Abstract
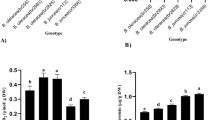
A method for in vitro screening and selection of drought (moisture stress) tolerant mango calli was developed. Poly ethylene glycol (PEG) (Molecular weight 6000) at various concentrations was used to induce moisture stress and reveal the underlying mechanisms used by mango calli for low moisture stress tolerance. The results demonstrated that there was significant impact of PEG induced moisture stress on various growth and biochemical parameters of mango calli. Based on the mortality rate, the calculated Lethal Dose 50 suggested that amongst the five mango genotypes, the polyembryonic (Olour, Kurakkan and K-2) had almost double tolerance level as compared to the monoembryonic (Amrapali and Dashehari) genotypes. Osmolytes like proline and total soluble sugars were higher in calli of the tolerant as compared to susceptible genotypes. The highest proline (0.683 µM g−1 FW) and total soluble sugars (24.61 mg g−1 FW) were observed in Olour. However, the highest malondialdehyde was in noted in Dashehari (20.25 µM g−1 FW) followed by Amrapali (19.68 µM g−1 FW). The activity of different antioxidant enzymes, namely, catalase, super oxide dismutase, peroxidase, and glutathione reductase were increased in the mango calli when grown in the PEG supplemented media though intensity varied in susceptible and tolerant genotypes. These results can be used for in vitro screening and selection for low moisture stress tolerance in other mango genotypes, somaclones, in vitro induced mutants etc.
Key message
PEG induced drought stress reduced the growth and modulated various biochemical parameters under in vitro conditions. Polyembryonic mango genotypes (Olour and Kurakkan) were observed to be distinctly moisture stress tolerant.




Similar content being viewed by others
Abbreviations
- PEG:
-
Poly ethylene glycol
- LD50 :
-
Lethal Dose 50
- CIM:
-
Callus induction media
- B5:
-
Gamborg B5 medium
- MS:
-
Murashige and Skoog’s medium
- 2,4-D:
-
2,4-Dichlorophenoxy acetic acid
- FW:
-
Fresh weight
- DW:
-
Dry weight
- CWC:
-
Callus water content
- RGR:
-
Relative growth rate
- MDA:
-
Malondialdehyde
- TBARS:
-
Thiobarbituric acid reactive substances
- CAT:
-
Catalase
- SOD:
-
Superoxide dismutase
- POD:
-
Peroxidase
- GR:
-
Glutathione reductase
References
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahmad MSA, Javed F, Ashraf M (2007) Iso–osmotic effect of NaCl and PEG on growth, cations and free proline accumulation in callus tissue of two indica rice (Oryza sativa L.) genotypes. Plant Growth Reg 53:53. https://doi.org/10.1007/s10725-007-9204-0
Akula R, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Sign Behav 6:1720–1731. https://doi.org/10.4161/psb.6.11.17613
Al-Khayri JM, Al-Bahrany AM (2004) Growth, water content, and proline accumulation in drought–stressed callus of date palm. Biol Planta 48:105–108. https://doi.org/10.1023/B:BIOP.0000024283.74919.4c
Bajji M, Lutts S, Kinet JM (2001) Water deficit effects on solute contribution to osmotic adjustment as a function of leaf ageing in three durum wheat (Triticum durum Desf.) cultivars performing differently in arid conditions. Plant Sci 160:669–681. https://doi.org/10.1016/S0168-9452(00)00443-X
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water–stress studies. Plant Soil 39(1):205–207. https://doi.org/10.1007/BF00018060
Bhaskaran S, Smith RH, Newton RJ (1985) Physiological changes in cultured sorghum cells in response to induced water stress: I. Free proline Plant Physiol 79:266–269. https://doi.org/10.1104/pp.79.1.266
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Castillo FJ, Penel C, Greppin H (1984) Peroxidase release induced by ozone in Sedum album leaves: involvement of Ca2+. Plant Physiol 74:846–851. https://doi.org/10.1104/pp.74.4.846
Dayal V, Dubey AK, Singh SK, Sharma RM, Dahuja A, Kaur C (2016) Growth, yield and physiology of mango (Mangifera indica L.) cultivars as affected by polyembryonic rootstocks. Scient Hortic 199:186–197. https://doi.org/10.1016/j.scienta.2015.12.042
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32:93–101. https://doi.org/10.1093/jxb/32.1.93
Elsheery NI, Cao KF (2008) Gas exchange, chlorophyll fluorescence, and osmotic adjustment in two mango cultivars under drought stress. Acta Physiol Plant 30:769–777. https://doi.org/10.1007/s11738-008-0179-x
Errabii T, Gandonou CB, Essalmani H, Abrini J, Idaomar M, Skali–Senhaji N (2006) Growth, proline and ion accumulation in sugarcane callus cultures under drought–induced osmotic stress and its subsequent relief. African J Biotech 5:1488–1493
Errabii T, Gandonou CB, Essalmani H, Abrini J, Idaomar M, Senhaji NS (2007) Effects of NaCl and mannitol induced stress on sugarcane (Saccharum sp.) callus cultures. Acta Physiol Plant 29:95. DOI https://doi.org/10.1007/s11738-006-0006-1
Fini A, Guidi L, Ferrini F, Brunetti C, Di Ferdinando M, Biricolti S, Tattini M (2012) Drought stress has contrasting effects on antioxidant enzymes activity and phenylpropanoid biosynthesis in Fraxinus ornus leaves: an excess light stress affair? J Plant Physiol 169:929–939. https://doi.org/10.1016/j.jplph.2012.02.014
Finney DJ (1952) Probit analysis: a statistical treatment of the sigmoid response curve, 2nd edn. Cambridge University Press, Cambridge
Gamborg OL, Miller R, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exper Cell Res 50:151–158. https://doi.org/10.1016/0014-4827(68)90403-5
Golkar P, Bakhshi G, Vahabi MR (2020) Phytochemical, biochemical, and growth changes in response to salinity in callus cultures of Nigella sativa L. Vitro Cell Develop Biol Plant 56:247–258. https://doi.org/10.1007/s11627-020-10058-z
Hare PD, Cress WA (1997) Metabolic implications of stress–induced proline accumulation in plants. Plant Growth Regul 21:79–102. https://doi.org/10.1023/A:1005703923347
Hassan NS, Shaaban LD, Hashem ESA, Seleem EE (2004) In vitro selection for water stress tolerant callus line of Helianthus annus L. cv. Myak International J Agric Biol 6:13–18
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Archiv Biochem Biophy 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Helaly MN, El-Hosieny HA, El–Sarkassy NM, Fuller MP, (2017) Growth, lipid peroxidation, organic solutes, and anti–oxidative enzyme content in drought–stressed date palm embryogenic callus suspension induced by polyethylene glycol. Vitro Cell Develop Biol Plant 53:133–141. https://doi.org/10.1007/s11627-017-9815-8
Jayasankar S, Litz RE, Gray DJ, Moon PA (1999) Responses of embryogenic mango cultures and seedling bioassays to a partially purified phytotoxin produced by a mango leaf isolate of Colletotrichum gloeosporioides Penz. Vitro Cell Develop Biol Plant 35:475–479. https://doi.org/10.1007/s11627-999-0071-4
Jones HG, Corlett JE (1992) Current topics in drought physiology. J Agric Sci 119:291–296. https://doi.org/10.1017/S0021859600012144
Joshi R, Shukla A, Sairam RK (2011) In vitro screening of rice genotypes for drought tolerance using polyethylene glycol. Acta Physiol Plant 33:2209. https://doi.org/10.1007/s11738-011-0760-6
Krishna H, Singh SK (2007) Biotechnological advances in mango (Mangifera indica L.) and their future implication in crop improvement-a review. Biotech Adv 25:223–243. https://doi.org/10.1016/j.biotechadv.2007.01.001
Kusvuran S, Ellialtıoglu SS, Talhouni M, Sonmez K, Kıran S (2014) Effects of salt and drought stresses on physiological and biochemical changes in callus tissues of melon cultivars. VI Balkan Symposium on Vegetables and Potatoes 1142:239–246
Litz RE, Schaffer B (1987) Polyamines in adventitious and somatic embryogenesis in mango (Mangifera indica L.). J Plant Physiol 128:251–258
Masoabi M, Lloyd J, Kossmann J, van der Vyver C (2018) Ethyl methanesulfonate mutagenesis and in vitro polyethylene glycol selection for drought tolerance in sugarcane (Saccharum spp.). Sugar Tech 20:50–59. https://doi.org/10.1007/s12355-017-0524-8
Misra N, Dwivedi UN (2004) Genotypic difference in salinity tolerance of green gram cultivars. Plant Sci 166:1135–1142. https://doi.org/10.1016/j.plantsci.2003.11.028
Misra N, Gupta AK (2005) Effect of salt stress on proline metabolism in two high yielding genotypes of green gram. Plant Sci 169:331–339. https://doi.org/10.1016/j.plantsci.2005.02.013
Misra N, Saxena P (2009) Effect of salicylic acid on proline metabolism in lentil grown under salinity stress. Plant Sci 177:181–189. https://doi.org/10.1016/j.plantsci.2009.05.007
Mohamed MF, Tawfik AA (2006) Dehydration-induced alterations in growth and osmotic potential of callus from six tepary bean lines varying in drought resistance. Plant Cell Tissue Organ Cult 87:255–262. https://doi.org/10.1007/s11240-006-9159-2
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Planta 15:473–497
Nath AK, Kumari S, Sharma DR (2005) In vitro selection and characterization of water stress tolerant cultures of bell pepper. Indian J Plant Physiol 10:14
Obaid OH, Reddy SK (2019) In vitro selection of drought stress tolerance in rice (Oryza sativa L.) var. Jarava Plant Tissue Cult Biotech 29:127–131. https://doi.org/10.3329/ptcb.v29i1.41985
Pandey SN, Dinesh MR (2010) Mango. Directorate of Information and Publications of Agriculture, Indian Council of Agricultural Research, Krishi Anusandhan Bhavan-1, Pusa, New Delhi, 110012, India.
Patade VY, Suprasanna P, Bapat VA (2008) Gamma irradiation of embryogenic callus cultures and in vitro selection for salt tolerance in sugarcane (Saccharum officinarum L.). Agricul Sci China 7:1147–1152. https://doi.org/10.1016/S1671-2927(08)60158-3
Patade VY, Bhargava S, Suprasanna P (2011) Salt and drought tolerance of sugarcane under iso–osmotic salt and water stress: growth, osmolytes accumulation, and antioxidant defense. J Plant Interac 6:275–282. https://doi.org/10.1080/17429145.2011.557513
Patade VY, Bhargava S, Suprasanna P (2012) Effects of NaCl and iso–osmotic PEG stress on growth, osmolytes accumulation and antioxidant defense in cultured sugarcane cells. Plant Cell Tissue Organ Cult 108:279–286. https://doi.org/10.1007/s11240-011-0041-5
Puente-Garza CA, Meza-Miranda C, Ochoa-Martinez D, Garcia-Lara S, (2017) Effect of in vitro drought stress on phenolic acids, flavonols, saponins, and antioxidant activity in Agave salmiana. Plant Physiol Biochem 115:400–407. https://doi.org/10.1016/j.plaphy.2017.04.012
Rao S, Jabeen FTZ (2013) In vitro selection and characterization of polyethylene glycol (PEG) tolerant callus lines and regeneration of plantlets from the selected callus lines in sugarcane (Saccharum officinarum L.). Physiol Mol Biol Plants 19:261–268. https://doi.org/10.1007/s12298-013-0162-x
Sadasivam S, Manickam A (1992) Biochemical Methods for Agricultural Sciences. Wiley Eastern Limited, New Delhi, pp 11–12
Seckin B, Sekmen AH, Turkan I (2009) An enhancing effect of exogenous mannitol on the antioxidant enzyme activities in roots of wheat under salt stress. J Plant Growth Regul 28:12. https://doi.org/10.1007/s00344-008-9068-1
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Meth Enzymol 299:152–178
Smith IK, Vierheller TL, Thorne CA (1988) Assay of glutathione reductase in crude tissue homogenates using 5, 5′–dithiobis (2–nitrobenzoic acid). Analyt Biochem 175:408–413. https://doi.org/10.1016/0003-2697(88)90564-7
Sofo A, Dichio B, Xiloyannis C, Masia A (2005) Antioxidant defences in olive trees during drought stress: changes in activity of some antioxidant enzymes. Func Plant Biol 32:45–53. https://doi.org/10.1071/FP04003
Srivastava DK, Gupta VK, Sharma DR (1995) In vitro selection and characterization of water stress tolerant callus cultures of tomato (Lycoperiscon esculentum L.). Indian J Plant Physiol 38:99–104
Tworkoski T, Fazio G, Glenn DM (2016) Apple rootstock resistance to drought. Sci Hortic 204:70–78. https://doi.org/10.1016/j.scienta.2016.01.047
Zhang C, Shi S, Wang B, Zhao J (2018) Physiological and biochemical changes in different drought–tolerant alfalfa (Medicago sativa L.) varieties under PEG–induced drought stress. Acta Physiol Plant 40:25. https://doi.org/10.1007/s11738-017-2597-0
Acknowledgements
Authors sincerely acknowledge the ICAR-Indian Agricultural Research Institute, New Delhi for providing the research facilities and Department of Science & Technology, Ministry of Science and Technology, Government of India for Grant of DST-INSPIRE fellowship to the senior author.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Sergio J. Ochatt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pradhan, S., Singh, S.K., Srivastav, M. et al. Poly ethylene glycol mediated in vitro screening and physico-biochemical changes induced in mango callus due to moisture stress. Plant Cell Tiss Organ Cult 145, 155–172 (2021). https://doi.org/10.1007/s11240-020-01999-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01999-9