Abstract
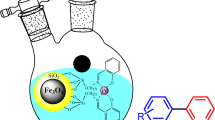
In this work, the mesoporous magnetic heterogeneous catalyst was synthesized by anchoring copper (II) onto the nitrogen-rich poly (melamine) Schiff base networks magnetic nanocomposite. First, MNP (Fe3O4@SiO2) was functionalized with chlorosilane (CPTS). Then, the chlorine of these functionalized magnetic nanoparticles could react with the –NH2 groups of the melamine through a nucleophilic substitution reaction. Simultaneously, the condensation polymerization of melamine and benzene-1, 3, 5-tricarbaldehyde was carried out in the presence of surface-modified magnetic nanoparticles. Later, Fourier-transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), thermogravimetric analysis (TGA), transmission electron microscopy (TEM), scanning electron microscopy (SEM), atomic absorption spectroscopy (AAS), Brunauer–Emmett–Teller surface analysis (BET), and energy dispersive analysis were deployed to characterize the catalyst. The results of the AAS measurement showed that the copper ion loading on the mesoporous nitrogen-rich supported catalyst was about 0.82 mmol g−1. The synthesized nanocomposite was used as a catalyst and shows excellent activity in the three-component coupling reaction of amines, carbon disulfide, and boronic acids. In the presence of 70 mg of the catalyst, a wide range of functionalized dithiocarbamates were synthesized leading to excellent yields. Moreover, this MNP@SNWMe@Cu catalyst was easily recovered and reused seven times without significant loss of its activity. The results of this research exhibited that the synthesized catalyst functioned with high catalytic activity in this reaction.











Similar content being viewed by others
Availability of data and materials
Not applicable.
References
M. Stasevych, V. Zvarych, V. Lunin, N.G. Deniz, Z. Gokmen, O. Akgun, E. Ulukaya, V. Poroikov, T. Gloriozova, V. Novikov, SAR QSAR Environ Res 28, 355 (2017)
R.D. Li, H.L. Wang, Y.B. Li, Z.Q. Wang, X. Wang, Y.T. Wang, Z.M. Ge, R.T. Li, Eur. J. Med. Chem. 93, 381 (2015)
M. Wang, X. Song, N. Ma, Catal. Lett. 144, 1233 (2014)
A.R. Sardarian, I. Dindarloo Inaloo, M. Zangiabadi, New J. Chem. 43, 8557 (2019)
R.K. Vishwakarma, S. Kumar, K.N. Singh, Org. Lett. 23, 4147 (2021)
A.R. Sardarian, I.D. Inaloo, A.R. Modarresi-Alam, Mol. Divers. 22, 863 (2018)
A.R. Modarresi-Alam, I.D. Inaloo, E. Kleinpeter, J. Mol. Struct. 1024, 156 (2012)
G. Li, Q. Yan, Z. Gan, Q. Li, X. Dou, D. Yang, Org. Lett. 21, 7938 (2019)
J.O. Adeyemi, G.M. Saibu, L.O. Olasunkanmi, A.O. Fadaka, M. Meyer, N.R.S. Sibuyi, D.C. Onwudiwe, A.O. Oyedeji, Heliyon 7, e07693 (2021)
D. Chaturvedi, S. Ray, Tetrahedron Lett. 47, 1307 (2006)
D. Chaturvedi, S. Ray, Tetrahedron Lett. 48, 149 (2007)
I.D. Inaloo, S. Majnooni, Chem Select 3, 4095 (2018)
G. Hogarth, Mini Rev. Med. Chem. 12, 1202 (2012)
N. Bagherzadeh, A.R. Sardarian, I.D. Inaloo, New J. Chem. 45, 11852 (2021)
A. Pourjavadi, M. Kohestanian, M. Yaghoubi, New J. Chem. 43, 18647 (2019)
A. Pourjavadi, S. Rahemipoor, M. Kohestanian, Compos. Sci. Technol. 188, 107951 (2020)
C. Qi, T. Guo, W. Xiong, Synlett 27, 2626 (2016)
R. Turgis, G. Arrachart, C. Delchet, C. Rey, Y. Barré, S. Pellet-Rostaing, Y. Guari, J. Larionova, A. Grandjean, Chem. Mater. 25, 4447 (2013)
S.D. Dhengale, V.M. Naik, G.B. Kolekar, C.V. Rode, P.V. Anbhule, Res. Chem. Intermed. 47, 3263 (2021)
T. Chowdhury, S. Chatterjee, P. Banerjee, D. Sukul, M. Shukla, T. Chattopadhyay, J. Coord. Chem. 73, 754 (2020)
M.J. Neufeld, J.L. Harding, M.M. Reynolds, A.C.S. Appl, Mater. Interfaces 7, 26742 (2015)
V.C. Niculescu, G. Paun, V. Parvulescu, Appl. Organomet. Chem. 32, 2 (2018)
Y. Su, Y. Zhang, C. Li, G. Xu, J. Bai, Catal. Lett. 150, 3196 (2020)
S. Sanaei-Rad, H. Saeidiroshan, B. Mirhosseini-Eshkevari, M.A. Ghasemzadeh, Res. Chem. Intermed. 47, 2143 (2021)
A. Pourjavadi, M. Kohestanian, N. Keshavarzi, Appl. Organomet. Chem. 34, 1 (2020)
A. Pourjavadi, N. Keshavarzi, S.H. Hosseini, F.M. Moghaddam, Ind. Eng. Chem. Res. 57, 12314 (2018)
J. Govan, Y.K. Gun’ko, Nanomaterials 4, 222 (2014)
A. Pourjavadi, N. Keshavarzi, F.M. Moghaddam, S.H. Hosseini, Appl. Organomet. Chem. 32, 1 (2018)
W. Sun, W. Jiang, G. Zhu, Y. Li, J. Organomet. Chem. 873, 91 (2018)
M. Qi, K. Zhang, S. Li, J. Wu, C. Pham-Huy, X. Diao, D. Xiao, H. He, New J. Chem. 40, 4480 (2016)
Y. Wang, Y. Wang, L. Yu, J. Wang, B. Du, X. Zhang, Chem. Eng. J. 368, 115 (2019)
A. Mouradzadegun, S. Elahi, F. Abadast, RSC Adv. 4, 31239 (2014)
R. Tamura, T. Kawata, Y. Hattori, N. Kobayashi, M. Kimura, Macromolecules 50, 7978 (2017)
P.A. Pourjavadi, N. Keshavarzi, F.M. Moghaddam, S.H. Hosseini, Chem. Sel. 3, 2716 (2018)
V. Kandathil, B.D. Fahlman, B.S. Sasidhar, S.A. Patil, S.A. Patil, New J. Chem. 41, 9531 (2017)
S. Omar, B. Dutta, S. Natour, R. Abu-Reziq, J. Organomet. Chem. 818, 48 (2016)
M.A. Barakat, R. Kumar, E.C. Lima, M.K. Seliem, J. Taiwan Inst. Chem. Eng. 119, 146 (2021)
H. Veisi, T. Tamoradi, A. Rashtiani, S. Hemmati, B. Karmakar, J. Ind. Eng. Chem. 90, 379 (2020)
L. Shiri, L. Heidari, M. Kazemi, Appl. Organomet. Chem. 32, 1 (2018)
C.I. Fernandes, M.D. Carvalho, L.P. Ferreira, C.D. Nunes, P.D. Vaz, J. Organomet. Chem. 760, 2 (2014)
I. Sarvi, M. Gholizadeh, M. Izadyar, Catal. Lett. 147, 1162 (2017)
G. Kang, J. Choi, T. Park, Sci. Rep. 6, 1 (2016)
Q. Li, X. Tian, N. Wu, Y. Li, T. Pan, B. Zhang, Y. Duan, S. Wang, Y. Li, J. Appl. Polym. Sci. 138, 1 (2021)
A. Pourjavadi, M. Nazari, M. Kohestanian, S.H. Hosseini, Colloid Polym. Sci. 297, 917 (2019)
M. Akkoç, N. Buğday, S. Altın, N. Kiraz, S. Yaşar, İ Özdemir, J. Organomet. Chem. 943, 121823 (2021)
B. Zakerinasab, M.A. Nasseri, H. Hassani, M.M. Samieadel, Res. Chem. Intermed. 42, 3169 (2016)
A. Radoń, A. Drygała, Ł Hawełek, D. Łukowiec, Mater. Charact. 131, 148 (2017)
G. Liu, Q. Chen, E. Oyunkhand, S. Ding, N. Yamane, G. Yang, Y. Yoneyama, N. Tsubaki, Carbon N. Y. 130, 304 (2018)
M.K. Bhunia, S.K. Das, P. Pachfule, R. Banerjee, A. Bhaumik, Dalt. Trans. 41, 1304 (2012)
J. Liu, X. Zhang, B. Wen, Y. Li, J. Wu, Z. Wang, T. Wu, R. Zhao, S. Yang, Catal. Sci. Technol. 11, 3119 (2021)
D. Liu, X. Wang, Y. Chen, S. Yuan, Sci. China Chem. 59, 975 (2016)
A. Pourjavadi, M. Kohestanian, C. Streb, Mater. Sci. Eng. C 108, 110418 (2020)
M. Kohestanian, H. Bouhendi, M. Ghiass, J. Polym. Res. 24, 1 (2017)
J. Grabska, J. Badzoka, C.W. Huck, K.B. Bec, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 262, 120085 (2021)
Y. Shi, X. Zhang, H. Liu, J. Han, Z. Yang, L. Gu, Z. Tang 2001998, 1 (2020)
K. Un, K. Un, T. Ft-ir, and T. Xrd, 8 (2015).
P. Puthiaraj, K. Pitchumani, Chem. A Eur. J. 20, 8761 (2014)
F. Siadatnasab, S. Farhadi, M. Dusek, V. Eigner, A.A. Hoseini, A. Khataee, Ultrason. Sonochem. 64, 104727 (2020)
U. Kalsoom, A. Peristyy, P.N. Nesterenko, B. Paull, RSC Adv. 6, 38140 (2016)
D. Schwarz, Y. Noda, J. Klouda, K. Schwarzová-Pecková, J. Tarábek, J. Rybáček, J. Janoušek, F. Simon, M.V. Opanasenko, J. Čejka, A. Acharjya, J. Schmidt, S. Selve, V. Reiter-Scherer, N. Severin, J.P. Rabe, P. Ecorchard, J. He, M. Polozij, P. Nachtigall, M.J. Bojdys, Adv. Mater. 29, 1 (2017)
Y. Chen, W. Bi, L. Chen, Q. Liu, Int. J. Hydrogen Energy 46, 27567 (2021)
M. Sadeghinia, M. Rezaei, A. Nemati Kharat, M. Namayandeh Jorabchi, B. Nematollahi, F. Zareiekordshouli, Mol. Catal. 484, 110776 (2020)
B. Ghods, M. Rezaei, F. Meshkani, Ceram. Int. 42, 6883 (2016)
L.D.S. Yadav, R. Patel, V.P. Srivastava, Tetrahedron Lett. 50, 1335 (2009)
S. Bhadra, A. Saha, B.C. Ranu, Green Chem. 10, 1224 (2008)
F. Nemati, A. Elhampour, S. Zulfaghari, Phosphorus Sulfur Silicon Relat. Elem. 190, 1692 (2015)
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Prof. AP: project administration, supervision, resources, review & editing, funding acquisition, and validation. NK and MK: conceptualization, investigation, methodology, validation, formal analysis, writing-original draft, and funding acquisition. AS: methodology, validation, and formal analysis.
Corresponding authors
Ethics declarations
Ethical approval
We confirm that this manuscript is original, has not been published elsewhere and is not under consideration by another journal.
Consent to participate
We confirm that this manuscript is original, has not been published elsewhere and is not under consideration by another journal.
Consent for publication
All authors have approved the manuscript and agree to submit it to the Research on Chemical Intermediates journal.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Keshavarzi, N., Kohestanian, M., Sabzevari, A. et al. Three-component coupling of boronic acids, amines, and carbon disulfide by a heterogeneous catalyst of copper (II) embedded in a highly porous and magnetic Schiff base network of melamine. Res Chem Intermed 49, 4423–4441 (2023). https://doi.org/10.1007/s11164-023-05081-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05081-9