Abstract
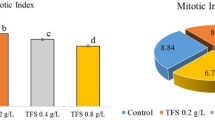
Polycyclic aromatic hydrocarbons (PAHs) present a significant threat to human health, in which 3,4-Benzopyrene as one of the three major carcinogens has been widely studied, while there are few researches of roots differentiation under 3,4-Benzopyrene exposure for understanding the mechanism of its phytoremediation. In this paper, the adventitious root differentiation of Triarrherca sacchariflora in vitro culture seedlings was observed under 3,4-Benzopyrene exposure. The results showed that 2.0 mg/L 3,4-Benzopyrene had significant positive effects on the adventitious root differentiation, while the higher dosage of 20.0 mg/L showed inhibition effects. 3,4-Benzopyrene could cause the remarkable reduction of the endogenous IAA and the ratios of IAA/ZR, IAA/DHZR, IAA/iPA and IAA/(ZR+DHZR+iPA) in stem explants of in vitro cultural seedlings of T. sacchariflora. The responses of coleoptile elongation of Zea mays and the DR5::GUS reporter gene of Arabidopsis thaliana to 3,4-Benzopyrene exposure were all positive comparing with the controls. It could be inferred from the results that the 3,4-Benzopyrene may be an auxin-like substance. In our knowledge, it was the first report concerning the 3,4-Benzopyrene with respect to the adventitious root differentiation of T. sacchariflora in vitro culture seedlings and its auxin-like biological effect analysis. It is the meaningful step to further understanding the 3,4-Benzopyrene effects on the plant endogenous signal substances that regulate the plant growth and development which closely related to the phytoremediation of PAHs.
Key message
3,4-Benzopyrene might be an auxin-like substance with the positive biological effect on the adventitious root differentiation via regulating some endogenous hormones of in vitro cultural seedlings of T. sacchariflora.




Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analysed during the current study are not publicly available due [REASON WHY DATA ARE NOT PUBLIC] but are available from the corresponding author on reasonable request.
References
Arumugam TV, Gleichmann M, Tang SC, Mattson MP (2006) Hormesis/preconditioning mechanisms, the nervous system and aging. Age Res Rev 5(2):165–178. https://doi.org/10.1016/j.arr.2006.03.003
Banks MK, Lee E, Schwab AP (1999) Evaluation of dissipation mechanisms for benzo [a] pyrene in the rhizosphere of tall fescue. Journal of Environmental Quality 28(1):294–298. https://doi.org/10.2134/jeq1999.00472425002800010036x
Belz RG, Piepho HP (2014) Interspecies variability of plant hormesis by the antiauxin PCIB in a laboratory bioassay. Journal of Plant Growth Regulation 33:499–512. https://doi.org/10.1007/s00344-013-9400-2
Binet P, Portal JM, Leyval C (2000) Dissipation of 3-6-ring Polycyclic aromatic hydrocarbons in the rhizosphere of ryegrass. Soil Biology & Biochemistry 32(14):2011–2017. https://doi.org/10.1016/s0038-0717(00)00100-0
Calabrese EJ (2013) Hormetic mechanisms. Crit Rev Toxicol 43(7):580–606. https://doi.org/10.3109/10408444.2013.808172
Calabrese EJ (2016) Preconditioning is hormesis. Part I: documentation, dose-response features and mechanistic foundations. Pharm Res 110:242–264. https://doi.org/10.1016/j.phrs.2015.12.021
Calabrese EJ, Baldwin LA (2001) The frequency of U-shaped dose responses in the toxicological literature. Toxicol Sci 62(2):330–338. https://doi.org/10.1093/toxsci/62.2.330
Calabrese EJ, Blain RB (2011) The hormesis database: the occurrence of hormetic dose responses in the toxicological literature. Regul Toxicol Pharmacol 61:73–81. https://doi.org/10.1016/j.yrtph.2011.06.003
Calabrese EJ, Blain R (2004) The occurrence of hormetic dose responses in the toxicological literature, the hormesis database: an overview. Toxicology and Applied Pharmacology 202:289–301. https://doi.org/10.1016/j.taap.2004.06.023
Cao HM, Wang CP, Liu HB, Jia WL, Sun HW (2020) Enzyme activities during Benzo[a] pyrene degradation by the fungus Lasiodiplodia theobromae isolated from a polluted soil. Scientific Reports 10(1):1–11. https://doi.org/10.1038/s41598-020-57692-6
Cerniglia CE (1984) Microbial metabolisms of polycyclic aromatic hydrocarbons. Adv Appl Micro 30:31–71. https://doi.org/10.1016/S0065-2164(08)70052-2
Chen SJ, Zhu XL, Feng XZ, Huang LQ, Mei YQ (2012) Effect of polycyclic aromatic hydrocarbons (PAHs) on plant. Bulletin of Biology 45(2):9–11
Christian M, Hannah WB, Lüthen H, Jones AM (2008) Identification of auxins by a chemical genomics approach. Experimental Botany 59(10):2757–2767. https://doi.org/10.1093/jxb/ern133
Gräf W, Nowak W (1966) Promotion of growth in lower and higher plants by carcinogenic PAH. Archiv für Hygiene und Bakteriologie 150(6):513–528
Hastings A, Clifton-Brown J, Wattenbach M, Mitchell CP, Stampfl P, Smith P (2009) Future energy potential of miscanthus in europe. Global Change Biology Bioenergy 1(2):180–196. https://doi.org/10.1111/j.1757-1707.2009.01012.x
Hunter RD, Ekunwe SIN, Dodor DE, Hwang HM, Ekunwe L (2005) Bacillus subtilis is a Potential Degrader of Pyrene and Benzo[a]pyrene. Int J Environ Res Public Health 2(2):267–271. https://doi.org/10.3390/ijerph2005020010
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. The EMBO Journal 6(13):3901–3907. https://doi.org/10.1002/j.1460-2075.1987.tb02730.x
Jouanneau Y, Willison JC, Meyer C, Krivobok S, Chevron N, Besombes JL, Blake G (2005) Stimulation of pyrene mineralization in freshwater sediments by bacterial and plant bioaugmentation. Environ Sci Technol 39(15):5729–5735. https://doi.org/10.2021/es050412d
Juhasz AL, Naidu R (2000) Bioremediation of high molecular weight polycyclic aromatic hydrocarbons: a review of the microbial degradation of benzo[a]pyrene. Int Biodeter Biodegr 45(1-2):57–88. https://doi.org/10.1016/s0964-8305(00)00052-4
Li SD, Zhang SL, Ma JC (1995) Degradation and forms of BaP in soil and rice plant. Acta Agriculturae Nucleatae 9(2):113–119
Liu Y, Zhou LL (2011) Comparative Studies on Absorption of Benzo(a) pyrene (BaP) in City Atmosphere by Leaves of 8 Street Plants. Urban Environment & Urban ecology 4:5–8. https://doi.org/10.1016/S1671-2927(11)60313-1
Lourdes RF, Melendez K, Hudson LG, Lauer FT, Burchiel SW (2009) EGF-receptor phosphorylation and downstream signaling are activated by benzo[a]pyrene 3,6-quinone and benzo[a]pyrene 1,6-quinone in human mammary epithelial cells. Toxicol Appl Pharm 235(3):321–328. https://doi.org/10.1016/j.taap.2008.12.022
Lüthen H, Bigdon M, Böttger M (1990) Re-examination of the acid growth theory of auxin action. Plant Physiology 93(3):931–939. https://doi.org/10.1104/pp.93.3.931
Malamy JE, Benfey PN (1997) Organization and cell differentiation in lateral roots of Arabidopsis thaliana. Development 124(1):33–44. https://doi.org/10.1146/annurev.cellbio.13.1.779
Masumura K, Toyoda-Hokaiwado N, Niimi N, Grúz P, Wada NA, Takeiri A, Jishage KI, Mishima M, Nohmi T (2017) Limited ability of DNA polymerase kappa to suppress benzo[a]pyrene-induced genotoxicity in vivo. Environ Mol Mutagen 58(9):644–653. https://doi.org/10.1002/em.22146
Mattson M, Calabrese E (2010) Hormesis: what it is and why it matters. In: Mattson MP, Calabrese E (eds) Hormesis: A Revolution in Biology, Toxicology and Medicine. Springer, New York, pp 1–14
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Orocio-Carrillo JA, del Carmen Rivera-Cruz M, Aranda-Ibañez EM, Trujillo-Narcía A, Hernández-Galvez G, Mendoza-López MR (2019) Hormesis under oil-induced stress in Leersia hexandra Sw. used as phytoremediator in clay soils of the Mexican humid tropic. Ecotoxicology (1):1–12. https://doi.org/10.1007/s10646-019-02106-1
Pang JF, Zheng MY, Wang AQ, Sun RY, Wang H, Jiang Y, Zhang T (2014) Catalytic conversion of concentrated miscanthus in water for ethylene glycol production. AIChE Journal 60(6):2254–2262. https://doi.org/10.1002/aic.14406
Qiu XJ, Leland TW, Shah SI, Sorensen D (1997) Field study: Grass remediation for clay soil contaminated with polycyclic aromatic hydrocarbons. ACS Symposium Series 664:186–199. https://doi.org/10.1021/bk-1997-0664.ch014
Rodriguez-Salus M, Bektas Y, Schroeder M, Knoth C, Vu T, Roberts P, Kaloshian I, Eulgem T (2015) The synthetic elicitor BHTC (2-(5-bromo-2-hydroxy-phenyl)-thiazolidine-4-carboxylic acid) links plant immunity to hormesis. Plant Physiology 170(1):444–458. https://doi.org/10.1104/pp.15.01058
Ulmasov T, Murfett J, Hagen G, Guilfoyle TJ (1997) Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin response elements. The Plant Cell 9(11):1963–1971. https://doi.org/10.2307/3870557
Wang CX, Yu SW (1984) Absorption of bap by corn roots and its accumulation in the plants. Acta Scientiae Circumstantiae 4(3):222–232
Wang ZL, Gao PH, Wei XY, Li Z, Li YL, Du KJ (2011) Establishment of plantlet regeneration system using seeds of Triarrherca sacchariflora (Maxin.)Nakai. Heber Journal of Forestry & Orchard Research, 2011 26(4):358–360
Wilson SC, Jones KC (1993) Bioremediation of soil contaminated with polynuclear aromatic hydrocarbons (PAHs): A review. Environ Pollut 81(3):229–249. https://doi.org/10.1016/0269-7491(93)90206-4
Zhao J, Li G, Wang BM, Li ZH, He ZP, Tian XL, Duan LS, Yin LN, Li J (2004) An enzyme immunoassay for the determination of chloramphenicol. Biotechnology 15(1):56–59
Acknowledgements
This work was supported by Natural Science Foundation of Hebei Province (Grant Number C2018204134).
Funding
This study was funded by Natural Science Foundation of Hebei Province (grant number C2018204134).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Ran Sun and Kejiu Du. The first draft of the manuscript was written by Ran Sun and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Ran Sun and Shuang Zhang contributed equally to the work.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethics approval
Informed consent.
Additional information
Communicated by Alison M.R. Ferrie.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sun, R., Zhang, S. & Du, K. Effects of 3,4-Benzopyrene on the adventitious root differentiation of in vitro culture seedlings of Triarrherca sacchariflora. Plant Cell Tiss Organ Cult 144, 641–647 (2021). https://doi.org/10.1007/s11240-020-01984-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-020-01984-2