Abstract
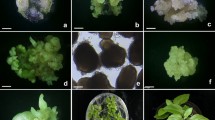
A procedure is described to regenerate plants from embryogenic suspension-derived protoplasts of ginger (Zingiber officinale Rosc.). Somatic embryogenic calli were induced from ginger shoot tips on solid MS medium with half the concentration of NH4NO3 and supplemented with 1.0 mg l−1 2,4-Dichloroacetic acid (2, 4-D) and 0.2 mg l−1 Kin. Rapid-growing and well-dispersed suspension cultures were established by subculturing the embryogenic calli in the same liquid medium. Protoplasts were isolated from embryogenic suspensions with an enzyme solution composed of 4.0 mg l−1 cellulase, 1.0 mg l−1 macerozyme, 0.1 mg l−1 pectolyase, 11% mannitol, 0.5% CaCl2 and 0.1% 2-(N-morpholino) ethane sulphonic acid (MES) for 12–14 h at 27°C with a yield of 6.27 × 106 protoplasts g−1 fresh weight. The protoplasts were cultured initially in liquid MS medium with 1.0 mg l−1 2, 4-D and 0.2 mg l−1 Kin. Then the protoplast-derived calli (1.5 cm2) were transferred to a basal MS medium containing 0.2 mg l−1 2, 4-D, 5.0 mg l−1 benzyladenine (BA), 3% sucrose and 0.7% agar. The white somatic embryos were transferred to MS medium lacking growth regulators for shoot development. Shoots developed into complete plantlets on a solid MS medium supplemented with 2.0 mg l−1 BA and 0.6 mg l−1 α-Naphthaleneacetic acid (NAA). In addition, the effects of AgNO3, activated charcoal (AC) and ascorbic acid (AA) on browning of protoplast-derived calli are discussed.




Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- AC:
-
Activated charcoal
- AA:
-
Ascorbic acid
- BA:
-
Benzyladenine
- CH:
-
Casein hydrolysate
- DTT:
-
Citric acid and 1,4-dithio-dl-threitol
- FDA:
-
Fluorescein diacetate
- Kin:
-
6-Furfurylaminopurine
- MES:
-
2-(N-morpholino) ethane sulphonic acid
- MS:
-
Murashige and Skoog medium (Murashige and Skoog 1962)
- NAA:
-
α-Naphthaleneacetic acid
- PVP:
-
Polyvinylpyrrolidone
References
Babu KN, Samsudeen K, Ravindran PN (1992) Direct regeneration of plantlets from immature inflorescence of ginger (Zingiber officinale Rosc.) by tissue culture. J Spices Aromatic Crops 1(1):43–48
Cao MD, Li JR, Qin DC, Wang JJ (2002) Effects of absorbents and components of medium on vanillin biosynthesis in cell cutures of Vanilla planifolia. Bull Bot Res 22(1):65–67
Chand S, Sahrawat AK (2001) Stimulatory effect of partial desiccation on plant regeneration in indica rice (Oryza sativa L.). J Plant Biochem Biotechnol 10:43–47
Dekker AJ, Rao AN, Gob CJ (1991) In vitro storage of multiple shoot cultures of ginger at ambient temperatures of 24–29°C. Sci Hortic 47:157–167
Feulgen R, Rossenbeck H (1924) Mikroskopisch-chemischer Nachweiseiner Nucleisäure vom Typus der Thymonucleinsäure unddie darauf beruhende elektive Färbung von Zellkernen inmikroskopischen Präparaten. Hoppe Seyler Z Physiol Chem 135:203–248
Geetha SP, Babu KN, Rema J (2000) Isolation of protoplasts from cardamom (Elettaria cardamomum Maton.) and ginger (Zingiber officinale Rosc.). J Spices Aromatic Crops 9(1):23–30
Guo YH, Zhang ZX (2005) Establishment and plant regeneration of somatic embryogenic cell suspension cultures of the Zingiber officinale Rosc. Sci Hortic 107:90–96
Hosoki T, Sagawa Y (1997) Clonal propagation of ginger (Zingiber officinale Rosc.) through tissue culture. HortScience 12:451–452
Jain RK, Jain S, Wang B, Wu R (1996) Stimulatory effect of water stress on plant regeneration in aromatic indica rice varieties. Plant Cell Rep 15:449–454
Kackar A, Bhat SR, Chandel KP (1993) Plant regeneration via somatic embryogenesis in ginger. Plant Cell Tissue Organ Cult 32(3):289–292
Mayer AM, Harel E (1979) Polyphenol oxidases in plants. Phytochemistry 18:193–215
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plantarum 15:473–497
Negrutiu I, Shillito R, Potrykus I, Biasini G, Sala F (1987) Hybrid genes in the analysis of transformation conditions: I. Setting up a simple method for direct gene transfer in plant protoplasts. Plant Mol Biol 8:363–373
Sharma TR, Singh BM (1997) High-frequency in vitro multiplication of disease-free Zingiber oficinale Rosc. Plant Cell Rep 17:68–72
Tang W, Harris LC, Outhavong V, Newton RJ (2004) Antioxidants enhance in vitro plant regeneration by inhibiting the accumulation of peroxidase in Virginia pine (Pinus virginiana Mill.). Plant Cell Rep 22(12):871–877
Taylor PWJ, Ko HL, Adkins SW (1992) Factors affecting protoplast isolation and the regeneration of shoot-like structures from protoplast-derived callus of sugarcane (Saccharum spp hybrids). Aust J Bot 40(6):863–876
Widholm J (1972) The use of FDA and phenosafranine for determining viability of cultured plant cells. Stain Technol 47:186–194
Zhu MY, Xu Ab, Yuan MB, Huang CN, Yu ZL, Wang LJ, Yu JJ (1990) Effects of amino acids on callus differentiation in barley anther culture. Plant Cell Tissue Organ Cult 22(3):201–204
Acknowledgments
This work was supported by The National Key Technologies Research and Development (R&D) Program of China (2001BA511B08-12 & 2006BAD13B06) and The Project of Agricultural Ministry (04-06-02B).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guo, Y., Bai, J. & Zhang, Z. Plant regeneration from embryogenic suspension-derived protoplasts of ginger (Zingiber officinale Rosc.). Plant Cell Tiss Organ Cult 89, 151–157 (2007). https://doi.org/10.1007/s11240-007-9223-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-007-9223-6