Abstract
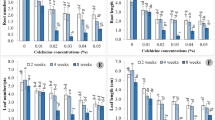
The hazelnut industry requires further development given the gap between production and demand. However, polyploid plantlet induction has not been reported in hazelnut varieties. As such, our aim in this study was to enrich the hazelnut germplasm resources. We used the shoot tips of in vitro propagated plantlets of Corylus heterophylla × Corylus avellana '84–226' as the material for inducing polyploidy. We supplemented the culture medium with 0, 100, 200, and 300 mg/L of colchicine, and we treated shoot tips for 20, 25, or 30 days. We found that the induction rate of tetraploids was the highest in treatments with 300 mg/L for 25 days; it was substantially higher by up to 20% than in the other treatments. In diploid and tetraploid tissue-cultured seedlings, tetraploid plantlets were much shorter, thicker, and stronger, as well as greener, than the diploid plantlets. The acquisition of polyploid hazelnuts provides an important prerequisite for hazelnut breeding.
Key message
We successful induced tetraploids of Corylus heterophylla × Corylus avellana '84–226', achieving a breakthrough as polyploid hazelnut has not previously appeared in nature, thereby enriching the hazelnut germplasm resources.




Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in the article.
References
Aleza P, Juárez J, Ollitrault P, Navarro L (2009) Production of tetraploid plants of non-apomictic citrus genotypes. Plant Cell Rep 28:1837–1846. https://doi.org/10.1007/s00299-009-0783-2
Allario T, Brumos J, Colmenero-Flores JM, Iglesias DJ, Pina JA, Navarro L, Talon M, Ollitrault P, Morillon R (2013) Tetraploid Rangpur lime rootstock increases drought tolerance via enhanced constitutive root abscisic acid production. Plant Cell Environ 36:856–868. https://doi.org/10.1111/pce.12021
Bhattarai K, Kareem A, Deng ZN (2021) In vivo induction and characterization of polyploids in gerbera daisy. Sci Hort 282:110054. https://doi.org/10.1016/j.scienta.2021.110054
Blakeslee AF, Avery AG (1937) Methods of inducing doubling of chromosomes in plants: by treatment with colchicine. J Hered 28:393–411. https://doi.org/10.1093/oxfordjournals.jhered.a104294
Blasco M, Badenes ML, Naval MM (2015) Colchicine-induced polyploidy in loquat (Eriobotrya japonica (Thunb.) Lindl.). Plant Cell Tissue Organ Cult 120:453–461. https://doi.org/10.1007/s11240-014-0612-3
Chakraborti SP, Vijayan K, Roy BN, Qadri SMH (1998) In vitro induction of tetraploidy in mulberry (Morus alba L.). Plant Cell Rep 17:799–803. https://doi.org/10.1007/s002990050486
Chen JT, Coate JE, Meru G (2020) Editorial: artificial polyploidy in plants. Front Plant Sci 11:621849. https://doi.org/10.3389/fpls.2020.621849
Chen RR, Feng ZY, Zhang XH, Song ZJ, Cai DT (2021) A New Way of Rice Breeding: Polyploid Rice Breeding. Plants 10:422. https://doi.org/10.3390/plants10030422
Cui YH, Hou L, Li X, Huang FY, Pang XM, Li YY (2017) In vitro induction of tetraploid Ziziphus jujuba Mill. var. spinosa plants from leaf explants. Plant Cell Tissue Organ Cult 131:175–182. https://doi.org/10.1007/s11240-017-1274-8
Eng WH, Ho WS (2019) Polyploidization using colchicine in horticultural plants: A review. Sci Hort 246:604–617. https://doi.org/10.1016/j. scien ta. 2018. 11. 010
Gallone A, Hunter A, Douglas GC (2014) Polyploid induction in vitro using colchicine and oryzalin on Hebe ‘Oratia Beauty’: Production and characterization of the vegetative traits. Sci Hort 179:59–66. https://doi.org/10.1016/j.scienta.2014.09.014
Gómez P, Germanà MA (2021) Production of haploid and doubled haploid lines in nut crops: Persian Walnut, Almond, and Hazelnut. In: Segui-Simarro JM (ed) Doubled Haploid Technology. Methods in Molecular Biology, vol 2289. Humana, New York, NY
Grouh MSH, Vahdati K, Lotfi M, Hassani D, Pirvali Biranvand N (2011) Production of haploids in Persian walnut through parthenogenesis induced by gamma-irradiated pollen. J Am Soc Hortic Sci 136(3):198–204
Guo YY, Xing SY, Ma YM, Tamg HX, Han KJ (2009) Analysis of karyotype on fifteen hazelnut germplasms. Acta Horticulturae Sinica 36(1):27–32.
Hu YL, Sun D, Hu HG, Zuo XD, Xia T, Xie JH (2021) A comparative study on morphological and fruit quality traits of diploid and polyploid carambola (Averrhoa carambola L.) genotypes. Sci Hort 277:109843. https://doi.org/10.1016/j.scienta.2020.109843
Huo HL, Ma QH, Li JJ, Zhao TT, Wang GX (2016) Study on the Distribution of Corylus L. in China and the Climatic Evaluation of the Suitable Areas. J Plant Genetic Resour 17(5):801–808. https://doi.org/10.13430/j.cnki.jpgr.2016.05.002
Huy NP, Tam DTT, Luan VQ, Tung HT, Hien VT, Ngan HTM, Duy PN, Nhut DT (2019) In vitro polyploid induction of Paphiopedilum villosum using colchicine. Sci Hort 252:283–290. https://doi.org/10.1016/j.scienta.2019.03.063
Jaretzky R (1930) Zur Zytologie der Fagales. Planta 10:120–137. https://doi.org/10.1007/BF01911539
Jia BY (2020) Cytological ploidy identification and its important characters transcriptome analysis of Acer buergerianum Miq. Dissertation, Shandong Agricultural University
Kadota M, Niimi Y (2002) In vitro induction of tetraploid plants from a diploid Japanese pear cultivar (Pyrus pyrifolia N. cv. Hosui). Plant Cell Rep 21:282–286. https://doi.org/10.1007/s00299-002-0509-1
Kang XY, Wei HL (2022) Breeding polyploid Populus: progress and perspective. Forestry Res 2:4. https://doi.org/10.48130/FR-2022-0004
Lei JJ, Wu LP, Dai HP, Hu WY, Ge HB (1999) Studies on Chromosome Doubling with Shoot-tip Culture in Strawberry. Acta horticulturea Sinica 26(1):13–18.
Li XS, Zhao K, Li L (2015) Tissue Culture of Vaccinium L.cv.“Legacy”and Mutagenic Effects of Colchicine. J Southwest Univ (Natural Sci Edition) 37(5):51–57. https://doi.org/10.13718/j.cnki.xdzk.2015.05.008
Loureiro J, Rodriguez E, Dolezel J, Santos C (2007) Two new nuclear isolation buffers for plant DNA flow cytometry: A Test with 37 Species. Ann Botany 100(4):875–888. https://doi.org/10.1093/aob/mcm152
Miguel TP, Leonhardt KW (2011) In vitro polyploid induction of orchids using oryzalin. Sci Hort 130:314–319. https://doi.org/10.1016/j.scienta.2011.07.002
Nas MN, Read PE (2004) A hypothesis for the development of a defined tissue culture medium of higher plants and micropropagation of hazelnuts. Sci Hort 101:189–200. https://doi.org/10.1016/j.scienta.2003.10.004
Nasirvand S, Asghari Zakaria R, Zare N, Esmaeilpoor B (2018) Polyploidy induction in parsley (Petroselinum crispum L.) by colchicine treatment. Cytologia 83(4):393–396. https://doi.org/10.1508/cytologia.83.393
Notsuka K, Tsuru T, Shiraishi M (2000) Induced Polyploid Grapes via in vitro Chromosome Doubling. J Japan Soc Hort Sci 69(5):543–551. https://doi.org/10.2503/jjshs.69.543
Podwyszyńska M, Sowik I, Machlańska A, Kruczyńska D, Dyki B (2017) In vitro tetraploid induction of Malus × domestica Borkh. using leaf or shoot explants. Sci Hort 226:379–388. https://doi.org/10.1016/j.scienta.2017.08.042
Portela de Carvalho JFR, de Portela de Carvalho CR, Otoni WC (2005) In vitro induction of polyploidy in annatto (Bixa orellana). Plant Cell Tissue Organ Cult 80:69–75. https://doi.org/10.1007/s11240-004-8833-5
Premjet D, Obeng AK, Kongbangkerd A, Premjet S (2019) Intergeneric Hybrid from Jatropha curcas L. and Ricinus communis L.: Characterization and Polyploid Induction. Biology 8(2):50. https://doi.org/10.3390/biology8020050
Qaderi A, Omidi M, Etminan A, Oladzad A, Ebrahimi C, Dehghani Mashkani MR, Mehrafarin A (2012) Hazel (Corylus avellana L.) as a new source of taxol and taxanes. J Med Plants 11:66–77.
Rovira M (2021) Advances in Hazelnut (Corylus avellana L.) Rootstocks Worldwide. Horticulturae 7:267. https://doi.org/10.3390/horticulturae7090267
Rovira M, Hermoso JF, Rufat J, Cristofori V, Silvestri C, Romero A (2022) Agronomical and Physiological Behavior of Spanish Hazelnut Selection “Negret-N9” Grafted on Non-suckering Rootstocks. Front Plant Sci 12:813902. https://doi.org/10.3389/fpls.2021.813902
Russell G (2004) Stomatal guard cell measurements using leaf prints. CSA J 4:137–139.
Salma U, Kundu S, Mandal N (2017) Artificial polyploidy in medicinal plants: advancement in the last two decades and impending prospects. J Crop Sci Biotechnol 20(1):9–19. https://doi.org/10.1007/s12892-016-0080-1
Sattler MC, Carvalho CR, Clarindo WR (2015) The polyploidy and its key role in plant breeding. Planta 243:281–296. https://doi.org/10.1007/s00425-015-2450-x
Thomasset M, Fernandez-Manjarrés JF, Douglas GC, Frascaria-Lacoste N, Raquin C, Hodkinson TR (2011) Molecular and Morphological Characterization of Reciprocal F1 Hybrid Ash (Fraxinus excelsior × Fraxinus angustifolia, Oleaceae) and Parental Species Reveals Asymmetric Character Inheritance. Int J Plant Sci 172:423–433. https://doi.org/10.1098/658169
Wang LH, Luo Z, Wang LL, Deng WP, Wei H, Liu P (2019) Morphological, cytological and nutritional changes of autotetraploid compared to its diploid counterpart in Chinese jujube (Ziziphus jujuba Mill.). Sci Hort 249:263–270. https://doi.org/10.1016/j.scienta.2019.01.063
Wang N, Liu M, Dai L, Qin Z (2005) In vitro tetraploid induction of Ziziphus jujuba ‘Dongzao’ and Z. acidojujuba (Z. spinosa Hu) with colchicine. Acta Horticulturae Sinica 32(6):1008–1012.
Wang QF, Mu XP, Gao YG, Zhang JC, Du JJ (2020) Successful induction and the systematic characterization of tetraploids in cerasus humilis for subsequent breeding. Sci Hort 265:109216. https://doi.org/10.1016/j.scienta.2020.109216
Wei TL, Wang Y, Liu JH (2020) Comparative transcriptome analysis reveals synergistic and disparate defense pathways in the leaves and roots of trifoliate orange (Poncirus trifoliata) autotetraploids with enhanced salt tolerance. Hortic Res 7:88. https://doi.org/10.1038/s41438-020-0311-7
Wu J, Cheng XT, Kong B, Zhou Q, SangYR, Zhang PD (2022) In vitro octaploid induction of Populus hopeiensis with colchicine. BMC Plant Biol 22:176. https://doi.org/10.1186/s12870-022-03571-3
Wu JH, Ferguson AR, Murray BG (2011) Manipulation of ploidy for kiwifruit breeding: in vitro chromosome doubling in diploid Actinidia chinensis Planch. Plant Cell Tissue Organ Cult 106:503–511. https://doi.org/10.1007/s11240-011-9949-z
Wu JH, Mooney P (2002) Autotetraploid tangor plant regeneration from in vitro Citrus somatic embryogenic callus treated with colchicine. Plant Cell Tissue Organ Cult 70:99–104. https://doi.org/10.1023/A:1016029829649
Xi YK, Dong X, Yang M, Meng QH, Huang HY (2021) In Vitro Polyploid Induction and Establishment of a Clone for Cyclocodon lancifolius (Roxb.) Kurz. Cytologia 86(4):367–374. https://doi.org/10.1508/cytologia.86.367
Xie X, Aguero CB, Wang Y, Walker MA (2015) In vitro induction of tetraploids in Vitis×Muscadinia hybrids. Plant Cell Tissue Organ Cult 122:675–683. https://doi.org/10.1007/s11240-015-0801-8
Zhang XQ, Gao JY (2021) Colchicine–induced tetraploidy in Dendrobium cariniferum and its effect on plantlet morphology, anatomy and genome size. Plant Cell Tissue Organ Cult 144:409–420. https://doi.org/10.1007/s11240-020-01966-4
Zhou HW, Zeng WD, Yan HB (2017) In vitro induction of tetraploids in cassava variety ‘Xinxuan 048’ using colchicine. Plant Cell Tissue Organ Cult 28:723–729. https://doi.org/10.1007/s11240-016-1141-z
Acknowledgements
This study was supported by the National Horticultural Germplasm Shenyang Hawthorn Branch Library Operation Service (NHGRC2020-NH08) and Collection, Identification, Numbering, Breeding, and Storage of Hawthorn Germplasm Resources (19200357).
Author information
Authors and Affiliations
Contributions
Ke Zheng carried out the experiment, collected and analysed data, and wrote the manuscript. Wenxuan Dong was involved in planning and supervising the study. Jian Wang performed part of the experiment. Xiao Zhang and Yali Hou assisted in the design of the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Maria Antonietta Germanà.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, K., Wang, J., Zhang, X. et al. Hazelnut germplasm innovation: induction of tetraploid hybrid hazelnut by colchicine. Plant Cell Tiss Organ Cult 152, 21–29 (2023). https://doi.org/10.1007/s11240-022-02385-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-022-02385-3