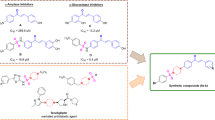
Abstract
In the current research work, a series of Co(II), Cu(II), Ni(II) and Zn(II) complexes of novel 5,5′-[(1-E,1′-E)-(4-nitro-1,2-phenylene)bis(azaneylylidene)bis(methaneylylidene) bis(2 methoxy phenol)] (NAMM) Schiff base ligand have been synthesized by the condensation reaction of 4-nitro-o-phenylenediammine with 3-hydroxy-4-methoxy-benzaldehyde. The characterization study of Schiff base ligand and its metal complexes is executed by various instrumental and spectral techniques such as 1H-nuclear magnetic resonance, 13C-nuclear magnetic resonance, infrared spectroscopy, mass analysis, ultraviolet–visible technique, elemental analysis, electron paramagnetic resonance, thermogravimetric analysis and powder X-ray diffraction techniques to know information about the structures of compounds. The spectral data reveal the hexadentate nature of synthesized metal complexes. To clarify the conductive and optical properties of complexes, direct optical energy band gap and Urbach spectral tail energy were estimated. The thermodynamic and kinetic parameters were calculated by Coats–Redfern method. In addition, the docking was carried out with two different proteins, COVID-19 main protease (PDB-6LU7) and human prostate specific antigen in a fab sandwich (PDB-3QUM) and the results revealed a large negative binding affinity value for zinc complex for both receptors, thereby exhibiting good protein–complex interactions. Further, the compounds were in silico screened for their biocompatibilities and pharmacokinetic behavior through ADMET and PASS studies which signifies the drug-like character of the synthesized compounds. Moreover, antimicrobial studies were evaluated against four different bacterial strains as well as two fungal strains and results evinced highest activity of copper complex (2) against E. coli (MIC-0.0055 μmol/mL) and C. albicans (MIC-0.0110 μmol/mL).













Similar content being viewed by others
Data availability
Data supporting the results of this study are available on fair request from the corresponding author. The data analyzed can be accessed from the manuscript for the current study.
References
S.K. Tadavi, A.A. Yadav, R.S. Bendre, J. Mol. Struct. (2017). https://doi.org/10.1016/j.molstruc.2017.09.112
A.S. Shekhawat, N.P. Singh, N.S. Chundawat, J. Sci. Res. (2022). https://doi.org/10.3329/jsr.v14i1.54814
B. Naureen, G.A. Miana, K. Shahid, M. Asghar, S. Tanveer, A. Sarwar, J. Mol. Struct. (2021). https://doi.org/10.1016/j.molstruc.2021.129946
A.A. Abu-Yamin, M.S. Abduh, S.A.M. Saghir, N. Al-Gabri, Pharmaceuticals 15, 454 (2022)
S. Nain, S. Asija, Y. Deswal, N. Kumar, P. Barwa, Indian J. Heterocycl. Chem. (2022) https://connectjournals.com/01951.2022.32.1
N. Kumar, S. Asija, Y. Deswal, S. Saroya, A. Kumar, Res. Chem. Intermed. (2022). https://doi.org/10.1007/s11164-022-04860-0
N. Kumar, Phosphorus Sulfur Silicon Relat. Elem. (2022). https://doi.org/10.1080/10426507.2022.2048386
S.S. Khaidir, H. Bahron, A.M. Tajuddin, K. Ramasamy, J. Sustain. Sci. Manag. (2022). https://doi.org/10.46754/jssm.2022.4.003
A. Singh, H.P. Gogoi, P. Barman, A.K. Guha, Appl. Organomet. Chem. (2022). https://doi.org/10.1002/aoc.6673
M.A. Diab, A.Z. El-Sonbati, S.M. Morgan, M.A. El-Mogazy, Appl. Organomet. Chem. (2018). https://doi.org/10.1002/aoc.4378
E. Sonbati, W.H. Mahmoud, G.G. Mohamed, M.A. Diab, S.M. Morgan, S.Y. Abbas, Appl. Organomet. Chem. (2019). https://doi.org/10.1002/aoc.5048
M. Kaute, E.M. Ngandung, F.A.N. Kamga, A.G. Paboudam, C.A. Mariam, C.N. Pecheu, T.K. Ignas, P.T. Ndifon, Egypt. J. Chem. (2022). https://doi.org/10.21608/EJCHEM.2022.82313.4087
K. Andiappan, A. Sanmugam, E. Deivanayagam, K. Karuppasamy, H.S. Kim, D. Vikraman, Int. J. Biol. Macromol. (2019). https://doi.org/10.1016/j.ijbiomac.2018.11.251
K. Mounika, J. Sci. Res. (2010). https://doi.org/10.3329/jsr.v2i3.4899
K. Subin, K.K. Aravindakshan, Res. Chem. (2021). https://doi.org/10.1016/j.rechem.2021.100129
C. Abate, F. Carnamucio, O. Giuffre, C. Foti, Biomolecules (2022). https://doi.org/10.3390/biom12070933
H.A. Ward, T.M. Musa, Z.N. Nasif, Al-Mustan. J. Sci. (2022). https://doi.org/10.23851/mjs.v33i2.1121
A. Singh, A. Chaudhary, Bioinorg. Chem. Appl. (2018). https://doi.org/10.1155/2018/2467463
P. Ghorai, R. Saha, S. Bhuiya, S. Das, P. Brandao, D. Ghosh, T. Bhaumik, P. Bandyopadhyay, D. Chattopadhyay, A. Saha, Polyhedron (2018). https://doi.org/10.1016/j.poly.2017.11.041
K. Kar, D. Ghosh, B. Kabi, A. Chandra, Polyhedron (2022). https://doi.org/10.1016/j.poly.2022.115890
R.S. Joseyphus, M.S. Nair, Mycobio (2008). https://doi.org/10.4489/MYCO.2008.36.2.093
S. Mahato, N. Meheta, K. Muddukrishnaiah, M. Joshi, M. Shit, A. Roy Choudhury, B. Biswas, Polyhedron (2020). https://doi.org/10.1016/j.poly.2020.114933
R. Atkins, G. Brewer, E. Kokot, G.M. Mockler, E. Sinn, Inorg. Chem. (1985). https://doi.org/10.1021/ic00196a003
H. Kargar, A.A. Ardakani, M.N. Tahir, M. Ashfaq, K.S. Munawar, J. Mol. Struct. (2021). https://doi.org/10.1016/j.molstruc.2021.130112
C. Shiju, D. Arish, S. Kumaresan, J. Mol. Struct. 1221, 128770 (2020)
N. Ahmad, M. Alam, R. Wahab, M. Ahmed, A. Ahmad, Open Chem. (2020). https://doi.org/10.1515/chem-2020-0168
H.A.E. Boraey, A.A.S. El-Din, Spectrochim. Acta (2014). https://doi.org/10.1016/j.saa.2014.05.018
M. Pervaiz, S. Sadiq, A. Sadiq, U. Younas, A. Ashraf, Z. Saeed, M. Zuber, A. Adnan, Coord. Chem. Rev. (2021). https://doi.org/10.1016/j.ccr.2021.214128
A.M.A. Alagha, Y.A. Ammar, H.A. Bayoumi, S.A. Aldhlmani, J. Mol. Struct. (2014). https://doi.org/10.1016/j.molstruc.2014.05.078
L. Rahman, M.S. Adam, N. Al-Zaqri, M.R. Shehata, H. El-Sayed Ahmed, S.K. Mohamed, Arab. J. Chem. (2022). https://doi.org/10.1016/j.arabjc.2022.103737
S. Basaleh, H.B. Howsaui, A.A. Sharfalddin, M.A. Hussien, Res. Chem. (2022). https://doi.org/10.1016/j.rechem.2022.100445
J. Saranya, S.J. Kirubabathy, S. Chitra, A. Zarrouk, K. Kalpana, K. Lavanya, B. Ravikiran, Arab. J. Sci. Eng. (2020). https://doi.org/10.1007/s13369-020-04416-7
H. Howsaui, A.A. Sharfalddin, M.H. Abdellattif, A.S. Basaleh, M.A. Hussien, Appl. Sci. (2021). https://doi.org/10.3390/app11199067
M. Chaudhary, Res. Equ. (2022). https://doi.org/10.21203/rs.3.rs-880545
V. Rangaswamy, S. Renuka, I. Venda, Mater. Today Proc. (2022). https://doi.org/10.1016/j.matpr.2021.10.360
A.Z. El-Sonbati, N.F. Omar, M.I. Abou-Dobara, M.A. Diab, M.A. El-Mogazy, Sh.M. Morgan, M.A. Hussien, A.A. El-Ghettany, J. Mol. Struct. (2021). https://doi.org/10.1016/j.molstruc.2021.130481
G.G. Mohamed, M.M. Omar, Y.M. Ahmed, J. Inorg. Gen. Chem. (2021). https://doi.org/10.1002/zaac.202100245
O.E. Gammal, F.S. Mohamed, G.N. Rezk, A.A. El-Bindary, J. Mol. Liq. (2021). https://doi.org/10.1016/j.molliq.2020.115223
T.A. Alorini, A.N. Al-Hakimi, S. El-S, E.H.L. Saeed, A.E.A.E.A. Alhamzi, Arab. J. Chem. (2022). https://doi.org/10.1016/j.arabjc.2021.103559
R. Shah, H. Katouah, A.A. Sedayo, M. Abualnaja, M.M. Aljohani, F. Saad, R. Zaky, N.M. El-Metwaly, J. Mol. Liq. (2020). https://doi.org/10.1016/j.molliq.2020.114116
M.S. Nair, D. Arish, R.S. Joseyphus, J. Saudi Chem. Soc. (2012). https://doi.org/10.1016/j.jscs.2010.11.002
A. Hamil, K.M. Khalifa, A.A. Almutaleb, M.Q. Nouradean, Adv. J. Chem. Sec. A (2020). https://doi.org/10.33945/SAMI/AJCA.2020.4.13
A.M.A. Adam, A. Alhadhrami, H.A. Saad, M.S. Refat, Appl. Organomet. Chem. (2017). https://doi.org/10.1002/aoc.3950
M. Manjunath, A.D. Kulkarni, G.B. Bagihalli, S. Malladi, S.A. Patil, J. Mol. Struct. (2017). https://doi.org/10.1016/j.molstruc.2016.07.123
K. Adilee, S.R. Hasan, IOP Conf. Ser. Earth Environ. Sci. (2021). https://doi.org/10.1088/1755-1315/790/1/012031
M. Kareem, PJMHS (2022). https://doi.org/10.53350/pjmhs22165655
T.J. Saritha, P. Metilda, J. Saudi Chem. Soc. (2021). https://doi.org/10.1016/j.jscs.2021.101245
A. Isiyaku, I.T. Siraj, A.U. Maigari, B. Magaji, Dutse J. Pure Appl. Sci. (2021). https://doi.org/10.4314/dujopas.v7i4a
H.M. Vinusha, S.P. Kollur, H.D. Revanasiddappa, R. Ramu, P.S. Shirahatti, M.N.N. Prasad, S. Chandrashekar, M. Begum, Res. Chem. (2019). https://doi.org/10.1016/j.rechem.2019.100012
V.P. Radha, S. Chitra, S. Jonekirubavathi, M. Chung, S. Kim, M. Prabakaran, J. Coord. Chem. (2020). https://doi.org/10.1080/00958972.2020.1752372
N.H. Mahmoud, G.H. Elsayed, A. Aboelnaga, A.M. Fahim, Appl. Organomet. Chem. (2022). https://doi.org/10.1002/aoc.6697
F.K. Ommenya, E.A. Nyawade, D.M. Andala, J. Kinyua, J. Chem. Hindawi (2020). https://doi.org/10.1155/2020/1745236
M. Bingol, N. Turan, J. Mol. Struct. (2020). https://doi.org/10.1016/j.molstruc.2019.127542
H.A. Kiwaan, T.M. Atwee, E.A. Azab, A.A. El-Bindary, J. Chin. Chem. Soc. (2019). https://doi.org/10.1002/jccs.201800092
F. Urbach, Phys. Rev. (1953). https://doi.org/10.1103/PhysRev.92.1324
V. Sangwan, D.P. Singh, J. Chin. Chem. Soc. (2019). https://doi.org/10.1002/jccs.201900290
A. Alothman, Z.M. Almarhoon, J. Mol. Struct. (2020). https://doi.org/10.1016/j.molstruc.2020.127704
V. Sangwan, D.P. Singh, Viet. J. Chem. (2021). https://doi.org/10.1002/vjch.201900122
H.H. Horowitz, G. Metzger, Anal. Chem. (1963). https://doi.org/10.1021/ac60203a013
A.Z. El-Sonbati, M.A. El-Mogazy, S.G. Nozha, M.A. Diab, M.I. Abou-Dobara, A.M. Eldesoky, S.M. Morgan, J. Mol. Struct. (2022). https://doi.org/10.1016/j.molstruc.2021.131498
A.Z. El-Sonbati, M.A. Diab, A.M. Eldesoky, S.M. Morgan, O.L. Salem, Appl. Organomet. Chem. (2019). https://doi.org/10.1002/aoc.4839
A.Z. El-Sonbati, M.A. Diab, S.M. Morgan, H.A. Seyam, J. Mol. Struct. (2018). https://doi.org/10.1016/j.molstruc.2017.10.020
P. Jeyaraman, A. Alagarraj, R. Natarajan, J. Biomol. Struct. Dyn. (2019). https://doi.org/10.1080/07391102.2019.1581090
A.A. Lagunin, V.I. Dubovskaja, A.V. Rudik, P.V. Pogodin, D.S. Druzhilovskiy, T.A. Gloriozova, D.A. Filimonov, G.N. Sastry, V.V. Poroikov, PLoS ONE (2018). https://doi.org/10.1371/journal.pone.0191838
C.A. Lipinski, F. Lombardo, B.W. Dominy, P.J. Feeney, Adv. Drug Deliv. Rev. (2001). https://doi.org/10.1016/s0169-409x(00)00129-0
S. Michael, P. Jeyaraman, B. Marimuthu, R. Rajasekar, R. Thanasamy, K. Kumar, N. Raman, J. Mol. Struct. (2023). https://doi.org/10.1016/j.molstruc.2023.134987
S. Hosny, M.S. Ragab, R.F.A. El-Baki, Sci. Rep. (2023). https://doi.org/10.1038/s41598-023-28402-9
S.G. Nozha, S.M. Morgan, S.E.A. Ahmed, M.A. El-Mogazy, M.A. Diab, A.Z. El-Sonbati, M.I. Abou-Dobara, J. Mol. Struct. (2021). https://doi.org/10.1016/j.molstruc.2020.129525
G.G. Mohamed, A.A. El-Sherif, M.A. Saad, S.E.A. El-Sawy, S.M. Morgan, J. Mol. Liq. (2016). https://doi.org/10.1016/j.molliq.2016.09.065
O. Trott, A.J. Olson, J. Comput. Chem. (2010). https://doi.org/10.1002/jcc.21334
M.A. Diab, S.G. Nozha, A.Z. El-Sonbati, M.A. El-Mogazy, S.M. Morgan, Appl. Organomet. Chem. (2019). https://doi.org/10.1002/aoc.5153
A.Z. El-Sonbati, M.A. Diab, S.M. Morgan, M.I. Abou-Dobara, A.A. El-Ghettany, J. Mol. Struct. (2020). https://doi.org/10.1016/j.molstruc.2019.127065
S.M. Morgan, M.A. Diab, A.Z. El-Sonbati, Appl. Organomet. Chem. (2018). https://doi.org/10.1002/aoc.4504
M.I. Abou-Dobara, N.F. Omar, A.Z. El-Sonbati, S.M. Morgan, O.L. Salem, A.M. Eldesoky, Mater. Sci. Eng. C (2019). https://doi.org/10.1016/j.msec.2019.05.012
M. Aljohani, Bull. Chem. Soc. Ethiop. (2023). https://doi.org/10.4314/bcse.v37i1.8
L. Ghasemi, M.H. Esfahani, A. Abbasi, M. Behzad, Polyhedron (2022). https://doi.org/10.1016/j.poly.2022.115825
M. Gulcan, M. Sonmez, I. Berber, Turk. J. Chem. (2012). https://doi.org/10.3906/kim-1104-2
M. Munjal, J. Pharmacog. Phytochem. (2018). https://doi.org/10.22271/phyto
S.A. Khan, M. Kumar, S. Saxena, Orient. J. Chem. 27, 3 (2011)
K.R.S. Gowda, H.S.B. Naik, B.V. Kumar, C.N. Sudhamani, H.V. Sudeep, T.R.R. Naik, G. Krishnamurthy, Spectrochim. Acta (2013). https://doi.org/10.1016/j.saa.2012.12.011
L.P. Nitha, R. Aswathy, N.E. Mathews, B.S. Kumari, K. Mohanan, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. (2014). https://doi.org/10.1016/j.saa.2013.08.075
Acknowledgements
The authors highly acknowledge SAIF, IIT Bombay, for providing (ESR JEOL) analytical facility.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
IS was involved in data curation, writing—original draft preparation, investigation, visualization. AS helped in conceptualization, methodology, project administration, validation, writing—review and editing, supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sindhu, I., Singh, A. Experimental and theoretical insight of Schiff base transition metal complexes: synthesis, characterization, antimicrobial and COVID-19 molecular docking studies. Res Chem Intermed 50, 413–436 (2024). https://doi.org/10.1007/s11164-023-05179-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05179-0