (DAD) operating at 254, 273 and 324 nm. A Kinetex® 2.6 mm XB-C18 100 Å, LC column 100 × 2.1 mm (Phenomenex, Torrance, CA, USA) was used at 30 °C temperature. The elution system (0.5 mL/min) involved two filtered solvents, namely acetonitrile (solvent A) and 0.1% formic acid in water (solvent B) with the following gradient: 0 min, 97% solvent B; 0–8 min, 85%; 8–11 min, 60%; and back to initial conditions. The injection volume was 2 mL. The identification of alkaloids and chlorogenic acids was achieved by comparison of specific retention times of standard solutions; the quantification was done by UV-Vis spectroscopy, using the DAD detector, by monitoring the absorbance at the maximum wavelength for each compound, i.e., 254 nm for trigonelline, 273 nm for theobromine and caffeine, and 324 nm for the CGAs. Each sample was characterized by its mean content of alkaloids and chlorogenic acids, expressed as mg/g of freeze-dried material (dry weight). Calibration curves were prepared in the appropriate range of concentration and the data were fitted using least-square linear regression. Good linearity (R2 > 0.9987), expressed as the determination coefficient, was obtained for all compounds. The limit of detection (LOD) and the limit of quantification (LOQ) previously published were verified.
Statistical analysis
Statistical analysis was performed using RStudio package 2021.09.1 of R software version 4.1.2 (R Core Team 2021). Levene’s and Shapiro-Wilk tests were performed to assess the homogeneity of variance and the normal distribution, respectively. Log transformation was applied to FW data of step 2 and 3 experiments to achieve normal distribution. Data regarding callogenesis were statistically evaluated using three-way (Step 1) or two-way analysis of variance (ANOVA) (Step 2 and 3). The latter analysis was performed also to test the effect of 2,4-D and 2,4,5-T on CSCs growth in the presence of different concentrations of KT. The significance of each source of variation was evaluated using the F-test. In case of significant differences, means were compared with Duncan’s multiple range test at a significance level of p-value ≤ 0.01.
For both cvs., CSC growth curves were fitted using the drda R package (Malyutina et al. 2023); 95% confidence intervals (CIs) of the curve parameters were calculated applying the Confint function. Curve parameters values were considered statistically different when their 95% CIs did not overlap.
Results
Callus induction at different concentrations of 2,4-D and KT
The results shown herein were achieved by collecting 2,430 leaf explants, out of which only 53 (2.2%) were lost due to microbial contamination. In the factorial experiment, in which 2,4-D and KT were applied at increasing concentrations, leaf explants of Castillo and Red Bourbon became swollen and began to develop undifferentiated cells along the cutting edges within 1 week of culture, regardless of medium composition. Nevertheless, after 6 weeks of culture, the amount of callus appeared significantly affected by both coffee genotype and PGRs combination (Table S1); these factors gave rise to significant first and second order interactions. In particular, the presence of KT was crucial for the induction and further development of the callus, as shown in Fig. 1. In both cvs., the lowest FW values were consistently recorded in KT-free media, with significant differences observed in respect to all the remaining media, in both statistical and absolute terms. In addition, the callus masses developed in the absence of KT turned completely brown by the end of the experiment, hindering any possible use for cell line production.
In KT-supplemented media, callus production was generally higher in Red Bourbon, reaching the maximum FW value at a concentration of 2 mg/L 2,4-D and 1 mg/L KT; however, other PGRs concentrations, namely 1 mg/L 2,4-D combined with 1 or 2 mg/L KT, yielded comparable outcomes (Fig. 1). Similarly, in Castillo, callogenesis was highest with 1 mg/L 2,4-D and 1 mg/L KT, but the difference with 1 mg/L 2,4-D and 0.5 mg/L KT was not significant (Fig. 1). Considering the data obtained for both cvs., the PGR combination of 1 mg/L 2,4-D and KT turned out to give more consistent results, also in terms of callus texture, which appeared highly friable. Interestingly, the highest PGR concentrations (e.g., 3 mg/L 2,4-D, 4 mg/L KT) generally resulted in significantly lower FW (Fig. 1).
Cytokinin selection
Nine cytokinins were tested in the presence of 1 mg/L 2,4-D. The type of cytokinin significantly affected both the FW (p < 0.001) (Table S2) and the callus texture. The effect of cytokinin was similar in Red Bourbon and Castillo, as no significant cv. x cytokinin interaction was observed (Table S2). Statistical analysis on aggregated data revealed that FW values in media supplemented with DHZ, BPA, KT and BAP (176.7–160.2 mg) were significantly higher than those achieved with DPHU and AHS (75.7–65.8 mg) (Fig. 2); moreover, the latter PGRs caused the formation of a brownish and watery callus. Amongst the four cytokinins eliciting the highest response, only BAP and KT consistently induced a friable texture in all explants. Consequently, considering all results, none of the eight cytokinins tested against KT exhibited better performance in terms of callus induction level and callus texture.
Auxin selection
The auxin comparison was carried out using a medium containing KT at a fixed concentration of 1 mg/L. The effect of different auxins on FW was highly significant (p < 0.001) (Table S3). Little or no callus was recorded for most leaf explants cultured on βNOA-, IAA-, IBA-, and NAA-enriched media, to such an extent that FW essentially corresponded to the explant weight. It is noteworthy that this result was achieved in both cvs. (Fig. 3). As shown in Fig. 3, the significant interaction between cv. and auxin (p < 0.05) (Table S3) could be explained by the varying responsiveness observed in 4-CPA medium. The highest FW values were attained in Red Bourbon onto 2,4,5-T and Di media (237.4 and 231.4 mg, respectively); similarly, these same media exhibited the best performances in Castillo (174.2 and 206.5 mg, respectively). However, although the effect of the two PGRs on callogenesis was comparable (Fig. 3), a mixed callus texture was observed in Di medium, as opposed to a highly friable callus consistently induced by 2,4,5-T. It should be noted that 2,4-D was found to be less efficient than 2,4,5-T, suggesting the use of the latter hormone in the callus induction medium.
Transition to liquid medium
The higher efficacy of 2,4,5-T compared to 2,4-D was confirmed in the liquid medium (p-value < 0.05) (Table S4); actually, the cell FW reached the highest values in media supplemented with 2,4,5-T (Fig. 4). An increased auxin to cytokinin ratio (w/w) did not result in significantly different effects on CSCs growth, as evidenced by results obtained in the presence of 2,4-D as well (Fig. 4). Consequently, this experiment suggested the application of same PGRs combination selected for callogenesis in cell suspension development and maintenance (Fig. 5).
Mean fresh weight of cell suspension culture (FWcsc, mg/mL) for each PGRs treatment. Auxin concentration (i.e., 2,4-D and 2,4,5-T) was set at 1 mg/L. The experiment was conducted on C. arabica cvs. Red Bourbon and Castillo (aggregated data). Different letters indicate significant differences at a p-value ≤ 0.01, according to Duncan’s multiple range test
Growth curve and cell viability
The growth pattern of CSCs for both cvs. was fitted using a 4-parameter logistic curve (Fig. 6). The equation of the curve, curve parameters values and associated 95% confidence intervals (CIs) are reported in Table 1. Concerning the “f” parameter, which represents the day value at which the CSC growth reached its mid-point, the 95% CIs did not overlap each other (Table 1). The mid-exponential phase was reached at day 14 and 22 for cv. Red Bourbon and Castillo, respectively. Furthermore, a significant difference was also detected in the upper asymptote (“d” parameter) (Table 1). At the stationary phase, Castillo reached a maximum FWCSC of 169.46 mg/mL, while Red Bourbon showed a value of 127.31 mg/mL. Remarkably, cell viability remained stable at high percentages (> 90%) throughout the experiment (Fig. 6).
Growth curves for Castillo (●) and Red Bourbon (○) suspension cultures. Lines represent the model output, while experimental data (FWcsc, mg/mL) are indicated by dots. The inset plot shows the time course of cell viability (percentage of FDA-stained cells on total) in suspension cultures of cv. Castillo (black bar) and Red Bourbon (white bar). Data are means of 3 replicates
Detection of secondary metabolites
As shown in Table 2, UHPLC analyses revealed the presence in our coffee cell suspension cultures of the main phytochemicals typically detected in coffee plant. Growth conditions (dark/photoperiod) did not affect SMs production, except for CGAs, which quantity strongly increased in CSCs of both cvs.
Discussion
The establishment of a productive CSC relies on the induction and growth of a friable callus (Mahood et al. 2022). Undifferentiated cell masses can be artificially produced by wounding plant tissues and culturing them in vitro (Efferth 2019). Although any parts of a plant can be used as an explant, immature organs with a higher content of juvenile cells are generally preferred due to their greater ability to express totipotency (Souza et al. 2014). In our trials, young leaves were selected as explant sources for several reasons, such as the prolonged availability on mother plants, ease of manipulation, effective removal of the microbial load with adequate concentration and timing of bactericidal and fungicidal agents. Actually, the protocol herein described for leaf surface sterilization resulted in a contamination occurrence as low as 2.2%, which was largely compatible with trial prosecution and completion.
In addition to type and stage of the explant source, PGRs, particularly auxins and cytokinins, represent key elements in the context of callus formation as they control specific cell division checkpoints (Ikeuchi et al. 2013). Our protocol involves the use of KT as this hormone appeared the most effective for the development of a callus suitable for establishing coffee cell suspensions. In fact, despite the highest callus formation induced by DHZ (Fig. 2), only KT and BAP favored the production of a white and friable callus. Along with 2-iP, the latter two hormones have been widely applied in callus induction media settled for subsequent somatic embryogenesis in Coffea spp. (Teixeira et al. 2004; Silva et al. 2011; Pádua et al. 2014; Suliansyah et al. 2021; Oliveira et al. 2021). Furthermore, KT has been the most commonly applied cytokinin in coffee cell suspensions (Frischknecht and Baumann 1980, 1985; Sartor and Mazzafera 2000; Gomes-Junior et al. 2006, 2007).
In our trials AHS and DPHU strongly inhibited callus development, unlike DHZ, BPA, KT or BAP. AHS, a known cytokinin precursor, has often been used as a helper PGR in regeneration studies, mostly in association to BAP or KT (van Staden et al. 2008; Khan et al. 2014; Rency et al. 2018). Our results confirm the limited activity of this growth stimulant applied alone and at a concentration (i.e., 1 mg/L) relatively low as compared to those used in the above-mentioned studies. Concerning DPHU, it should be noted that FW was rather poor also with TDZ, another phenyl urea-type cytokinin with recognized stronger activity (Mok et al. 1982; Carra et al. 2006). It appeared, therefore, that in coffee, phenyl-urea derivatives are largely ineffective for callus formation and viability. In either case, the callus was compact, showing brown portions indicative of oxidative damage.
According to our protocol, a cytokinin is critical for callus proliferation. Indeed, after the initial step of callus induction, in all cytokinin-free media the cell mass ceased to grow and turned brown. Several studies have shown that exogenous cytokinins are strictly necessary for mitotic initiation (John et al. 1993; Laureys et al. 1998; Valente et al. 1998; Zhang et al. 2005) by promoting nuclear shuttling of Myb-domain protein 3R4 (MYB3R4), a transcription factor that activates mitotic gene expression (Yang et al. 2021).
Synergic effects were often noted with auxins: Kaur and Kothari (2004) recorded a significant increase in callus FW originated from cultured immature inflorescences of Kodo millet (Paspalum scrobiculatum) by adding KT to 2,4-D. Similarly, callus induction from Oryza sativa mature seeds occurred independently from KT presence, but its growth was significantly enhanced by KT addition (Sidek et al. 2022). Results of our trials on coffee are in full accordance with former evidence on the dual role of cytokinins in cell dedifferentiation and mitotic division; they also indicate the possible presence of non-additive effects caused by associations between cytokinins and auxins on callus induction and growth. Finally, they point out that a correct ratio between the two hormones must be defined to tune the process of friable callus formation and viability.
As shown before, the best FW values were recorded in both cvs. when the exogenous auxin: cytokinin was perfectly balanced (i.e., 1:1) or doubled in favour of the auxin. However, consistent outcomes were observed only when the induction medium contained 1 mg/L of both 2,4-D and KT, making it the selected combination of PGRs in our protocol. These results are in line with the fundamental principles of chemical regulation in tissue culture. Indeed, a pluripotent callus can be obtained by both an equal and a high auxin-to-cytokinin ratio (Skoog and Miller 1957; Ikeuchi et al. 2013; Nakashima et al. 2023). Furthermore, a 1:1 ratio has been selected for callus induction in plant species known as valuable sources of phytochemicals: Kapoor et al. (2019) produced callus from leaf explants of Rhodiola imbricata by using a MS medium supplemented with 3 mg/L of both NAA and BAP; Thacker et al. (2018) determined that a combination of 2 mg/L of both 2,4-D and KT was optimal for callogenesis in four different cvs. of Cannabis sativa; other combinations of auxins and cytokinins giving a 1:1 ratio were also well performing.
A hormonal balance is equally important for callus quality; according to our factorial experiment, friable callus usable as a starting material for the setting up of CSCs or cell line backups was preferably obtained with the administration of 1 mg/L of both 2,4-D and KT; the same hormonal composition was formerly applied by Domínguez et al. (2009) to induce and maintain friable callus from leaves of Magnolia dealbata.
Interestingly, most of the studies aimed at the production of coffee CSC achieved callogenesis through an induction medium conceived by Söndahl and Sharp (1977) for indirect somatic embryogenesis in C. arabica cv. Red Bourbon (Sartor and Mazzafera 2000; Gomes-Junior et al. 2006, 2007; Bottcher et al. 2012). According to our results, this PGRs combination (i.e., 1 mg/L 2,4-D + 4 mg/L KT) was inadequate to sustain friable callus formation and growth (Fig. 1); especially in cv. Castillo, poor FW values and a mixed callus texture were recorded with this medium. Several studies have demonstrated that high cytokinin concentrations can affect cell viability by accelerating cell senescence processes, thereby promoting programmed cell death both in plants and in cultured plant cells (Carimi et al. 2003, 2004; Zottini et al. 2006; Vescovi et al. 2012).
Coupled with KT, 2,4-D is the most extensively used auxin for callus induction in Coffea spp. (Söndahl and Sharp 1977; Sartor and Mazzafera 2000; Gomes-Junior et al. 2006, 2007; Oliveira et al. 2021; Aisala et al. 2023); occasionally, this hormone was replaced by IBA (Teixeira et al. 2004; Etienne 2005; Pádua et al. 2014) or NAA (Staritsky 1970). In support of the limited application of alternative auxins in coffee tissue culture, our data clearly indicated that β-NOA, IAA, IBA and NAA cannot replace 2,4-D for sufficient callus development. Poor results were somewhat expected for IAA and IBA, two naturally occurring PGRs that are known to progressively lose their efficacy with time in both agarized and liquid media (Nissen and Sutter 1990). Since their higher stability, the use of synthetic PGRs has increased in tissue culture techniques. Poor callus masses were also achieved with NAA, likely due to the necessity of higher hormone concentrations, as reported in previous studies (Al-Hamidi et al. 2023).
Callus proliferation was highest when chemically stable, chlorinated auxins were used; Di and 2,4,5-T exceeded the performance of 2,4-D, but callus quality and its long-term viability were remarkably different in the media enriched with the two hormones. When Di was used, callus consistency was mixed, especially in cv. Castillo; furthermore, in subsequent subcultures, callus masses turned completely brown, indicating a cell sensitivity to prolonged Di exposure. These drawbacks were absent when adding 2,4,5-T, as this hormone consistently promoted friable callus proliferation with no apparent loss of efficacy over time. In respect to 2,4-D, 2,4,5-T has been used to a much lesser extent in plant tissue culture, however, its stronger activity has been recognized in callus growth of some species (Suprasanna et al. 1989; Rani et al. 2003; Ahmad et al. 2010). Notably, its effectiveness decreased at high doses (Suprasanna et al. 1989), suggesting a narrow effective concentration range (Conger et al. 1978).
The combination of 1 mg/L of both KT and 2,4,5-T was successful not only in callus induction and proliferation, but also in cell suspension establishment. Indeed, the same medium supported the development of a fine and homogeneous suspension of whitish cells, in amounts higher than those achieved with other hormonal treatments; in particular, the application of equal concentrations of 2,4-D significantly reduced cells growth (Fig. 4). In a former work, 2,4,5-T was also selected for the initiation and maintenance of Solanum elaeagnifolium cell suspensions but, contrary to our results, 2,4-D promoted the highest cell growth (Alvarez et al. 1993). Moreover, Suprasanna et al. (1989) reported a better performance of 2,4-D in obtaining a fine and homogeneous cell suspension in respect to the 2,4-D analogues 2,4,5-P and 2,4,5-T; with the latter auxins only large aggregates were produced, evidencing the fundamental role of chemical structure in the cell separation process. According to our microscopic inspections, no relevant differences in aggregates dimensions were detected between 2,4-D and 2,4,5-T. In either case, the suspension consisted of a mixture of single cells, cell couples, clumps made up of 3–4 cells, and aggregates smaller than 500 μm.
The selected hormonal treatment allowed the establishment of viable CSCs with typical sigmoidal growth curves and well-defined lag, exponential and stationary phases (Fig. 6). Growth rates shown by the two cvs. were clearly different; in particular, Castillo cell suspensions remained for a longer time in the lag phase, reaching the mid-exponential phase 8 days later than Red Bourbon. On the other hand, Red Bourbon suspension cultures entered the stationary phase when the cell biomass was comparably lower. The definition of cell growth parameters is crucial in planning subculture intervals. It is widely recognized that harvesting cells before entering the stationary phase ensures a more active cell-fraction and a shorter recovery time, maintaining a cell suspension with a constant growth kinetic (Freshney 2005). By examining the growth curves, a subculture time should be set within the 16th and 27th day for Red Bourbon and Castillo, respectively. Nevertheless, given the fact that the signaling pathway for coming in and out the G0 and G1 phase can be diversely modulated according to genotype, the above-mentioned subculture intervals could be approximate.
In addition to a considerable biomass production, coffee cell cultures showed a phytochemical profile enriched with the most-studied compounds of coffee, namely alkaloids (i.e., trigonelline and theobromine) and phenolics (i.e., chlorogenic acids) (Table 2). Despite caffeine has been detected, its quantification was not possible because it was present in quantity lower than LOQ (< 0.001 mg/g). This is consistent with previous studies performed on coffee CSCs, which detected a higher concentration of this alkaloid released in the culture medium compared to that within the cells (Sartor and Mazzafera 2000; Pech-Kú et al. 2018). Concerning CGAs, approximately > 90% was represented by 5-CQA (Table S5), which is also the most abundant isomer found in coffee green bean or leaf (Monteiro et al. 2020; Rojas-González et al. 2022). When exposed to photoperiod, as expected, cells considerably increased their CGAs content, especially in CSCs of cv. Red Bourbon (Table 2). In addition to 5-CQA, 3,5-diCQA was the second most-induced isomer (Table S5), reflecting the CGAs composition of the leaf (Monteiro et al. 2020). For this class of molecules, our results are in agreement with the most recent phytochemical characterization performed by Khushvakov et al. (2024) on cell-cultured coffee. Indeed, in the dark-cultured cells, they reported a relatively low concentration of 5-CQA, counting for the 0.07% of DW. However, as supported by the widely-known effect of light in stimulating the synthesis of phenolic compounds, the application of suitable stressors through elicitation strategies could help in the optimization of relevant SMs production.
Conclusions
Despite the great interest for tissue culture from Coffea spp., the long-term maintenance of undifferentiated cell mass has been poorly debated so far. The results described therein identify a culture medium suitable for the development of cell suspensions from the most preliminary step of the process, namely the callus induction. Since their strong control over cell differentiation, growth regulators were deeply investigated in order to find the best culture conditions for the induction of a friable callus, recognized as the most suitable cell suspension establishment easy to crumble into a fine and homogeneous texture. Furthermore, the basic composition of the culture medium, requiring only nutrients, an energy source, and growth regulators, highlights what is strictly necessary for a satisfactory biomass production. Finally, the presence of phytochemicals commonly produced in Coffea spp. provides evidence for not only commercial but also therapeutic applications, which are currently unexploited through such tissue culture techniques. Optimization steps concerning the high-scale production of such valuable phytochemicals require further investigation and might not be solely dependent from growth regulators, but also rely on specific culture conditions (e.g., light/dark), and could be enhanced by elicitation or metabolic engineering strategies.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- SMs:
-
Secondary metabolites
- CSCs:
-
Cell suspension cultures
- PGRs:
-
Plant Growth Regulators
- 2,4,5-T:
-
2,4,5-Trichlorophenoxyacetic acid
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- 2-iP:
-
2-Isopentenyladenine
- 4-CPA:
-
p-Chlorophenoxyacetic acid
- 3-CQA:
-
3-Caffeoylquinic acid
- 4-CQA:
-
4-Caffeoylquinic acid
- 5-CQA:
-
5-Caffeoylquinic acid
- 3,4-diCQA:
-
3,4-Caffeoylquinic acid
- 3,5-diCQA:
-
3,5-Caffeoylquinic acid
- 4,5-diCQA:
-
4,5-Caffeoylquinic acid
- βNOA:
-
β-Naphthoxyacetic acid
- AHS:
-
Adenine Hemisulfate
- BAP:
-
6-Benzylaminopurine
- BPA:
-
N-Benzyl-9-(tetrahydropyranyl)-adenine
- Di:
-
Dicamba
- DHZ:
-
Dihydrozeatin
- DPHU:
-
1,3-Diphenylurea
- IAA:
-
Indole-3-acetic acid
- IBA:
-
Indol-3-butyric acid
- KT:
-
Kinetin
- NAA:
-
α-Naphthaleneacetic acid
- TDZ:
-
Thidiazuron
- ZT:
-
Zeatin
References
Ahmad N, Faisal M, Anis M, Aref IM (2010) In vitro callus induction and plant regeneration from leaf explants of Ruta graveolens L. South Afr J Bot 76:597–600. https://doi.org/10.1016/j.sajb.2010.03.008
Aisala H, Kärkkäinen E, Jokinen I et al (2023) Proof of concept for cell culture-based coffee. J Agric Food Chem 71:18478–18488. https://doi.org/10.1021/acs.jafc.3c04503
Al-Hamidi AOA, Al-Hadedy SHA, Bashi AZAK (2023) Effect of 2,4-D and NAA in callus induction and differentiation from different explants of Moringa oleifera Lam. IOP Conf Ser: Earth Environ Sci 1252:012091. https://doi.org/10.1088/1755-1315/1252/1/012091
Alvarez MA, Nigra HM, Giulietti AM (1993) Solasodine production by Solanum Eleagnifolium Cav. In vitro cultures: influence of plant growth regulators, age and inoculum size. Large-scale production. Nat Prod Lett 3:9–19. https://doi.org/10.1080/10575639308043832
Baumann TW, Röhrig L (1989) Formation and intracellular accumulation of caffeine and chlorogenic acid in suspension cultures of Coffea arabica. Phytochemistry 28:2667–2669. https://doi.org/10.1016/S0031-9422(00)98064-6
Bottcher A, Nobile PM, Martins PF et al (2012) Antioxidative responses of cell suspension cultures of two Coffea arabica varieties to low aluminum levels at pH 5.8. Hoehnea 39:01–10. https://doi.org/10.1590/S2236-89062012000100001
Carimi F, Zottini M, Formentin E et al (2003) Cytokinins: new apoptotic inducers in plants. Planta 216:413–421. https://doi.org/10.1007/s00425-002-0862-x
Carimi F, Terzi M, De Michele R et al (2004) High levels of the cytokinin BAP induce PCD by accelerating senescence. Plant Sci 166:963–969. https://doi.org/10.1016/j.plantsci.2003.12.016
Carra A, De Pasquale F, Ricci A, Carimi F (2006) Diphenylurea derivatives induce somatic embryogenesis in Citrus. Plant Cell Tiss Organ Cult 87:41–48. https://doi.org/10.1007/s11240-006-9132-0
Conger BV, Carabia JV, Lowe KW (1978) Comparison of 2,4-D and 2,4,5-T on callus induction and growth in three Gramineae species. Environ Exp Bot 18:163–168. https://doi.org/10.1016/0098-8472(78)90034-5
Domínguez F, Chávez M, Garduño-Ramírez ML et al (2009) Production of honokiol and magnolol in suspension cultures of Magnolia dealbata Zucc. Nat Prod Commun 4:939–943
Efferth T (2019) Biotechnology applications of plant callus cultures. Engineering 5:50–59. https://doi.org/10.1016/j.eng.2018.11.006
Etienne H (2005) Somatic embryogenesis protocol: coffee (Coffea arabica L. and C. canephora P.). In: Protocol for somatic embryogenesis in woody plants. https://agritrop.cirad.fr/528249/. Accessed 28 Nov 2023
Freshney RI (2005) Basic principles of Cell Culture. Culture of cells for tissue engineering. Wiley, Ltd, pp 1–22
Frischknecht PM, Baumann TW (1980) The pattern of purine alkaloid formation in suspension cultures of Coffea arabica. Planta Med 40:245–249. https://doi.org/10.1055/s-2008-1074965
Frischknecht PM, Baumann TW (1985) Stress induced formation of purine alkaloids in plant tissue culture of Coffea arabica. Phytochemistry 24:2255–2257. https://doi.org/10.1016/S0031-9422(00)83020-4
Furuya T, Koge K, Orihara Y (1990) Long term culture and caffeine production of immobilized coffee (Coffea arabica) L. cells in polyurethane foam. Plant Cell Rep 9:125–128. https://doi.org/10.1007/BF00232086
Gomes-Junior RA, Moldes CA, Delite FS et al (2006) Antioxidant metabolism of coffee cell suspension cultures in response to cadmium. Chemosphere 65:1330–1337. https://doi.org/10.1016/j.chemosphere.2006.04.056
Gomes-Junior RA, Gratão PL, Gaziola SA et al (2007) Selenium-induced oxidative stress in coffee cell suspension cultures. Funct Plant Biol 34:449–456. https://doi.org/10.1071/FP07010
Herman EB, Haas GJ (1975) Clonal propagation of Coffea arabica L. from callus culture. HortScience 10:588–589. https://doi.org/10.21273/HORTSCI.10.6.588
Ikeuchi M, Sugimoto K, Iwase A (2013) Plant callus: mechanisms of induction and repression. Plant Cell 25:3159–3173. https://doi.org/10.1105/tpc.113.116053
John PCL, Zhang K, Dong C et al (1993) p34cdc2 related proteins in control of cell cycle progression, the switch between division and differentiation in tissue development, and stimulation of division by auxin and cytokinin. Funct Plant Biol 20:503–526. https://doi.org/10.1071/pp9930503
Kapoor S, Sharma A, Bhardwaj P et al (2019) Enhanced production of phenolic compounds in compact callus aggregate suspension cultures of Rhodiola Imbricata Edgew. Appl Biochem Biotechnol 187:817–837. https://doi.org/10.1007/s12010-018-2851-y
Kaur P, Kothari SL (2004) Vitro culture of Kodo millet: influence of 2,4-D and Picloram in combination with kinetin on callus initiation and regeneration. Planr Cell Tissue Organ Cult 77:73–79. https://doi.org/10.1023/B:TICU.0000016505.20448.44. in
Khan KM, Misra P, Sharma T et al (2014) Effect of adenine sulphate on in vitro mass propagation of Stevia rebaudiana Bertoni. J Med Plants Res 8:543–549. https://doi.org/10.5897/JMPR2013.5217
Khushvakov J, Opitz S, Plüss N et al (2024) Analytical platform to determine similarities and dissimilarities between cell-cultured coffee and farm-grown coffee. ACS Food Sci Technol. https://doi.org/10.1021/acsfoodscitech.4c00238
Kurata H, Seki M, Furusaki S, Furuya T (1991) Influence of light irradiation rates and irradiation modes on caffeine production and cell growth in suspension culture of Coffea arabica cells. J Chem Eng Jpn 24:783–788. https://doi.org/10.1252/jcej.24.783
Kurata H, Achioku T, Furusaki S (1998) The light/dark cycle operation with an hour-scale period enhances caffeine production by Coffea arabica cells. Enzym Microb Technol 23:518–523. https://doi.org/10.1016/S0141-0229(98)00081-7
Laureys F, Dewitte W, Witters E et al (1998) Zeatin is indispensable for the G2-M transition in tobacco BY-2 cells. FEBS Lett 426:29–32. https://doi.org/10.1016/s0014-5793(98)00297-x
Linden JC, Phisalaphong M (2000) Oligosaccharides potentiate methyl jasmonate-induced production of paclitaxel in Taxus canadensis. Plant Sci 158:41–51. https://doi.org/10.1016/S0168-9452(00)00306-X
Mahood HE, Sarropoulou V, Tzatzani T-T (2022) Effect of explant type (leaf, stem) and 2,4-D concentration on callus induction: influence of elicitor type (biotic, abiotic), elicitor concentration and elicitation time on biomass growth rate and costunolide biosynthesis in gazania (Gazania rigens) cell suspension cultures. Bioresources Bioprocess 9:100. https://doi.org/10.1186/s40643-022-00588-2
Malyutina A, Tang J, Pessia A (2023) Drda: an R package for dose-response data analysis using logistic functions. J Stat Softw 106:1–26. https://doi.org/10.18637/jss.v106.i04
McCabe PF, Leaver CJ (2000) Programmed cell death in cell cultures. Plant Mol Biol 44:359–368. https://doi.org/10.1023/A:1026500810877
Mirjalili N, Linden JC (1996) Methyl jasmonate induced production of taxol in suspension cultures of Taxus cuspidata: ethylene interaction and induction models. Biotechnol Prog 12:110–118. https://doi.org/10.1021/bp9500831
Moeenfard M, Cortez A, Machado V et al (2016) Anti-angiogenic properties of cafestol and kahweol palmitate diterpene esters. J Cell Biochem 117:2748–2756. https://doi.org/10.1002/jcb.25573
Mok MC, Mok DW, Dixon SC et al (1982) Cytokinin structure-activity relationships and the metabolism of N-(delta-isopentenyl)adenosine-8-C in Phaseolus callus tissues. Plant Physiol 70:173–178. https://doi.org/10.1104/pp.70.1.173
Monteiro Â, Colomban S, Azinheira HG et al (2020) Dietary antioxidants in coffee leaves: impact of botanical origin and maturity on chlorogenic acids and xanthones. Antioxidants 9:6. https://doi.org/10.3390/antiox9010006
Munyendo LM, Njoroge DM, Owaga EE, Mugendi B (2021) Coffee phytochemicals and post-harvest handling—A complex and delicate balance. J Food Compos Anal 102:103995. https://doi.org/10.1016/j.jfca.2021.103995
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Nakashima Y, Kobayashi Y, Murao M et al (2023) Identification of a pluripotency-inducing small compound, PLU, that induces callus formation via heat shock protein 90-mediated activation of auxin signaling. Front Plant Sci 14. https://doi.org/10.3389/fpls.2023.1099587
Nissen SJ, Sutter EG (1990) Stability of IAA and IBA in nutrient medium to several tissue culture procedures. HortSci 25:800–802. https://doi.org/10.21273/HORTSCI.25.7.800
Ogita S, Uefuji H, Morimoto M, Sano H (2004) Application of RNAi to confirm theobromine as the major intermediate for caffeine biosynthesis in coffee plants with potential for construction of decaffeinated varieties. Plant Mol Biol 54:931–941. https://doi.org/10.1007/s11103-004-0393-x
Oliveira JP, de Sanglard M, Ferreira NA, Clarindo A WR (2021) Genomic methylated cytosine level during the dedifferentiation and cellular competence in Coffea arabica lines: insights about the different in vitro responses. Forests 12:1536. https://doi.org/10.3390/f12111536
Pádua MS, Paiva LV, Silva LC da, et al (2014) Morphological characteristics and cell viability of coffee plants calli. Cienc Rural 44:660–665. https://doi.org/10.1590/S0103-84782014000400014
Pawar KD, Yadav AV, Shouche YS, Thengane SR (2011) Influence of endophytic fungal elicitation on production of inophyllum in suspension cultures of Calophyllum inophyllum L. Plant Cell Tiss Organ Cult 106:345–352. https://doi.org/10.1007/s11240-011-9928-4
Pech-Kú R, Muñoz-Sánchez JA, Monforte-González M et al (2018) Relationship between aluminum stress and caffeine biosynthesis in suspension cells of Coffea arabica L. J Inorg Biochem 181:177–182. https://doi.org/10.1016/j.jinorgbio.2017.07.032
Praveen N, Murthy HN, Chung IM (2011) Improvement of growth and gymnemic acid production by altering the macro elements concentration and nitrogen source supply in cell suspension cultures of Gymnema sylvestre R. Br. Ind Crops Prod 33:282–286. https://doi.org/10.1016/j.indcrop.2010.12.015
Rani G, Virk GS, Nagpal A (2003) Callus induction and plantlet regeneration in Withania somnifera (L.) dunal. In Vitro Cellular & Developmental Biology Plant 39:468–474. https://doi.org/10.1079/IVP2003449
Rency AS, Pandian S, Ramesh M (2018) Influence of adenine sulphate on multiple shoot induction in Clitoria ternatea L. and analysis of phyto-compounds in in vitro grown plants. Biocatal Agric Biotechnol 16:181–191. https://doi.org/10.1016/j.bcab.2018.07.034
R Core Team (2021). R: a language and environment for statisticalcomputing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rojas-González A, Figueroa-Hernández CY, González-Rios O et al (2022) Coffee chlorogenic acids incorporation for bioactivity enhancement of foods: a review. Molecules 27:3400. https://doi.org/10.3390/molecules27113400
Sartor RM, Mazzafera P (2000) Caffeine formation by suspension cultures of Coffea dewevrei. Braz Arch Biol Technol 43:61–69. https://doi.org/10.1590/S1516-89132000000100009
Sidek N, Nulit R, Kong YC et al (2022) Callogenesis and somatic embryogenesis of Oryza sativa L. (Cv. MARDI Siraj 297) under the influence of 2, 4-dichlorophenoxyacetic acid and kinetin. AIMSAGRI 7:536–552. https://doi.org/10.3934/agrfood.2022033
Silva AS, Luz JMQ, Rodrigues TM et al (2011) Callus induction and embryo regeneration in Coffea arabica L. anthers by silver nitrate and ethylene. Rev Ciênc Agron 42:921–929. https://doi.org/10.1590/S1806-66902011000400014
Skoog F, Miller CO (1957) Chemical regulation of growth and organ formation in plant tissues cultured in vitro. Symp Soc Exp Biol 11:118–130
Söndahl MR, Sharp WR (1977) High frequency induction of somatic embryos in cultured leaf expiants of Coffea arabica L. Z für Pflanzenphysiologie 81:395–408. https://doi.org/10.1016/S0044-328X(77)80175-X
Souza JMM, Berkov S, Santos AS (2014) Improvement of friable callus production of Boerhaavia paniculata Rich and the investigation of its lipid profile by GC/MS. Acad Bras Ciênc 86:1015–1027. https://doi.org/10.1590/0001-3765201420130098
Staritsky G (1970) Embryoid formation in callus tissues of coffee. Acta Bot Neerlandica 19:509–514. https://doi.org/10.1111/j.1438-8677.1970.tb00677.x
Suliansyah I, Yusniwati, Resigia E et al (2021) Multiplication of Arabica Solok Rajo coffee through bio cellular technology. IOP Conf Ser: Earth Environ Sci 741:012053. https://doi.org/10.1088/1755-1315/741/1/012053
Suprasanna P, Rao KV, Reddy GM (1989) Genetically controlled anthocyanin synthesis in callus cultures of Zea mays L. Proceedings: Plant Sciences 99:293–295. https://doi.org/10.1007/BF03053605
Teixeira MC, Telo JP, Duarte NF, Sá-Correia I (2004) The herbicide 2,4-dichlorophenoxyacetic acid induces the generation of free-radicals and associated oxidative stress responses in yeast. Biochem Biophys Res Commun 324:1101–1107. https://doi.org/10.1016/j.bbrc.2004.09.158
Thacker X, Thomas K, Fuller M et al (2018) Determination of optimal hormone and mineral salts levels in tissue culture media for callus induction and growth of industrial hemp (Cannabis sativa L). AS 09:1250–1268. https://doi.org/10.4236/as.2018.910088
Townsley PM (1974) Production of coffee from plant cell suspension cultures. Can Inst Food Sci Technol J 7:79–81. https://doi.org/10.1016/S0315-5463(74)73854-8
Valente P, Tao W, Verbelen J-P (1998) Auxins and cytokinins control DNA endoreduplication and deduplication in single cells of tobacco. Plant Sci 134:207–215. https://doi.org/10.1016/S0168-9452(98)00062-4
van Staden J, Zazimalova E, George EF (2008) Plant growth regulators II: cytokinins, their analogues and antagonists. Plant Propag Tissue Cult 1: Backgr 205–226
Vescovi M, Riefler M, Gessuti M et al (2012) Programmed cell death induced by high levels of cytokinin in Arabidopsis cultured cells is mediated by the cytokinin receptor CRE1/AHK4. J Exp Bot 63:2825–2832. https://doi.org/10.1093/jxb/ers008
Wintgens JN (ed) (2012) Coffee - growing, processing, sustainable production: a guidebook for growers, processors, traders and researchers, 2nd, Revised edition edn. Wiley-VCH, Weinheim, Germany
World Health Organization (2023) The selection and use of essential medicines 2023: web annex A. World Health Organization model list of essential medicines: 23rd list (2023)‎
Yang W, Cortijo S, Korsbo N et al (2021) Molecular mechanism of cytokinin-activated cell division in Arabidopsis. Science 371:1350–1355. https://doi.org/10.1126/science.abe2305
Zhang K, Diederich L, John PCL (2005) The cytokinin requirement for cell division in cultured Nicotiana plumbaginifolia cells can be satisfied by yeast Cdc25 protein tyrosine phosphatase: implications for mechanisms of cytokinin response and plant development. Plant Physiol 137:308–316. https://doi.org/10.1104/pp.104.051938
Zottini M, Barizza E, Bastianelli F et al (2006) Growth and senescence of Medicago truncatula cultured cells are associated with characteristic mitochondrial morphology. New Phytol 172:239–247. https://doi.org/10.1111/j.1469-8137.2006.01830.x
Acknowledgements
We would like to thank dr. Claudio Ferfuia for his help in statistical analyses and dr. Carla Pappalardo for her technical assistance.
Funding
Open access funding provided by Università degli Studi di Udine within the CRUI-CARE Agreement. Azzurra Di Bonaventura fellowship has been granted by the financial support from the Italian Ministry of Education “Programma Operativo Nazionale Ricerca e Innovazione 2014–2020” (CCI2014IT16M2OP005, risorse FSE REACT-EU, Azione IV.4 “Dottorati e contratti di ricerca su tematiche dell’innovazione” e Azione IV.5 “Dottorati su tematiche Green”), grant n. DOT13QKUB3-3. We also acknowledge illycaffé S.p.A. for its financial support.
Open access funding provided by Università degli Studi di Udine within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
Azzurra Di Bonaventura, Stefano Marchetti and Elisa Petrussa performed the experiments. Azzurra Di Bonaventura and Enrico Braidot analysed the data, prepared tables and figures. Azzurra Di Bonaventura and Silvia Colomban performed SMs chromatographic analysis. Azzurra Di Bonaventura, Stefano Marchetti and Marco Zancani designed and monitored the experiments. Luciano Navarini and Marco Zancani provided the plant material. All authors revised the draft of the manuscript. All authors read the manuscript and approved it.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Communicated by Francisco de Assis Alves Mourão Filho.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Di Bonaventura, A., Marchetti, S., Petrussa, E. et al. A protocol for the development and maintenance of Coffea arabica (L.) cell suspension cultures. Plant Cell Tiss Organ Cult 158, 48 (2024). https://doi.org/10.1007/s11240-024-02848-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11240-024-02848-9