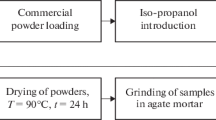
Results obtained from differential scanning calorimetry and a thermogravimetric analysis of zirconia-based nanocrystalline powder systems are presented. Heating is found to cause intense mass loss that increases with increase in the MgO content. Differential scanning calorimetry has revealed that the total energy expended for reactions involved in the powder heating process increases with increase in the MgO content. The heated powders are characterized by desorption of water. For 10 wt. % MgO, residual nitrates are seen to decompose into NO2, N2O, or NO.
Similar content being viewed by others
References
I. P. Suzdalev and P. I. Suzdalev, Usp. Khim., 75, No. 8, 715–752 (2006).
S. N. Kulkov, S. P. Buyakova, and V. I. Maslovskii, Vestn. TGU, No. 13, 61–87 (2003).
M. Rezai and S. M. Alavi, J. Porous Mater., No. 10, 117–125 (2008).
W. Kucza and J. Oblakowskii, J. Therm. An. Calorimetry, 88, No. 1, 65–69 (2007).
A. V. Shevchenko, A. K. Ruban, E. V. Dudnik and V. A. Mel’nikova, Powder Metall. Met. Ceramics, 36, No. 7–8, 420–424 (1997).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Izvestiya Vysshikh Uchebnykh Zavedenii, Fizika, No. 12, pp. 45–48, December, 2010.
Rights and permissions
About this article
Cite this article
Kanaki, A.V., Buyakova, S.P., Volkov, S.A. et al. Differential scanning calorimetry and a thermogravimetric analysis of nanozirconia-based powder systems. Russ Phys J 53, 1271–1274 (2011). https://doi.org/10.1007/s11182-011-9559-6
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11182-011-9559-6