Abstract
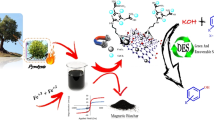
Inexpensive and recyclable catalysts, waste regeneration, that utilize safe and available solvents are important factors in laboratory and industrial applications. On the other hand, biochar nanoparticles are new precursors for the solid phase of carbon materials with novel characteristics, which exists synthetically or naturally. Therefore in this work, biochar nanoparticles were prepared from the pyrolysis of biomass resulting from chicken manure. Therefore, it is a suitable method for biomass waste recycling. Then, the surface of biochar nanoparticles was modified by (3-aminoopropyl)trimethoxysilane in n-hexane for 24 h at 60 °C, which the modified biochar nanoparticles were named nPr-NH2@biochar. Next, the modified biochar nanoparticles were functionalized by di(pyridin-2-yl)methanone (DPMI) in ethanol at 80 °C for 24 h, and the functionalized biochar nanoparticles were named as DPMI@biochar. Finally, a new copper Schiff-base complex of di(pyridin-2-yl)methanimine (DPMI) was immobilized on the surface of functionalized biochar nanoparticles (Cu-DPMI@biochar). This biochar-based catalyst nanostructure (Cu-DPMI@biochar) was used as a practical and reusable catalyst in the homoselective synthesis of 5-substituted-1H-tetrazole compounds by [3 + 2] cycloaddition reaction of sodium azide and nitrile derivatives. This catalyst was characterized by scanning electron microscopy (SEM), wavelength-dispersive X-ray spectroscopy (WDX), energy-dispersive X-ray spectroscopy (EDS), thermogravimetric analysis (TGA), X-ray powder diffraction (XRD), N2 adsorption/desorption isotherms, Fourier transform infrared spectroscopy (FTIR), and atomic absorption spectroscopy (AAS) techniques. Cu-DPMI@biochar can be recovered and reused several times.













Similar content being viewed by others
Data availability
Data are available in the main article file and supplementary material.
References
D. Wang, D. Astruc, Chem. Rev. 114, 6949 (2014)
V. Polshettiwar, R. Luque, A. Fihri, H. Zhu, M. Bouhrara, J.M. Basset, Chem. Rev. 111, 3036 (2011)
Y. Zhu, L.P. Stubbs, F. Ho, R. Liu, C.P. Ship, J.A. Maguire, N.S. Hosmane, ChemCatChem 2, 365 (2010)
M. Miceli, P. Frontera, A. Macario, A. Malara, Catalysts 11, 591 (2021)
D.J. Cole-Hamilton, Science 299, 1702 (2003)
V.S. Shende, V.B. Saptal, B.M. Bhanage, Chem. Rec. 19, 1 (2019)
J. Zhu, H. Li, L. Zhong, P. Xiao, X. Xu, X. Yang, Z. Zhao, J. Li, ACS Catal. 4, 2917 (2014)
C. Woo Lim, I.S. Lee, Nano Today 5, 412 (2010)
A. Vojvodic, J.K. Nørskov, Natl. Sci. Rev. 2, 140 (2015)
R. Schlögl, Angew. Chem. Int. 54, 3465 (2015)
A. Wang, J. Li, T. Zhang, Nat. Rev. Chem. 2, 65 (2018)
A. Jabbari, P. Moradi, M. Nikoorazm, J. Porous Mater. 30, 1395 (2023)
P. Moradi, RSC Adv. 12, 33459 (2022)
H. Keypour, J. Kouhdareh, S. Alavinia, K. Rabiei, M. Mohammadi, A. Maryamabadi, S. Babaei, J. Organomet. Chem. 989, 15 (2023)
B. Maleki, R. Sandaroos, S. Naderi, S. Peiman, J. Organomet. Chem. 990, 122666 (2023)
Z. Mohammadkhani, K. Rabiei, H. Keypour, J. Kouhdareh, I. Karakaya, J. Organomet. Chem. 999, 122822 (2023)
A. Ghorbani-Choghamarani, M. Nikoorazm, H. Goudarziafshar, B. Tahmasbi, Bull. Korean Chem. Soc. 30, 1388 (2009)
A.L. Corcho-Valdés, C. Iriarte-Mesa, J. Calzadilla-Maya, Y. Matos-Peralta, L.F. Desdín-García, M. Antuch, Carbon Compos. Catal. 223, 1 (2022)
T.T. Li, Y. Mei, H. Li, J. Qian, M. Wu, Y.Q. Zheng, Inorg. Chem. 59, 14184 (2020)
K. Biradha, A. Goswami, R. Moi, Chem. Commun. 56, 10824 (2020)
P. Moradi, M. Hajjami, RSC Adv. 11, 25867 (2021)
A.H. Chughtai, N. Ahmad, H.A. Younus, A. Laypkov, F. Verpoort, Chem. Soc. Rev. 44, 6804 (2015)
A. Jabbari, P. Moradi, M. Hajjami, B. Tahmasbi, Sci. Rep. 12, 11660 (2022)
P. Moradi, M. Hajjami, RSC Adv. 12, 13523 (2022)
P. Moradi, B. Zarei, Y. Abbasi Tyula, M. Nikoorazm (2023) Appl. Organomet. Chem. 37, e7020.
D.S. Khan, Appl. Organomet. Chem. 37, e7007 (2023)
H.S. Sobh, H.H. Mihsen, Baghdad Sci. J. 16(4), 886 (2019)
A.D. Mohsin, H.H. Mihsen, J. Inorg. Organomet. Polym. 30, 2172 (2020)
S.K. Abbas, Z.M. Hassan, H.H. Mihsen, M.T. Eesa, D.H. Attol, SILICON 12, 1103 (2020)
A.D. Mohsin, H.H. Mihsen, A.I.P. Conf, Proc. 2386, 030027 (2022)
M. Ghasemirad, M. Norouzi, P. Moradi, J. Nanopart. Res. 26, 14 (2024)
A. Jabbari, P. Moradi, B. Tahmasbi, RSC Adv. 13, 8890 (2023)
A. Jabbari, M. Nikoorazm, P. Moradi, Res. Chem. Intermed. 49, 1485 (2023)
M. Norouzi, P. Moradi, M. Khanmoradi, RSC Adv. 13, 35569 (2023)
X. Xiong, I.K.M. Yu, L. Cao, D.C.W. Tsang, S. Zhang, Y.S. Ok, Bioresour. Technol. 246, 254 (2017)
A. Kumar, K. Saini, T. Bhaskar, Bioresour. Technol. 299, 122564 (2020)
S.Z. Zhang, L.N. Dong, L.P. Mo, Z.H. Zhang, Appl. Organometal. Chem. 37, e6949 (2023)
S.Z. Zhang, Z.S. Cui, M. Zhang, Z.H. Zhang, Curr. Opin. Green Sustain. Chem. 38, 100713 (2022)
X. Li, Y. Jia, J. Zhang, Y. Qin, Y. Wu, M. Zhou, J. Sun, Chin. Chem. Lett. 33, 2105 (2022)
L.N. Dong, Y.M. Wang, W.L. Zhang, L.P. Mo, Z.H. Zhang, Appl. Organometal. Chem. 36, e6667 (2022)
L.N. Dong, S.Z. Zhang, W.L. Zhang, Y. Dong, L.P. Mo, Z.H. Zhang, Res. Chem. Intermed. 48, 1249 (2022)
P. Moradi, M. Hajjami, B. Tahmasbi, Polyhedron 175, 114169 (2020)
P. Moradi, M. Hajjami, F. Valizadeh-Kakhki, Appl Organometal Chem. 33, e5205 (2019)
S. Layek, B. Agrahari, S. Dey, R. Ganguly, D.D. Pathak, J. Organomet. Chem. 896, 194 (2019)
H. Sharghi, S. Ebrahimpourmoghaddam, M.M. Doroodmand, J. Organomet. Chem. 738, 41 (2013)
J.R. Bikas, N. Heydari, T. Lis, J. Mol. Struct. 1281, 135120 (2023)
F. Pirani, H. Eshghi, S.A. Rounaghi, RSC Adv. 13, 6160 (2023)
S. Molaei, N. Moeini, M. Ghadermazi, J. Organomet. Chem. 977, 122459 (2022)
L.V. Myznikov, S.V. Vorona, Y.E. Zevatskii, Chem. Heterocycl. 57, 224 (2021)
M.A. Vatmanidou, K. Stathopoulou, E. Gikas, N. Aligiannis, P.P. Dallas, D.M. Rekkas, Sci. Technol. 67, 102869 (2022)
J. Fenna, C. Chu, R. Hassan, T. Gomes, M. Tadrous, CMAJ Open 9, E1128 (2021)
A.K. Kabi, S. Sravani, R. Gujjarappa, A. Garg, N. Vodnala, U. Tyagi, D. Kaldhi, R. Velayutham, V. Singh, S. Gupta, C.C. Malakar, An overview on biological evaluation of tetrazole derivatives, in S. P. Swain, (eds) Nanostructured Biomaterials. (2022) 307–349. Materials Horizons: From Nature to Nanomaterials. Springer, Singapore
D. Kommula, P. Kumar Chintakunta, K. Garikapati, M.S.R. Murty, Mol. Divers. 27, 425 (2023)
J. Devasia, S. Chinnam, K. Khatana, S. Shakya, F. Joy, M. Rudrapal, A. Nizam, Polycycl. Aromat. Comp. 43, 1941 (2023)
V. Aureggi, G. Sedelmeier, Angew. Chem. Int. Ed. 46, 8440 (2007)
A. Najafi Chermahini, M. Azadi, E. Tafakori, A. Teimouri, M. Sabzalian, J. Porous Mater. 23, 441 (2016)
R. Kant, V. Singh, A. Agarwal, C. R. Chim. 19, 305 (2016)
P. Mani, A. Kumar Singh, S. Kumar Awasthi, Tetrahedron Lett. 55, 2014 (1879)
I.B. Seiple, Z. Zhang, P. Jakubec, A. Langlois-Mercier, P.M. Wright, D.T. Hog, K. Yabu, S. RaoAllu, T. Fukuzaki, P.N. Carlsen, Y. Kitamura, X. Zhou, M.L. Condakes, F.T. Szczypiński, W.D. Green, A.G. Myers, Nature 533, 338 (2016)
D.R. Patil, Y.B. Wagh, P.G. Ingole, K. Singh, D.S. Dalal, New J. Chem. 37, 3261 (2013)
R. Jahanshahi, B. Akhlaghinia, RSC Adv. 5, 104087 (2015)
A.M. Dehkhoda, N. Ellis, E. Gyenge, J. Appl. Electrochem. 44, 141 (2014)
Y. Liu, X. Zhao, J. Li, D. Ma, R. Han, Desalin. Water Treat. 46, 115 (2012)
T. Zhang, P. Li, C. Fang, R. Jiang, S. Wu, H. Nie, Adv. Mater. Res. 726–731, 1679 (2013)
X. Zheng, J. Wu, X. Yan, G. Qin, R. Zhou, Z. Wei, J. Soils Sediments 20, 3846 (2020)
M. Nikoorazm, P. Moradi, N. Noori, G. Azadi, J. Iran. Chem. Soc. 18, 467 (2021)
Q. Li, S.W. Zhang, Y. Zhang, C. Chen, Nanotechnology 17, 4981 (2006)
G. Zhang, B. Xu, H. Chong, W. Wei, C. Wang, G. Wang, RSC Adv. 9, 14016 (2019)
Y. Dong, K. Wang, Y. Tan, Q. Wang, J. Li, H. Mark, S. Zhang, Nanoscale Res. Lett. 13, 119 (2018)
T. Kikhavani, P. Moradi, M. Mashari-Karir, J. Naji, Appl. Organomet. Chem. 36, e6895 (2022)
Y. Abbasi Tyula, P. Moradi, M. Nikoorazm, ChemistrySelect 8, e202301674 (2023)
Y. Liu, G. Zhao, D. Wang, Y. Li, Natl. Sci. Rev. 2, 150 (2015)
C. Wen, A. Yin, W.L. Dai, Appl. Catal. B Environ. 160–161, 730 (2014)
B.K. Min, C.M. Friend, Chem. Rev. 107, 2709 (2007)
V. Polshettiwar, R.S. Varma, Green Chem. 12, 743 (2010)
P. Gupta, S. Paul, Catal. Today 236, 153 (2014)
H. Li, P.S. Bhadury, B. Song, S. Yang, RSC Adv. 2, 12525 (2012)
V. Rama, K. Kanagaraj, K. Pitchumani, J. Org. Chem. 76, 9090 (2011)
G. Aridoss, K.K. Laali, Eur. J. Org. Chem. 2011, 6343 (2011)
S. Kumar Prajapti, A. Nagarsenkar, B. Nagendra Babu, Tetrahedron Lett. 55, 3507 (2014)
G. Qi, W. Liu, Z. Bei, Chin. J. Chem. 29, 131 (2011)
L. Lang, H. Zhou, M. Xue, X. Wang, Z. Xu, Mater. Lett. 106, 443 (2013)
B. Sreedhar, A. SureshKumar, D. Yada, Tetrahedron Lett. 52, 3565 (2011)
S.M. Agawane, J.M. Nagarkar, Catal. Sci. Technol. 2, 1324 (2012)
P. Moradi, A. Ghorbani-Choghamarani, Appl. Organometal. Chem. 31, e3602 (2017)
M. Nikoorazm, P. Moradi, N. Noori, J. Porous Mater. 27, 1159 (2020)
M. Nikoorazm, A. Ghorbani-Choghamaranai, M. Khanmoradi, P. Moradi, J. Porous Mater. 25, 2018 (1831)
P. Moradi, T. Kikhavani, Y. Abbasi Tyula, Sci. Rep. 13, 5902 (2023)
M. Nikoorazm, B. Tahmasbi, S. Gholami, M. Khanmoradi, Y. AbbasiTyula, M. Darabi, M. Koolivand, Polyhedron 244, 116587 (2023)
Acknowledgements
The authors thank the research facilities of Dezful Branch, Islamic Azad University, Dezful, Iran, and also Ilam University, Ilam, Iran, for financial support of this research project.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. No funding was received.
Author information
Authors and Affiliations
Contributions
MA helped in data collection and methodology. SH supervised the study. BT was involved in supervision, writing—original draft, writing review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest and competing interests.
Ethical approval
Human and/or animal studies have not been conducted in this article. All authors give their consent for the publication of this article. This article is the only version submitted for “Research on Chemical Intermediates.” No version or part of this article is not published or not under review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alekasir, M., Heydarian, S. & Tahmasbi, B. The synthesis of biochar from biomass waste recycling and its surface modification for immobilization of a new Cu complex as a reusable nanocatalyst in the homoselective synthesis of tetrazoles. Res Chem Intermed 50, 2031–2049 (2024). https://doi.org/10.1007/s11164-024-05252-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-024-05252-2