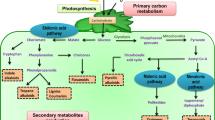
Abstract
This report focuses on the crucial role of lipids and starch metabolism in the growth and ultrastructure of the cell wall (CW) in rice calli. Non-habituated calli (NH-calli) were isolated from a rice embryo-derived callus culture supplemented with 2,4-D (2,4-Dichlorophenoxyacetic acid); however, the habituated calli (H-calli) were isolated from NH ones and cultured subsequently on exogenous hormone-free medium. The phenotypes of the H- and NH-calli differed significantly between each other as reported in our recently published work. The H-calli CW was thinner, the endoplasmic reticulum (ER), mitochondria (MT) and starch grains (SGs) were less in the H-calli. The intracellular free fatty acids (FAs) and starch content were much less in the H- if compared to NH-calli. By microarray analysis, the differentially expressed genes (DEGs) related to primary and secondary metabolism, lipid and starch metabolism were down-regulated in H- vs NH-calli. The response at the transcriptomic level involved in these changes, as well as the corresponding variations in cytology and FAs metabolism were also accurately elucidated herein. The study reveals that the decreased levels of lipids and starch in the H-calli are mainly attributable to their (H-calli) elevated consumption, in the absence of exogenous hormonal supply to ensure the demand in energy of the calli to survive and maintain an optimum growth and development with the available resource of energy (starch and lipids). This report displays as well the slowdown of the H-calli metabolism, in the absence of exogenous growth regulators supply, if compared to that of the NH-ones.
Key message
Lipids and starch decreased in the H-calli is mainly attributable to their elevated consumption to survive and maintain an optimal growth and development with the available resource of energy.











Similar content being viewed by others
Availability of data and material
This study includes two supplemental tables.
Abbreviations
- ACBP:
-
acyl-CoA-binding protein
- ACS:
-
acyl-CoA synthetase
- CW:
-
cell wall
- Cryo-SEM:
-
Cryo-scanning electron microscopy
- DAG:
-
1,3-diacylglycerol
- DEGs:
-
differentially expressed genes
- DGAT:
-
diacylglycerol O-acyltransferase
- d:
-
day
- FAs:
-
fatty acids
- FAE:
-
fatty acid elongase
- FAR:
-
fatty acyl-CoA reductase
- FM:
-
Fluorescence microscopy
- KCS:
-
3-ketoacyl-CoA synthase
- LD:
-
lipid drop
- LM:
-
light microscopy
- LTPs:
-
lipid transfer proteins
- PM:
-
plasmalemma
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- TAG:
-
triacylglycerol
- TEM:
-
Transmission electron microscopy
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid.
References
Ballicora MA, Iglesias AA, Preiss J (2004) ADP-Glucose pyrophosphorylase: a regulatory enzyme for plant starch synthesis. Photosynth Res 79(1):1–24. https://doi.org/10.1023/B:PRES.0000011916.67519.58
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B 57(1):289–300 https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Bindea G, Mlecnik B, Hackl H, Charoentong P, Tosolini M, Kirilovsky A, Fridman WH, Pages F, Trajanoski Z, Galon J (2009) ClueGO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 25(8):1091–1093. https://doi.org/10.1093/bioinformatics/btp101
Bindea G, Galon J, Mlecnik B (2013) CluePedia Cytoscape plugin: pathway insights using integrated experimental and in silico data. Bioinformatics 29(5):661–663. https://doi.org/10.1093/bioinformatics/btt019
Browse John SC (1991) Glycerolipid metabolism: biochemistry and regulation. Plant Mol Biol 42(1):467–506
Brundrett MC, Kendrick B, Peterson CA (1991) Efficient lipid staining in plant material with sudan red 7B or fluorol [correction of fluoral] yellow 088 in polyethylene glycol-glycerol. Biotech Histochem 66(3):111–116. https://doi.org/10.3109/10520299109110562
Cai Y, Goodman JM, Pyc M, Mullen RT, Dyer JM, Chapman KD (2015) Arabidopsis SEIPIN proteins modulate triacylglycerol accumulation and influence lipid droplet proliferation. Plant Cell 27(9):2616–2636. https://doi.org/10.1105/tpc.15.00588
Chapman KD, Ohlrogge JB (2012) Compartmentation of triacylglycerol accumulation in plants. J Biol Chem 287(4):2288–2294. https://doi.org/10.1074/jbc.R111.290072
Chotipa Sakulsingharoj S-BC, Hwanga S-K, Edwardsc GE, Borkd J, Meyerd CR, Preissd J, Okitaa TW (2004) Engineering starch biosynthesis for increasing rice seed weight: the role of the cytoplasmic ADP-glucose pyrophosphorylase. Plant Sic 167:11
Chu CC, Wang CC, Sun CS, Hsu KC, Yen KC, Chu CY, FY B (1975) Establishment of an efficient medium for anther culture of rice through comparative experimentation on the nitrogen source. Sci China 13:659–688
Dal Santo S, Fasoli M, Cavallini E, Tornielli GB, Pezzotti M, Zenoni S (2011) PhEXPA1, a Petunia hybrida expansin, is involved in cell wall metabolism and in plant architecture specification. Plant Signal Behav 6(12):2031–2034. https://doi.org/10.4161/psb.6.12.18110
Domergue F, Vishwanath SJ, Joubes J, Ono J, Lee JA, Bourdon M, Alhattab R, Lowe C, Pascal S, Lessire R, Rowland O (2010) Three Arabidopsis fatty acyl-coenzyme A reductases, FAR1, FAR4, and FAR5, generate primary fatty alcohols associated with suberin deposition. Plant Physiol 153(4):1539–1554. https://doi.org/10.1104/pp.110.158238
Fiehn O, Kopka J, Dormann P, Altmann T, Trethewey RN, Willmitzer L (2000) Metabolite profiling for plant functional genomics. Nat Biotechnol 18(11):1157–1161. https://doi.org/10.1038/81137
Fujita N, Satoh R, Hayashi A, Kodama M, Itoh R, Aihara S, Nakamura Y (2011) Starch biosynthesis in rice endosperm requires the presence of either starch synthase I or IIIa. J Exp Bot 62(14):4819–4831. https://doi.org/10.1093/jxb/err125
Gaspar T, Kevers C, Penel C (1988) Biochemical characterization of normal and habituated sugarbeet calli. Relationship with anatomy, habituation and organogenesis. Potsdamer Forsch 57:21–30
Gautheret G (1942) Hétéro-auxine et cultures de tissues végétaux. Bull Soc Chim Biol 24:13–47
Goold H, Beisson F, Peltier G, Li-Beisson Y (2015) Microalgal lipid droplets: composition, diversity, biogenesis and functions. Plant Cell Rep 34(4):545–555. https://doi.org/10.1007/s00299-014-1711-7
Hayashi M, Toriyama K, Kondo M, Kato A, Mano S, De Bellis L, Hayashi-Ishimaru Y, Yamaguchi K, Hayashi H, Nishimura M (2000) Functional transformation of plant peroxisomes. Cell Biochem Biophys 32 Spring:295–304
Jako C, Kumar A, Wei Y, Zou J, Barton DL, Giblin EM, Covello PS, Taylor DC (2001) Seed-specific over-expression of an Arabidopsis cDNA encoding a diacylglycerol acyltransferase enhances seed oil content and seed weight. Plant Physiol 126(2):861–874. https://doi.org/10.1104/pp.126.2.861
Jin J, Chen H, Cai W (2015) Transcriptome analysis of Oryza sativa calli under microgravity. Microgravity Sci Technol 27(6):437–453. https://doi.org/10.1007/s12217-015-9432-2
Jin J, Essemine J, Duan J, Xie Q, Zhu J, Cai W (2020) Regeneration of active endogenous IAA in rice calli following acclimation to 2,4-D free medium. Plant Growth Regul. https://doi.org/10.1007/s10725-020-00679-0
Judy SJS, John B (2004) The acyl-CoA synthetase encoded by LACS2 is essential for normal cuticle development in Arabidopsis. Plant Cell 16(16):629–642
Khor VK, Shen WJ, Kraemer FB (2013) Lipid droplet metabolism. Curr Opin Clin Nutr Metab Care 16(6):632–637. https://doi.org/10.1097/MCO.0b013e3283651106
Kong X, Li C, Zhang F, Yu Q, Gao S, Zhang M, Tian H, Zhang J, Yuan X, Ding Z (2018) Ethylene promotes cadmium-induced root growth inhibition through EIN3 controlled XTH33 and LSU1 expression in Arabidopsis. Plant Cell Environ 41(10):2449–2462. https://doi.org/10.1111/pce.13361
Kunst L, Samuels L (2009) Plant cuticles shine: advances in wax biosynthesis and export. Curr Opin Plant Biol 12(6):721–727. https://doi.org/10.1016/j.pbi.2009.09.009
Lee SK, Hwang SK, Han M, Eom JS, Kang HG, Han Y, Choi SB, Cho MH, Bhoo SH, An G, Hahn TR, Okita TW, Jeon JS (2007) Identification of the ADP-glucose pyrophosphorylase isoforms essential for starch synthesis in the leaf and seed endosperm of rice (Oryza sativa L.). Plant Mol Biol 65(4):531–546. https://doi.org/10.1007/s11103-007-9153-z
Lee HG, Kim H, Suh MC, Kim HU, Seo PJ (2018) The MYB96 transcription factor regulates triacylglycerol accumulation by activating DGAT1 and PDAT1 expression in Arabidopsis seeds. Plant Cell Physiol 59(7):1432–1442. https://doi.org/10.1093/pcp/pcy073
Li N, Xu C, Li-Beisson Y, Philippar K (2016) Fatty acid and lipid transport in plant cells. Trends Plant Sci 21(2):145–158. https://doi.org/10.1016/j.tplants.2015.10.011
Li Z, Ma S, Song H, Yang Z, Zhao C, Taylor D, Zhang M (2020) A 3-ketoacyl-CoA synthase 11 (KCS11) homolog from Malania oleifera synthesizes nervonic acid in plants rich in 11Z-Eicosenoic acid. Tree Physiol. https://doi.org/10.1093/treephys/tpaa125
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lock YY, Snyder CL, Zhu W, Siloto RM, Weselake RJ, Shah S (2009) Antisense suppression of type 1 diacylglycerol acyltransferase adversely affects plant development in Brassica napus. Physiol Plant 137(1):61–71. https://doi.org/10.1111/j.1399-3054.2009.01258.x
Lokesh U, kiranmai MP, babu S, Koundal N, Sudhakar C (2013) Role of plant fatty acid elongase (3 keto acyl-CoA Synthase) gene in cuticular wax biosynthesis. J Agric Allied Sci 2:35–42
Moreau P, Bessoule JJ, Mongrand S, Testet E, Vincent P, Cassagne C (1998) Lipid trafficking in plant cells. Prog Lipid Res 37(6):371–391. https://doi.org/10.1016/s0163-7827(98)00016-2
Mutch DM, Temanni MR, Henegar C, Combes F, Pelloux V, Holst C, Sorensen TI, Astrup A, Martinez JA, Saris WH, Viguerie N, Langin D, Zucker JD, Clement K (2007) Adipose gene expression prior to weight loss can differentiate and weakly predict dietary responders. PLoS One 2(12):e1344. https://doi.org/10.1371/journal.pone.0001344
Nookaraju A, Pandey SK, Fujino T, Kim JY, Suh MC, Joshi CP (2014) Enhanced accumulation of fatty acids and triacylglycerols in transgenic tobacco stems for enhanced bioenergy production. Plant Cell Rep 33(7):1041–1052. https://doi.org/10.1007/s00299-014-1582-y
Oshima Y, Mitsuda N (2013) The MIXTA-like transcription factor MYB16 is a major regulator of cuticle formation in vegetative organs. Plant Signal Behav 8(11):e26826. https://doi.org/10.4161/psb.26826
Ozseyhan ME, Kang J, Mu X, Lu C (2018) Mutagenesis of the FAE1 genes significantly changes fatty acid composition in seeds of Camelina sativa. Plant Physiol Biochem 123:1–7. https://doi.org/10.1016/j.plaphy.2017.11.021
Philippe G, Sorensen I, Jiao C, Sun X, Fei Z, Domozych DS, Rose JK (2020) Cutin and suberin: assembly and origins of specialized lipidic cell wall scaffolds. Curr Opin Plant Biol 55:11–20. https://doi.org/10.1016/j.pbi.2020.01.008
Popielarska-Konieczna M, Kozieradzka-Kiszkurno M, Bohdanowicz J (2011) Cutin plays a role in differentiation of endosperm-derived callus of kiwifruit. Plant Cell Rep 30(11):2143–2152. https://doi.org/10.1007/s00299-011-1120-0
Prathap V, Tyagi A (2020) Correlation between expression and activity of ADP glucose pyrophosphorylase and starch synthase and their role in starch accumulation during grain filling under drought stress in rice. Plant Physiol Biochem 157:239–243. https://doi.org/10.1016/j.plaphy.2020.10.018
Regmi A, Shockey J, Kotapati HK, Bates PD (2020) Oil-Producing metabolons containing DGAT1 use separate substrate pools from those containing DGAT2 or PDAT. Plant Physiol 184(2):720–737. https://doi.org/10.1104/pp.20.00461
Reina-Pinto JJ, Voisin D, Kurdyukov S, Faust A, Haslam RP, Michaelson LV, Efremova N, Franke B, Schreiber L, Napier JA, Yephremov A (2009) Misexpression of FATTY ACID ELONGATION1 in the Arabidopsis epidermis induces cell death and suggests a critical role for phospholipase A2 in this process. Plant Cell 21(4):1252–1272. https://doi.org/10.1105/tpc.109.065565
Rowland O, Domergue F (2012) Plant fatty acyl reductases: enzymes generating fatty alcohols for protective layers with potential for industrial applications. Plant Sci 193-194:28–38. https://doi.org/10.1016/j.plantsci.2012.05.002
Saripalli G, Gupta PK (2015) AGPase: its role in crop productivity with emphasis on heat tolerance in cereals. Theor Appl Genet 128(10):1893–1916. https://doi.org/10.1007/s00122-015-2565-2
Souza CD, Kim SS, Koch S, Kienow L, Schneider K, McKim SM, Haughn GW, Kombrink E, Douglas CJ (2009) A novel fatty Acyl-CoA Synthetase is required for pollen development and sporopollenin biosynthesis in Arabidopsis. Plant Cell 21(2):507–525. https://doi.org/10.1105/tpc.108.062513
Turchetto-Zolet AC, Maraschin FS, de Morais GL, Cagliari A, Andrade CM, Margis-Pinheiro M, Margis R (2011) Evolutionary view of acyl-CoA diacylglycerol acyltransferase (DGAT), a key enzyme in neutral lipid biosynthesis. BMC Evol Biol 11:263. https://doi.org/10.1186/1471-2148-11-263
Vanhercke T, Belide S, Taylor MC, El Tahchy A, Okada S, Rolland V, Liu Q, Mitchell M, Shrestha P, Venables I, Ma L, Blundell C, Mathew A, Ziolkowski L, Niesner N, Hussain D, Dong B, Liu G, Godwin ID, Lee J, Rug M, Zhou XR, Singh SP, Petrie JR (2019) Up-regulation of lipid biosynthesis increases the oil content in leaves of Sorghum bicolor. Plant Biotechnol J 17(1):220–232. https://doi.org/10.1111/pbi.12959
Walther TC, Farese RV Jr (2012) Lipid droplets and cellular lipid metabolism. Annu Rev Biochem 81:687–714. https://doi.org/10.1146/annurev-biochem-061009-102430
Wang XL, Li XB (2009) The GhACS1 gene encodes an acyl-CoA synthetase which is essential for normal microsporogenesis in early anther development of cotton. Plant J 57(3):473–486. https://doi.org/10.1111/j.1365-313X.2008.03700.x
Wang Y, Wang M, Sun Y, Wang Y, Li T, Chai G, Jiang W, Shan L, Li C, Xiao E, Wang Z (2015) FAR5, a fatty acyl-coenzyme A reductase, is involved in primary alcohol biosynthesis of the leaf blade cuticular wax in wheat (Triticum aestivum L.). J Exp Bot 66(5):1165–1178. https://doi.org/10.1093/jxb/eru457
Xue Y, Xiao S, Kim J, Lung SC, Chen L, Tanner JA, Suh MC, Chye ML (2014) Arabidopsis membrane-associated acyl-CoA-binding protein ACBP1 is involved in stem cuticle formation. J Exp Bot 65(18):5473–5483. https://doi.org/10.1093/jxb/eru304
Yadav V, Molina I, Ranathunge K, Castillo IQ, Rothstein SJ, Reed JW (2014) ABCG transporters are required for suberin and pollen wall extracellular barriers in Arabidopsis. Plant Cell 26(9):3569–3588. https://doi.org/10.1105/tpc.114.129049
Yang T, Li Y, Liu Y, He L, Liu A, Wen J, Mysore KS, Tadege M, Chen J (2020) The 3-ketoacyl-CoA synthase WFL is involved in lateral organ development and cuticular wax synthesis in Medicago truncatula. Plant Mol Biol. https://doi.org/10.1007/s11103-020-01080-1
Yeung E, Chan C (2015) The Glycol Methacrylate Embedding Resins—Technovit 7100 and 8100. pp. 67–82
Zhang CL, Hu X, Zhang YL, Liu Y, Wang GL, You CX, Li YY, Hao YJ (2020) An apple long-chain acyl-CoA synthetase 2 gene enhances plant resistance to abiotic stress by regulating the accumulation of cuticular wax. Tree Physiol 40(10):1450–1465. https://doi.org/10.1093/treephys/tpaa079
Zhao L, Katavic V, Li F, Haughn GW, Kunst L (2010) Insertional mutant analysis reveals that long-chain acyl-CoA synthetase 1 (LACS1), but not LACS8, functionally overlaps with LACS9 in Arabidopsis seed oil biosynthesis. Plant J 64(6):1048–1058. https://doi.org/10.1111/j.1365-313X.2010.04396.x
Zhao L, Haslam TM, Sonntag A, Molina I, Kunst L (2019) Functional overlap of Long-Chain Acyl-CoA synthetases in Arabidopsis. Plant Cell Physiol 60(5):1041–1054. https://doi.org/10.1093/pcp/pcz019
Zheng P, Allen WB, Roesler K, Williams ME, Zhang S, Li J, Glassman K, Ranch J, Nubel D, Solawetz W, Bhattramakki D, Llaca V, Deschamps S, Zhong GY, Tarczynski MC, Shen B (2008) A phenylalanine in DGAT is a key determinant of oil content and composition in maize. Nat Genet 40(3):367–372. https://doi.org/10.1038/ng.85
Acknowledgements
The authors thank Wenli Hu (Center for Excellence in Molecular Plant Sciences, Shanghai Institute of Plant Physiology and Ecology, Chinese Academy of Sciences) for assistance with the GC-MS analysis; and Xiaoyan Gao, Jiqin Li and Zhiping Zhang (Center for Excellence in Molecular Plant Sciences, Shanghai Institute of Plant Physiology and Ecology, Chinese Academy of Sciences) for assistance with the cryo-SEM and TEM analyses.
Funding
This work was supported by the funding from the national natural science foundation of China, NSFC (Grant Nos. 31600684, U1738107, 31570859 and 31370214).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and experiment conduction were performed by J.J. and J.D.. Data collection and analysis were performed by J.J., J.Z., J.E. and W.C.. The first draft of the manuscript was written by J.J. and edited by J.E.. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Manoj Prasad.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jin, J., Essemine, J., Duan, J. et al. Rice calli may decelerate its metabolism to adapt hormone free medium. Plant Cell Tiss Organ Cult 145, 223–238 (2021). https://doi.org/10.1007/s11240-020-02004-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-02004-z