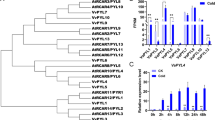
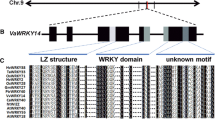
Abstract
The PYR/PYL/RCAR (hereafter referred to PYLs) proteins, which act as abscisic acid (ABA) receptors, have been reported to play a crucial role in response to drought stress. In this study, we used a drought-resistant accession of the wild Chinese grapevine, Vitis yeshanensis-’Yanshan-1’, as a model to understand the function of PYLs in grape and other fruit crops. We cloned six PYL genes from V. yeshanensis. Phylogenetic analysis showed that the encoded proteins could be classified into three subfamilies. All six grapevine PYL proteins contained conserved amino acids at positions implicated in ‘gate-and-latch’ mechanism of ABA binding and in the interaction with PP2C proteins. Gene expression profiles of VyPYLs in various tissues of ‘Yanshan-1’ plants revealed they were relatively strongly expressed in old leaves compared to young leaves. Analysis of the subcellular localization of the VyPYL proteins showed that they were localized in the cytoplasm and the nucleus. Yeast two-hybrid assays revealed that six VyPYL proteins selectively interacted with three VyPP2C proteins in an ABA-dependent or ABA-independent manner. Expression of one of the genes, VyPYL9, was induced by exogenous ABA and drought conditions. Heterologous expression of VyPYL9 in Arabidopsis enhanced ABA sensitivity during seed germination and primary root growth. Moreover, VyPYL9-expressing Arabidopsis plants showed enhanced drought tolerance, and this was associated with decreased water loss and increased activity of antioxidant defense systems. In addition, we found that ABA promoted leaf senescence in Arabidopsis plants expressing VyPYL9. Taken together, our results suggest that VyPYL9 may be involved in response of grapevine to drought stress via the ABA signaling pathway.
Key message
VyPYL9-expressing Arabidopsis plants showed enhanced drought tolerance, and this was associated with decreased water loss and increased activity of antioxidant defense systems. In addition, we found that ABA promoted leaf senescence in Arabidopsis plants expressing VyPYL9.









Similar content being viewed by others
References
Altschul S, Gish W, Miller W, Myers E, Lipman D (1990) Basic local alignment search tool. J Mol Biol 215(3):403–410
Antoni R, Gonzalez-Guzman M, Rodriguez L, Rodrigues A, Pizzio GA, Rodriguez PL (2012) Selective inhibition of clade A phosphatases type 2C by PYR/PYL/RCAR abscisic acid receptors. Plant Physiol 158(2):970–980. https://doi.org/10.1104/pp.111.188623
Antoni R, Gonzalez-Guzman M, Rodriguez L, Peirats-Llobet M, Pizzio GA, Fernandez MA, De Winne N, De Jaeger G, Dietrich D, Bennett MJ, Rodriguez PL (2013) PYRABACTIN RESISTANCE1-LIKE8 plays an important role for the regulation of abscisic acid signaling in root. Plant Physiol 161(2):931–941. https://doi.org/10.1104/pp.112.208678
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Bai G, Yang DH, Zhao Y, Ha S, Yang F, Ma J, Gao XS, Wang ZM, Zhu JK (2013) Interactions between soybean ABA receptors and type 2C protein phosphatases. Plant Mol Biol 83(6):651–664. https://doi.org/10.1007/s11103-013-0114-4
Balazadeh S, Kwasniewski M, Caldana C, Mehrnia M, Zanor MI, Xue G-P, Mueller-Roeber B (2011) ORS1, an H2O2-Responsive NAC Transcription Factor, Controls Senescence in Arabidopsis thaliana. Mol Plant 4(2):346–360. https://doi.org/10.1093/mp/ssq080
Blum A (2005) Drought resistance, water-use efficiency, and yield potential: are they compatible, dissonant, or mutually exclusive? Aust J Agric Res 56(11):1159–1168. https://doi.org/10.1071/ar05069
Boneh U, Biton I, Zheng C, Schwartz A, Ben-Ari G (2012) Characterization of potential ABA receptors in Vitis vinifera. Plant Cell Rep 31(2):311–321. https://doi.org/10.1007/s00299-011-1166-z
Chen Y, Feng L, Wei N, Liu Z-H, Hu S, Li X-B (2017) Overexpression of cotton PYL genes in Arabidopsis enhances the transgenic plant tolerance to drought stress. Plant Physiol Biochem 115:229–238. https://doi.org/10.1016/j.plaphy.2017.03.023
Choi H, Hong J, Ha J, Kang J, Kim SY (2000) ABFs, a family of ABA-responsive element binding factors. J Biol Chem 275(3):1723–1730
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant Journal 16(6):735–743
Cutler SR, Rodriguez PL, Finkelstein RR, Abrams SR (2010) Abscisic acid: emergence of a core signaling network. Annu Rev Plant Biol 61:651–679. https://doi.org/10.1146/annurev-arplant-042809-112122
Des Marais DL, McKay JK, Richards JH, Sen S, Wayne T, Juenger TE (2012) Physiological genomics of response to soil drying in diverse Arabidopsis accessions. Plant Cell 24(3):893–914. https://doi.org/10.1105/tpc.112.096180
Fang Y, Xiong L (2015) General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci 72(4):673–689. https://doi.org/10.1007/s00018-014-1767-0
Fang L, Su L, Sun X, Li X, Sun M, Karungo SK, Fang S, Chu J, Li S, Xin H (2016) Expression of Vitis amurensis NAC26 in Arabidopsis enhances drought tolerance by modulating jasmonic acid synthesis. J Exp Bot 67(9):2829–2845. https://doi.org/10.1093/jxb/erw122
Fujii H, Chinnusamy V, Rodrigues A, Rubio S, Antoni R, Park SY, Cutler SR, Sheen J, Rodriguez PL, Zhu JK (2009) In vitro reconstitution of an abscisic acid signalling pathway. Nature 462(7273):660–664. https://doi.org/10.1038/nature08599
Gonzalez-Guzman M, Pizzio GA, Antoni R, Vera-Sirera F, Merilo E, Bassel GW, Fernandez MA, Holdsworth MJ, Perez-Amador MA, Kollist H, Rodriguez PL (2012) Arabidopsis PYR/PYL/RCAR receptors play a major role in quantitative regulation of stomatal aperture and transcriptional response to abscisic acid. Plant Cell 24(6):2483–2496. https://doi.org/10.1105/tpc.112.098574
Gonzálezguzmán M, Rodríguez L, Lorenzoorts L, Pons C, Sarriónperdigones A, Fernández MA, Peiratsllobet M, Forment J, Morenoalvero M, Cutler SR (2014) Tomato PYR/PYL/RCAR abscisic acid receptors show high expression in root, differential sensitivity to the abscisic acid agonist quinabactin, and the capability to enhance plant drought resistance. J Exp Bot 65(15):4451–4464
Guo R, Zhao J, Wang X, Guo C, Li Z, Wang Y, Wang X (2015) Constitutive expression of a grape aspartic protease gene in transgenic Arabidopsis confers osmotic stress tolerance. Plant Cell, Tissue Organ Cult 121(2):275–287
Haines A, Kovats RS, Campbell-Lendrum D, Corvalán C (2006) Climate change and human health: impacts, vulnerability and public health. Public Health 120:585–596
Hao Q, Yin P, Li W, Wang L, Yan C, Lin Z, Wu JZ, Wang J, Yan SF, Yan N (2011) The molecular basis of ABA-independent inhibition of PP2Cs by a subclass of PYL proteins. Mol Cell 42(5):662–672. https://doi.org/10.1016/j.molcel.2011.05.011
He PC (2012) Wild resources of grapevine in China (in Chinese). China Agricultural Press, Beijing
He R, Zhuang Y, Cai Y, Aguero CB, Liu S, Wu J, Deng S, Walker MA, Lu J, Zhang Y (2018) Overexpression of 9-cis-epoxycarotenoid dioxygenase cisgene in grapevine increases drought tolerance and results in pleiotropic effects. Front Plant Sci 9:970. https://doi.org/10.3389/fpls.2018.00970
Hou H, Jia H, Yan Q, Wang X (2018) Overexpression of a SBP-box gene (VpSBP16) from Chinese wild vitis species in Arabidopsis improves salinity and drought stress tolerance. Int J Mol Sci 19(4):940. https://doi.org/10.3390/ijms19040940
Hu Y, Han YT, Zhang K, Zhao F-L, Li Y-J, Zheng Y, Wang Y-J, Wen Y-Q (2016) Identification and expression analysis of heat shock transcription factors in the wild Chinese grapevine (Vitis pseudoreticulata). Plant Physiol Biochem 99:1–10. https://doi.org/10.1016/j.plaphy.2015.11.020
Kim T-H (2014) Mechanism of ABA signal transduction: agricultural highlights for improving drought tolerance. J Plant Biol 57(1):1–8. https://doi.org/10.1007/s12374-014-0901-8
Kim H, Hwang H, Hong JW, Lee YN, Ahn IP, Yoon IS, Yoo SD, Lee S, Lee SC, Kim BG (2012) A rice orthologue of the ABA receptor, OsPYL/RCAR5, is a positive regulator of the ABA signal transduction pathway in seed germination and early seedling growth. J Exp Bot 63(2):1013–1024. https://doi.org/10.1093/jxb/err338
Klingler JP, Batelli G, Zhu JK (2010) ABA receptors: the START of a new paradigm in phytohormone signalling. J Exp Bot 61(12):3199–3210. https://doi.org/10.1093/jxb/erq151
Lee SC, Lim CW, Lan W, He K, Luan S (2013) ABA signaling in guard cells entails a dynamic protein-protein interaction relay from the PYL-RCAR family receptors to ion channels. Mol Plant 6(2):528–538. https://doi.org/10.1093/mp/sss078
Li X, Li G, Li Y, Kong X, Zhang L, Wang J, Li X, Yang Y (2018) ABA receptor subfamily III enhances abscisic acid sensitivity and improves the drought tolerance of Arabidopsis. Int J Mol Sci 19(7):1938. https://doi.org/10.3390/ijms19071938
Liang C, Liu Y, Li Y, Meng Z, Yan R, Zhu T, Wang Y, Kang S, Ali Abid M, Malik W, Sun G, Guo S, Zhang R (2017) Activation of ABA receptors gene GhPYL9-11A is positively correlated with cotton drought tolerance in transgenic Arabidopsis. Front Plant Sci 8:1453. https://doi.org/10.3389/fpls.2017.01453
Liu H, Yu C, Li H, Ouyang B, Wang T, Zhang J, Wang X, Ye Z (2015) Overexpression of ShDHN, a dehydrin gene from Solanum habrochaites enhances tolerance to multiple abiotic stresses in tomato. Plant Sci 231:198–211. https://doi.org/10.1016/j.plantsci.2014.12.006
Ma Y, Szostkiewicz I, Korte A, Moes D, Yang Y, Christmann A, Grill E (2009) Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 324(5930):1064–1068
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7(9):405–410
Miyazono K, Miyakawa T, Sawano Y, Kubota K, Kang HJ, Asano A, Miyauchi Y, Takahashi M, Zhi Y, Fujita Y, Yoshida T, Kodaira KS, Yamaguchi-Shinozaki K, Tanokura M (2009) Structural basis of abscisic acid signalling. Nature 462(7273):609–614. https://doi.org/10.1038/nature08583
Myles S, Boyko AR, Owens CL, Brown PJ, Grassi F, Aradhya MK, Prins B, Reynolds A, Chia JM, Ware D (2011) Genetic structure and domestication history of the grape. Proc Natl Acad Sci USA 108(9):3530–3535
Nishimura N, Sarkeshik A, Nito K, Park S-Y, Wang A, Carvalho PC, Lee S, Caddell DF, Cutler SR, Chory J, Yates JR, Schroeder JI (2009) PYR/PYL/RCAR family members are major in vivo ABI1 protein phosphatase 2C-interacting proteins in Arabidopsis. Plant J 61(2):290–299. https://doi.org/10.1111/j.1365-313X.2009.04054.x
Osakabe Y, Yamaguchi-Shinozaki K, Shinozaki K, Tran LSP (2014) ABA control of plant macroelement membrane transport systems in response to water deficit and high salinity. New Phytol 202(1):35–49
Park S-Y, Fung P, Nishimura N, Jensen DR, Fujii H, Zhao Y, Lumba S, Santiago J, Rodrigues A, Chow T-FF, Alfred SE, Bonetta D, Finkelstein R, Provart NJ, Desveaux D, Rodriguez PL, McCourt P, Zhu J-K, Schroeder JI, Volkman BF, Cutler SR (2009) Abscisic acid inhibits type 2C protein phosphatases via the PYR/PYL family of START proteins. Science 324(5930):1068–1071. https://doi.org/10.1126/science.1173041
Pizzio GA, Rodriguez L, Antoni R, Gonzalez-Guzman M, Yunta C, Merilo E, Kollist H, Albert A, Rodriguez PL (2013) The PYL4 A194T mutant uncovers a key role of PYR1-LIKE4/PROTEIN PHOSPHATASE 2CA interaction for abscisic acid signaling and plant drought resistance. Plant Physiol 163(1):441–455. https://doi.org/10.1104/pp.113.224162
Pou A, Flexas J, Alsina MdM, Bota J, Carambula C, de Herralde F, Galmes J, Lovisolo C, Jimenez M, Ribas-Carbo M, Rusjan D, Secchi F, Tomas M, Zsofi Z, Medrano H (2008) Adjustments of water use efficiency by stomatal regulation during drought and recovery in the drought-adapted Vitis hybrid Richter-110 (V. berlandieri × V. rupestris). Physiol Plantarum 134(2):313–323. https://doi.org/10.1111/j.1399-3054.2008.01138.x
Quan WL, Hu YL, Mu ZX, Shi HT, Chan ZL (2018) Overexpression of AtPYL5 under the control of guard cell specific promoter improves drought stress tolerance in Arabidopsis. Plant Physiol Biochem 129:150–157. https://doi.org/10.1016/j.plaphy.2018.05.033
Raghavendra AS, Gonugunta VK, Christmann A, Grill E (2010) ABA perception and signalling. Trends Plant Sci 15(7):395–401
Reichstein M, Bahn M, Ciais P, Frank D, Mahecha MD, Seneviratne SI, Zscheischler J, Beer C, Buchmann N, Frank DC, Papale D, Rammig A, Smith P, Thonicke K, van der Velde M, Vicca S, Walz A, Wattenbach M (2013) Climate extremes and the carbon cycle. Nature 500(7462):287–295. https://doi.org/10.1038/nature12350
Saavedra X, Modrego A, Rodríguez D, Gonzálezgarcía MP, Sanz L, Nicolás G, Lorenzo O (2010) The nuclear interactor PYL8/RCAR3 of Fagus sylvatica FsPP2C1 is a positive regulator of abscisic acid signaling in seeds and stress. Plant Physiol 152(1):133–150
Santiago J, Dupeux F, Round A, Antoni R, Park SY, Jamin M, Cutler SR, Rodriguez PL, Marquez JA (2009a) The abscisic acid receptor PYR1 in complex with abscisic acid. Nature 462(7273):665–668. https://doi.org/10.1038/nature08591
Santiago J, Rodrigues A, Saez A, Rubio S, Antoni R, Dupeux F, Park SY, Marquez JA, Cutler SR, Rodriguez PL (2009b) Modulation of drought resistance by the abscisic acid receptor PYL5 through inhibition of clade A PP2Cs. Plant J 60(4):575–588. https://doi.org/10.1111/j.1365-313X.2009.03981.x
Scholander PF, Bradstreet ED, Hemmingsen EA, Hammel HT (1965) Sap pressure in vascular plants: negative hydrostatic pressure can be measured in plants. Science 148(3668):339–346. https://doi.org/10.1126/science.148.3668.339
Sharp RE, Poroyko V, Hejlek LG, Spollen WG, Springer GK, Bohnert HJ, Nguyen HT (2004) Root growth maintenance during water deficits: physiology to functional genomics. J Exp Bot 55(407):2343–2351. https://doi.org/10.1093/jxb/erh276
Sheard LB, Zheng N (2009) Plant biology: signal advance for abscisic acid. Nature 462(7273):575–576. https://doi.org/10.1038/462575a
Su X, Zhou P, Wang R, Luo Z, Xia Z (2014) Overexpression of the maize psbA gene enhances sulfur dioxide tolerance in transgenic tobacco. Plant Cell, Tissue Organ Cult 120(1):303–311. https://doi.org/10.1007/s11240-014-0607-0
Sucu S, Yagci A, Yildirim K (2018) Changes in morphological, physiological traits and enzyme activity of grafted and ungrafted grapevine rootstocks under drought stress. Erwerbs-Obstbau 60(2):127–136. https://doi.org/10.1007/s10341-017-0345-7
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739. https://doi.org/10.1093/molbev/msr121
Tian X, Wang Z, Li X, Lv T, Liu H, Wang L, Niu H, Bu Q (2015) Characterization and functional analysis of pyrabactin resistance-like abscisic acid receptor family in rice. Rice 8(1):28. https://doi.org/10.1186/s12284-015-0061-6
Touma D, Ashfaq M, Nayak MA, Kao S-C, Diffenbaugh NS (2015) A multi-model and multi-index evaluation of drought characteristics in the 21st century. J Hydrol 526:196–207. https://doi.org/10.1016/j.jhydrol.2014.12.011
Tu M, Wang X, Huang L, Guo R, Zhang H, Cai J, Wang X (2016) Expression of a grape bZIP transcription factor, VqbZIP39, in transgenic Arabidopsis thaliana confers tolerance of multiple abiotic stresses. Plant Cell, Tissue Organ Cult 125(3):537–551. https://doi.org/10.1007/s11240-016-0969-6
Uauy C, Distelfeld A, Fahima T, Blechl A, Dubcovsky J (2006) A NAC gene regulating senescence improves grain protein, zinc, and iron content in wheat. Science 314(5803):1298–1301. https://doi.org/10.1126/science.1133649
Walter M, Chaban C, Schutze K, Batistic O, Weckermann K, Nake C, Blazevic D, Grefen C, Schumacher K, Oecking C, Harter K, Kudla J (2004) Visualization of protein interactions in living plant cells using bimolecular fluorescence complementation. Plant J 40(3):428–438. https://doi.org/10.1111/j.1365-313X.2004.02219.x
Wang Y, Yang Y, Zhang J, Pan X, Yizhen Wan (2004) Preliminary identification of drought resistance of chinese wild vitis species and its interspecific hybrids (in Chinese). Acta Horticulturae Sinica 31(6):711–714. https://doi.org/10.16420/j.issn.0513-353x.2004.06.001
Wang YG, Yu HQ, Zhang YY, Lai CX, She YH, Li WC, Fu FL (2014) Interaction between abscisic acid receptor PYL3 and protein phosphatase type 2C in response to ABA signaling in maize. Gene 549(1):179–185. https://doi.org/10.1016/j.gene.2014.08.001
Wei W, Cui MY, Hu Y, Gao K, Xie YG, Jiang Y, Feng JY (2018) Ectopic expression of FvWRKY42, a WRKY transcription factor from the diploid woodland strawberry (Fragaria vesca), enhances resistance to powdery mildew, improves osmotic stress resistance, and increases abscisic acid sensitivity in Arabidopsis. Plant Sci 275:60–74. https://doi.org/10.1016/j.plantsci.2018.07.010
Weiner JJ, Peterson FC, Volkman BF, Cutler SR (2010) Structural and functional insights into core ABA signaling. Curr Opin Plant Biol 13(5):495–502. https://doi.org/10.1016/j.pbi.2010.09.007
Xia XJ, Wang YJ, Zhou YH, Tao Y, Mao WH, Shi K, Asami T, Chen Z, Yu JQ (2009) Reactive oxygen species are involved in brassinosteroid-induced stress tolerance in cucumber. Plant Physiol 150(2):801–814. https://doi.org/10.1104/pp.109.138230
Xiong L, Schumaker KS, Zhu JK (2002) Cell signaling during cold, drought, and salt stress. Plant Cell 14(Suppl):S165
Xu H (2004) Study on drought resistance of grape rootstocks and varieties. Thesis for Master’s Degree. Shandong Agricultural University, Tai’an, Shandong, China
Yang YZ (2003) Identification of drought resistance and RAPD maker slinked to drought resistance genes in Chinese wild species. Thesis for Master’s Degree. Northwest A&F University, Yangling, Shaanxi, China
Yildirim K, Kaya Z (2017) Gene regulation network behind drought escape, avoidance and tolerance strategies in black poplar (Populus nigra L.). Plant Physiol Biochem 115:183–199. https://doi.org/10.1016/j.plaphy.2017.03.020
Yin P, Fan H, Hao Q, Yuan X, Wu D, Pang Y, Yan C, Li W, Wang J, Yan N (2009) Structural insights into the mechanism of abscisic acid signaling by PYL proteins. Nat Struct Mol Biol 16(12):1230–1236. https://doi.org/10.1038/nsmb.1730
Yoshida T, Mogami J, Yamaguchishinozaki K (2014) ABA-dependent and ABA-independent signaling in response to osmotic stress in plants. Curr Opin Plant Biol 21(21C):133–139
Yu JL, Yang L, Liu XB, Tang RJ, Wang Y, Ge HM, Wu MT, Zhang J, Zhao FG, Luan S, Lan WZ (2016) Overexpression of poplar pyrabactin resistance-like abscisic acid receptors promotes abscisic acid sensitivity and drought resistance in transgenic Arabidopsis. PLoS ONE 11(12):17. https://doi.org/10.1371/journal.pone.0168040
Yu J, Ge H, Wang X, Tang R, Wang Y, Zhao F, Lan W, Luan S, Yang L (2017) Overexpression of pyrabactin resistance-like abscisic acid receptors enhances drought, osmotic, and cold tolerance in transgenic poplars. Front Plant Sci 8:1752. https://doi.org/10.3389/fpls.2017.01752
Zhao Y, Chan ZL, Xing L, Liu XD, Hou YJ, Chinnusamy V, Wang PC, Duan CG, Zhu JK (2013) The unique mode of action of a divergent member of the ABA-receptor protein family in ABA and stress signaling. Cell Res 23(12):1380–1395. https://doi.org/10.1038/cr.2013.149
Zhao F-L, Li Y-J, Hu Y, Gao Y-R, Zang X-W, Ding Q, Wang Y-J, Wen Y-Q (2015) A highly efficient grapevine mesophyll protoplast system for transient gene expression and the study of disease resistance proteins. Plant Cell, Tissue Organ Cult 125(1):43–57. https://doi.org/10.1007/s11240-015-0928-7
Zhao Y, Chan Z, Gao J, Xing L, Cao M, Yu C, Hu Y, You J, Shi H, Zhu Y, Gong Y, Mu Z, Wang H, Deng X, Wang P, Bressan RA, Zhu JK (2016) ABA receptor PYL9 promotes drought resistance and leaf senescence. Proc Natl Acad Sci USA 113(7):1949–1954. https://doi.org/10.1073/pnas.1522840113
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53(53):247–273
Acknowledgements
This study was funded by the National Natural Science Foundation of China (Grant No. 31772264 to YQW) and by the open Project of the State Key Laboratory of Crop Stress Biology for Arid Areas (CSBAA2017014 to XCF).
Author information
Authors and Affiliations
Contributions
FLZ, XCF, and YQW conceived and designed the research. FLZ conducted most of the experiments. YG participated in the experiments. JL wrote the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Klaus Eimert.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, J., Zhao, FL., Guo, Y. et al. The ABA receptor-like gene VyPYL9 from drought-resistance wild grapevine confers drought tolerance and ABA hypersensitivity in Arabidopsis. Plant Cell Tiss Organ Cult 138, 543–558 (2019). https://doi.org/10.1007/s11240-019-01650-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01650-2