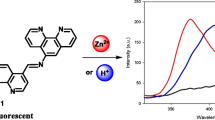
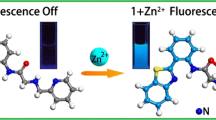
Abstract
A previously undescribed fluorescent sensor for zinc cations based on a 4-methoxynaphthalimide fluorophore and a receptor fragment represented by a dipicolylamine derivative was synthesized, and its spectral and luminescent properties were studied. The resulting fluorescent sensor demonstrated fluorescence enhancement upon complexation with Zn2+ cations due to the formation of ligand—metal complexes with a composition of 1: 1, in which the electron transfer process is suppressed.
Similar content being viewed by others
References
D. Wu, A. C. Sedgwick, T. Gunnlaugsson, E. U. Akkaya, J. Yoon, T. D. James, Chem. Soc. Rev., 2017, 46, 7105; DOI: https://doi.org/10.1039/c7cs00240h.
M. Formica, V. Fusi, L. Giorgi, M. Micheloni, Coord. Chem. Rev., 2012, 256, 170.
B. L. Vallee, K. H. Falchuk, Physiol. Rev., 1993, 73, 79; DOI: https://doi.org/10.1152/physrev.1993.73.1.79.
Q. Truong-Tran, J. Carter, R. C. Ruffin, P. D. Zalewski, Biometals, 2001, 14, 315; DOI: https://doi.org/10.1023/a:1012993017026.
E. L. Que, D. W. Domaille, C. J. Chang, Chem. Rev., 2008, 108, 1517; DOI: https://doi.org/10.1021/cr078203u.
B. Valeur, I. Leray, Coord. Chem. Rev., 2000, 205, 3; DOI: https://doi.org/10.1016/S0010-8545(00)00246-0.
T. L. Mako, J. M. Racicot, M. Levine, Chem. Rev., 2019, 119, 322; DOI: https://doi.org/10.1021/acs.chemrev.8b00260.
J. R. Lakowicz, Principles of Fluorescent Spectroscopy, 3rd ed., Springer Science + Business Media, New York, 2006, 954 pp.
B. T. Huy, D. T. Thangadurai, M. Sharipov, N. N. Nghia, N. Van Cuong, Y. Lee, Microchem. J., 2022, 179, 107511; DOI: https://doi.org/10.1016/j.microc.2022.107511.
P. A. Panchenko, A. S. Polyakova, Yu. V. Fedorov, O. A. Fedorova, Russ. Chem. Bull., 2021, 70, 1939; DOI: https://doi.org/10.1007/s11172-021-3300-6.
P. A. Panchenko, N. V. Leichu, Yu. V. Fedorov, O. A. Fedorova, Makrogeterotsikly [Macroheterocycles], 2019, 12, 319 (in Russian); DOI: https://doi.org/10.6060/MHC190339p.
A. M. Lunev, Yu. A. Belousov, Russ. Chem. Bull., 2022, 71, 825; DOI: https://doi.org/10.1007/s11172-022-3485-3.
L. Dong, Y. Zang, D. Zhou, X. He, G. Chen, T. D. James, J. Li, Chem. Commun., 2015, 51, 11852; DOI: https://doi.org/10.1039/C5CC04357C.
Z. Xu, K. Baek, H. N. Kim, J. Cui, X. Qian, D. R. Spring, I. Shin, J. Yoon, J. Am. Chem. Soc., 2010, 132, 601; DOI: https://doi.org/10.1021/ja907334j.
X. Liu, N. Zhang, J. Zhou, T. Chang, C. Fang, D. Shangguan, Analyst, 2013, 138, 901; DOI: https://doi.org/10.1039/c2an36203a.
M. L. Zastrow, R. J. Radford, W. Chyan, C. T. Anderson, D. Y. Zhang, A. Loas, T. Tzounopoulos, S. J. Lippard, ACS Sens., 2016, 1, 32; DOI: https://doi.org/10.1021/acssensors.5b00022.
M. D. Pluth, E. Tomat, S. J. Lippard, Ann. Rev. Biochem., 2011, 80, 333; DOI: https://doi.org/10.1039/C7OB02235B.
K. Komatsu, K. Kikuchi, H. Kojima, Y. Urano, T. Nagano, J. Am. Chem. Soc., 2005, 127, 10197; DOI: https://doi.org/10.1021/ja050301e.
B. A. Wong, S. Friedle, S. J. Lippard, J. Am. Chem. Soc., 2009, 131, 7142; DOI: https://doi.org/10.1021/ja900980u.
S. Mizukami, S. Okada, S. Kimura, K. Kikuchi, Inorg. Chem., 2009, 48, 7630; DOI: https://doi.org/10.1021/ic900247r.
H. Wang, H. Wu, L. Xue, Y. Shia, X. Li, Org. Biomol. Chem., 2011, 9, 5436; DOI: https://doi.org/10.1039/c1ob05481c.
D. Liu, J. Qi, X. Liu, H. He, J. Chen, Inorg. Chem. Commun., 2014, 43, 173; DOI: https://doi.org/10.1016/j.inoche.2014.02.035.
H. Duan, Y. Ding, Ch. Huang, W. Zhu, R. Wang, Y. Xu, Chin. Chem. Lett., 2019, 30, 55; DOI: https://doi.org/10.1016/j.cclet.2018.03.016.
J. Wang, Y. Xiao, Z. Zhang, X. Qian, Y. Yanga, Q. Xu, J. Mater. Chem., 2005, 15, 2836; DOI: https://doi.org/10.1039/b500766f.
A. Pardo, J. M. L. Poyato, E. Martin, J. J. Camacho, D. Reyman, M. F. Braña, J. M. Castellano, J. Photochem. Photobiol. A, 1989, 46, 323; DOI: https://doi.org/10.1016/1010-6030(89)87049-2.
P. A. Panchenko, A. S. Polyakova, Yu. V. Fedorov, O. A. Fedorova, Makrogeterotsikly [Macroheterocycles], 2022, 15, 44 (in Russian); DOI: https://doi.org/10.6060/mhc214029p.
M. A. Zakharko, P. A. Panchenko, P. A. Ignatov, Yu. V. Fedorov, O. A. Fedorova, Mendeleev Commun., 2020, 30, 332; DOI: https://doi.org/10.1016/j.mencom.2020.05.024.
P. A. Panchenko, A. S. Polyakova, Yu. V. Fedorov, O. A. Fedorova, Mendeleev Commun., 2019, 29, 155; DOI: https://doi.org/10.1016/j.mencom.2019.03.012.
P. A. Panchenko, P. A. Ignatov, M. A. Zakharko, Yu. V. Fedorov, O. A. Fedorova, Mendeleev Commun., 2020, 30, 55; DOI: https://doi.org/10.1016/j.mencom.2020.01.018.
S. Nad, M. Kumbhakar, H. Pal, J. Phys. Chem. A., 2003, 107, 4808; DOI: https://doi.org/10.1021/JP021543T.
K. A. Connors, Binding Constants: the Mesurement of Molecular Complex Stability, John Wiley & Sons, New York, 1987.
M. T. Beck, I. Nagypál, Chemistry of Complex Equilibria, John Wiley & Sons, New York, 1990.
Q. Mei, Y. Shi, Q. Hua, B. Tong, RSC Adv., 2015, 5, 74924; DOI: https://doi.org/10.1039/C5RA09609J.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no competing interests.
Additional information
The study of the complexation of sensor 5 with metal cations by optical and NMR spectroscopy was carried out with the support of the Ministry of Science and Higher Education of the Russian Federation using the scientific equipment of the Center for Research of the Structure of Molecules, A. N. Nesmeyanov Institute of Organoelement Compounds, Russian Academy of Sciences.
This work was financially supported by the Russian Science Foundation (Project No. 20-73-10186).
No human or animal subjects were used in this research.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, Vol. 72, No. 9, pp. 2154–2161, September, 2023.
Rights and permissions
About this article
Cite this article
Pavlova, M.A., Panchenko, P.A., Vlasova, M.N. et al. Fluorescent sensor for Zn2+ cations based on a 4-methoxy-1,8-naphthalimide derivative containing a dipicolylamine receptor fragment. Russ Chem Bull 72, 2154–2161 (2023). https://doi.org/10.1007/s11172-023-4011-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-023-4011-y