Abstract
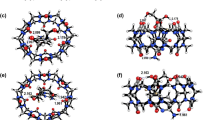
The structural geometries of cucurbit[n]uril CB[n] with n = 6–9 and their complexes with oseltamivir (OST) drug were obtained using the density functional theory computations. The stationary points of the most stable complexes were confirmed using vibrational frequency calculation. The complexation energies and electronic properties of CB[n]/OST complexes were investigated. The calculated results indicate that the intermolecular interactions in all the studied complexes occur via a large number of dipole–dipole interactions, especially hydrogen bonds between oxygen atoms of CB[n] and hydrogen atoms of amine of oseltamivir drug. The negative complexation energies of CB[n]/OST complexes in both gas and water phases indicate that the host–guest complexes are exothermic process and the complexes are more stable than its bare CB[n]. In addition, the CB[7]/OST complex is more stable than that of all studied CB[n]/OST complexes. The frequency calculation results of the most stable complexes for each of CBs indicate that complexations occur via a spontaneous process. The NBO analysis of complexes shows the transferring of partial charge from CB[n]s to oseltamivir which correspond to their MEP contours. The HOMO and the LUMO orbitals are localized on the oseltamivir in CB[n]/OST complexes. After drug complexation, the electronic properties also display that the energy gaps of CB[n] are significantly changed. All of the complexation properties point out that CB[n]s can act as a host for appropriately oseltamivir guest, even in aqueous solution.










Similar content being viewed by others
Availability of data and material
Not applicable (all data generated or analyzed during this study are included in this published article).
References
Webber MJ, Langer R (2017) Chem Soc Rev 46:6600–6620
Miskolczy Z, Megyesi M, Tarkanyi G, Mizsei R, Biczok L (2011) Org Biomol Chem 9:1061–1070
Cong H, Li CR, Xue SF, Tao Z, Zhub QJ, Wei G (2011) Org Biomol Chem 9:1041–1046
Krause-Heuer AM, Grant MP, Orkey N, Aldrich-Wright JR (2008) Aust J Chem 61:675–681
Kemp S, Wheate NJ, Wang S, Collins JG, Ralph SF, Day AI, Higgins VJ, Aldrich-Wright JR (2007) J Biol Inorg Chem 12:969–979
Zhang J, Ma PX (2013) Adv Drug Deliv Rev 65:1215–1233
Bani-Yaseen AD, Mo’ala A (2014) Spectrochim Acta A 131:424–431
Gallego-Yerga L, de la Torre C, Sansone F, Casnati A, Mellet CO, García Fernandez JM, Cena V (2021) Carbohydr Polym 252:117135
Wang J, Ding X, Guo X (2019) Adv Colloid Interface Sci 269:187–202
Basilotta R, Mannino D, Filippone A, Casili G, Prestifilippo A, Colarossi L, Raciti G, Esposito E, Campolo M (2021) Molecules 26:3963
Muzzalupo R, Nicoletta FP, Trombino S, Cassano R, Iemma F, Picci (2007) Colloids Surf B 58:197–202
Kemp S, Wheate NJ, Stootman FH, Aldrich-Wright JR (2007) Supramol Chem 19:475–484
Shchepotina EG, Pashkina EA, Yakushenko EV, Kozlov VA (2011) Nanotechnol Russ 6:773–779
Walker S, Oun R, McInnes FJ, Wheate NJ (2011) Isr J Chem 51:616–624
Wheate NJ, Limantoro C (2016) Supramol Chem 28:849–856
Suvitha A, Venkataramanan NS, Mizuseki H, Kawazoe Y, Ohuchi N (2010) J Incl Phenom Macrocycl Chem 66:213–218
Behrend R, Meyer E, Rusche F (1905) Justus Liebigs Ann Chem 339:1–37
Freeman WA, Mock WL, Shih NY (1981) J Am Chem Soc 103:7367–7368
Kim J, Jung I-S, Kim S-Y, Lee E, Kang J-K, Sakamoto S, Yamaguch K, Kim K (2000) J Am Chem Soc 122:540–541
Uzunova VD, Cullinane C, Brix K, Nau WM, Day AI (2010) Org Biomol Chem 8:2037–2042
Zhao Y, Buck DP, Morris DL, Pourgholami MH, Day AI, Collins JG (2008) Org Biomol Chem 6:4509–4515
Wheate NJ (2008) J Inorg Biochem 102:2060–2066
Corda E, Hernandez M, Sanchez-Cortes S, Sevilla P (2018) Colloids Surf A: Physicochem Eng Asp 557:66–75
Jeon YJ, Kim S-Y, Ko YH, Sakamoto S, Yamaguchi K, Kim K (2005) Org Biomol Chem 3:2122–2125
Nojini ZB, Yavari F, Bagherifar S (2012) J Mol Liq 166:53–61
Sabet M, Ganji MD (2013) J Mol Model 19:4013–4023
Assaf KI, Nau WM (2015) Chem Soc Rev 44:394–418
Gurbuz S, Idris M, Tuncel D (2015) Org Biomol Chem 13:330–347
Kim K, Selvapalam N, Ko YH, Park KM, Kim D, Kim J (2007) Chem Soc Rev 36:267–279
Hayden F, Treanor JJ, Fritz RS, Lobo M, Betts RF, Miller M, Kinnersley N, Mills RG, Ward P, Straus SE (1999) JAMA 282:1240–1246
Lindemann L, Jacobsen H, Schuhbauer D, Knoflach F, Gatti S, Wettstein JG, Loetscher H, Chu T, Ebeling M, Paulson JC, Prinssen E, Brockhaus M (2010) Eur J Pharmacol 628:6–10
Bartels P, von Tumpling W (2008) Sci Total Environ 405:215–225
Accinelli C, Caracciolo AB, Grenni P (2007) Int J Environ Anal Chem 87:579–587
Sacca ML, Accinelli C, Fick J, Lindberg R, Olsen B (2009) Chemosphere 75:28–33
Pinjari RV, Khedkar JK, Gejji SP (2010) J Incl Phenom Macrocycl Chem 66:371–380
Becke AD (1988) Phys Rev A 38:3098–3100
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji , Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JrJE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) GAUSSIAN 09, Revision A.02, Gaussian Inc, Wallingford CT
Barone V, Cossi M, Tomasi J (1998) J Comput Chem 19:404–417
Cossi M, Barone V (1998) J Chem Phys 109:6246–6254
Flükiger P, Lüthi HP, Portmann S (2000) MOLEKEL 4.3, Swiss center for scientific computing, Manno, Switzerland
Pichierri F (2006) J Mol Struct (Theochem) 765:151–152
Ochterski JW (2000) Thermochemistry in Gaussian, Gaussian Inc., Pittsburgh, PA
Safia H, Fatiha M, Belgacem B, Leila N (2020) J Mol Struct 1217:128390
Parr RG, Donelly RA, Levy M, Palke WE (1978) J Chem Phys 68:3801–3807
Janak JF (1978) Phys Rev B 18:7165–7168
Funding
This research project was financially supported by Mahasarakham University 2021, Thailand.
Author information
Authors and Affiliations
Contributions
W. Rakrai and B. Wanno contributed to the study conception and design. The DFT calculations were performed by W. Rakrai, C. Tabtimsai, and B. Wanno. The data analysis and the first draft of the manuscript were made by W. Rakrai and B. Wanno. Revising the manuscript critically for important intellectual content on subsequent versions of the manuscript has been done by W. Rakrai, C. Kaewtong, and B. Wanno. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The ethical standards have been met.
Consent for publication
All co-authors have seen and approved the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rakrai, W., Tabtimsai, C., Kaewtong, C. et al. Theoretical investigation of the complexation, structural, and electronic properties of complexes between oseltamivir drug and cucurbit[n = 6–9]urils. Struct Chem 33, 757–768 (2022). https://doi.org/10.1007/s11224-022-01888-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-022-01888-1