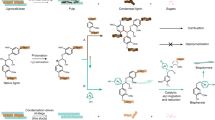
Abstract
1,8-Diazabicyclo [5.4.0] undec-7-ene functionalized cellulose nanofibers (CNF@DBU[Cl]) were simply prepared and characterized by analytical techniques. Scanning electron microscope confirmed that CNF@DBU[Cl] was formed with average size of 30–60 nm. X-ray diffraction of CNF@DBU[Cl] showed that the crystalline structure of the cellulose nanofibers was remained unchanged after functionalization. According to elemental analyses and thermal gravimetric analysis, the loading amount of organic group on cellulose nanofibers was found to be 1.46 mmol/g. The catalytic activity of DBU functionalized cellulose nanofibers was studied for the synthesis of biologically important tetraketone derivatives in the reaction of aldehydes with 1,3-dicarbonyl compounds. In the presence of CNF@DBU[Cl], the corresponding tetraketones were obtained in 80–96% yields under very mild reaction conditions. The catalyst was recovered and reused four successive runs without significant loss of catalytic activity. This method showed several significant advantages including short reaction times, high yields of products, use of various substrates, convenient work-up, no necessity of extraction or chromatographic purification steps, environmentally friendly conditions and lack of use of any harmful solvent.
Graphic abstract








Similar content being viewed by others
References
Q. Du, Y. Li, Beilstein J. Org. Chem. 7, 378 (2011)
W. Yu, L. Jiang, C. Shen, W. Xu, P. Zhang, Catal. Commun. 79, 11 (2016)
S. Hajar, MdS Islam, MdL Rahman, S.S. Rashid, Z.Z. Chowdhury, MdE Ali, S.M. Sarkar, Bioressources 12, 882 (2017)
MdS Islam, MdL Rahman, M.M. Yusoff, S.M. Sarkar, J. Clean. Prod. 149, 1045 (2017)
D. Li, J. Zhang, C. Cai, J. Org. Chem. 83, 7534 (2018)
S. Shojaei, Z. Ghasemi, A. Shahrisa, Appl. Organomet. Chem. 31, e3788 (2017)
P. Kaur, B. Kumar, V. Kumar, R. Kumar, Tetrahedron Lett. 59, 1986 (2018)
H.P.S. Abdul Khalil, C.K. Saurabha, A.S. Adnan, M.R. Nurul Fazitaa, M.I. Syakira, Y. Davoudpour, M. Rafatullah, C.K. Abdullah, M.K.M. Haafiz, R. Dungani, Carbohydr. Polym. 150, 216 (2016)
T. Baran, A. Mentes, J. Organomet. Chem. 803, 30 (2016)
S. Keshipour, S.S. Mirmasoudi, Adv. Environ. Technol. 3, 59 (2017)
A. Matsuoka, T. Isogawa, Y. Morioka, B.R. Knappett, A.E.H. Wheatley, S. Saito, H. Naka, RSC Adv. 5, 12152 (2015)
Y. Huang, Y. Fang, L. Chen, A. Lu, L. Zhang, Chem. Eng. J. 315, 573 (2017)
Y. Cao, M. Tang, M. Li, J. Deng, F. Xu, L. Xie, Y. Wang, ACS Sustain. Chem. Eng. 5, 9894 (2017)
K. Murali, M.S. Neelakandan, S. Thomas, JSM Nanotechnol. Nanomed 6(1), 1064 (2018)
L. Dykman, N. Khlebtsov, Chem. Soc. Rev. 41, 2256 (2012)
L. Chen, E. Hwang, J. Zhang, Sensors 18, 1440 (2018)
MdR Awual, Chem. Eng. J. 307, 456 (2017)
MdR Awual, G.E. Eldesoky, T. Yaita, M. Naushad, H. Shiwaku, Z.A. Alothman, S. Suzuki, Chem. Eng. J. 279, 639 (2015)
A. Shahat, MdR Awual, M. Naushad, Chem. Eng. J. 271, 155 (2015)
MdR Awual, Chem. Eng. J. 303, 539 (2016)
MdR Awual, Chem. Eng. J. 300, 264 (2016)
MdR Awual, T. Yaita, S. Suzuki, H. Shiwaku, J. Hazard. Mater. 291, 111 (2015)
MdR Awual, T. Yaita, H. Shiwaku, Chem. Eng. J. 228, 327 (2013)
MdR Awual, T. Kobayashi, Y. Miyazaki, R. Motokawa, H. Shiwaku, S. Suzuki, Y. Okamoto, T. Yaita, J. Hazard. Mater. 252–253, 313 (2013)
MdR Awual, MdM Hasan, MdA Khaleque, MdC Sheikh, Chem. Eng. J. 288, 368 (2016)
MdR Awual, Chem. Eng. J. 289, 65 (2016)
MdR Awual, Chem. Eng. J. 307, 85 (2017)
S. Duan, Zh Du, H. Fan, R. Wang, Nanomaterials 8, 949 (2018)
M. Kaushik, A. Moores, Green Chem. 18, 622 (2016)
T. Nypelö, H. Pynnönen, M. Österberg, J. Paltakari, J. Laine, Cellulose 19, 779 (2012)
T. Saito, A. Isogai, Carbohydr. Polym. 61, 183 (2005)
K. Yoon, B.S. Hsiao, B. Chu, J. Mater. Chem. 18, 5326 (2008)
I. Siro, D. Plackett, Cellulose 17, 459 (2010)
H. Takagi, A. Asano, Compos. Part A Appl. Sci. Manuf. 39, 685 (2008)
A. Isogai, T. Saito, H. Fukuzumi, Nanoscale 3, 71 (2011)
S. Iwamoto, A. Isogai, T. Iwata, Biomacromol 12, 831 (2011)
Z. Ma, M. Kotaki, S. Ramakrishna, J. Membrane Sci. 265, 115 (2005)
W. Chen, H. Yu, Y. Liu, Y. Hai, M. Zhang, P. Chen, Cellulose 18, 433 (2011)
D. Klemm, T. Heinze, B. Philipp, W. Wagenknecht, Acta Polym. 48, 277 (1997)
D. Roy, M. Semsarilar, J.T. Guthrie, S. Perrier, Chem. Soc. Rev. 38, 2046 (2009)
K.R. Reddy, N.S. Kumar, P.S. Reddy, B. Sreedhar, M.L. Kantam, J. Mol. Catal. A: Chem. 252, 12 (2006)
C. Li, J. Wang, Zh Yang, Zh Hu, Z. Lei, Catal. Commun. 8, 1202 (2007)
G.M. Maharvi, S. Ali, N. Riaz, N. Afza, A. Malik, M. Ashraf, L. Iqbal, M. Lateef, J. Enzyme Inhib. Med. Chem. 23, 62 (2008)
K.M. Khan, G.M. Maharvi, M.T.H. Khan, A. Jabbar Shaikh, S. Perveen, S. Begum, M.I. Choudhary, Bioorg. Med. Chem. 14, 344 (2006)
M. Saha, A.K. Pal, Tetrahedron Lett. 52, 4872 (2011)
M. Brito-Arias, M. Tapia-Albarran, I. Padilla-Martınez, F. Martınez-Martınez, G. Espinosa, E. Molins, E. Espinosa, J. Chem. Crystallogr. 29, 759 (1999)
A. Fallah, M. Tajbakhsh, H. Vahedi, A. Bekhradnia, Res. Chem. Intermed. 43, 29 (2017)
T.S. Jin, A.Q. Wang, H. Ma, J. Zhang, T. Li, Indian J. Chem. B 45, 470 (2006)
A. Pramanik, S. Bhar, Catal. Commun. 20, 17 (2012)
V.K. Rao, M.M. Kumar, A. Kumar, Indian J. Chem. Sec. B 50, 1128 (2011)
S. Kantevari, R. Bantu, L. Nagarapu, J. Mol. Catal. A: Chem. 269, 53 (2007)
S. Ray, A. Bhaumik, M. Pramanik, R.J. Butcher, S.O. Yildirim, C. Mukhopadhyay, Catal. Commun. 43, 173 (2014)
M. Gupta, M. Gupta, J. Chem. Sci. 128, 849 (2016)
A. Ilangovan, S. Malayappasamy, S. Muralidharan, S. Maruthamuthu, Chem. Cent. J. 5, 1 (2011)
G. Song, B. Wang, H. Luo, L. Yang, Catal. Commun. 8, 673 (2007)
V. Rama, K. Kanagaraj, K. Pitchumani, Tetrahedron Lett. 53, 1018 (2012)
M. Tajbakhsh, M. Heidary, R. Hosseinzadeh, Res. Chem. Intermed. 42, 1425 (2016)
A.M. Akondi, M.L. Kantam, R. Trivedi, B. Sreedhar, S.K. Buddana, R.S. Prakasham, S. Bhargava, J. Mol. Catal. A: Chem. 386, 49 (2014)
J.K. Rajput, G. Kaur, Catal. Sci. Technol. 4, 142 (2014)
M. Tajbakhsh, M. Farhang, H.R. Mardani, R. Hosseinzadeh, Y. Sarrafi, Chin. J. Catal. 34, 2217 (2013)
M. Tajbakhsh, M. Farhang, R. Hosseinzadeh, Y. Sarrafi, RSC Adv. 4, 23116 (2014)
M. Tajbakhsh, M. Bazzar, S.F. Ramzanian, M. Tajbakhsh, Appl. Clay Sci. 178, 88 (2014)
Y. Sarrafi, M. Sadatshahabi, K. Alimohammadi, M. Tajbakhsh, Green Chem. 13, 2851 (2011)
Z. Lasemi, E. Mehrasbi, Res. Chem. Intermed. 41(2855–2866), 51 (2015)
J.T. Li, Y.W. Li, Y.L. Song, G.F. Chen, Ultrason. Sonochem. 19, 1 (2012)
J.J. Yu, L.M. Wang, J.Q. Liu, F.L. Guo, Y. Liu, N. Jiao, Green Chem. 12, 216 (2010)
J.M. Khurana, K. Vij, J. Chem. Sci. 124, 907 (2012)
M.R. Awual, J. Environ. Chem. Eng. 7, 103087 (2019)
M.R. Awual, M.M. Hasan, M.A. Khaleque, Sens. Actuators B Chem. 209, 194 (2015)
M.R. Awual, M.M. Hasan, J. Ind. Eng. Chem. 21, 507 (2015)
M.R. Awual, M.M. Hasan, Sens. Actuators B Chem. 202, 395 (2014)
M.R. Awual, T. Yaita, T. Taguchi, H. Shiwaku, Y. Okamoto, J. Hazard. Mater. 278, 227 (2014)
M.R. Awual, S. Suzuki, T. Taguchi, H. Shiwaku, T. Yaita, Chem. Eng. J. 242, 127 (2014)
M.R. Awual, M.M. Hasan, H. Znad, Chem. Eng. J. 259, 611 (2015)
M.R. Awual, T. Kobayashi, H. Shiwaku, Y. Miyazaki, T. Yaita, Chem. Eng. J. 225, 558 (2013)
M.R. Awual, Mater. Sci. Eng. C 101, 686 (2019)
M.R. Awual, J. Environ. Chem. Eng. 7, 103378 (2019)
J. Bian, F. Peng, X.P. Peng, P. Peng, F. Xu, R.C. Sun, BioResources 7, 4626 (2012)
S.G. Khokarale, J.P. Mikkola, RSC Adv. 8, 18531 (2018)
K.Y. Lee, T. Tammelin, K. Schulfter, H. Kiiskinen, J. Samela, A. Bismarck, A.C.S. Appl, Mater. Interfaces 4, 4078 (2012)
A. Fatoni, S. Koesnarpadi, N. Hidayati, Indones J. Chem. 15, 194 (2015)
T.T.T. Ho, T. Zimmermann, R. Hauert, W. Caseri, Cellulose 18, 1391 (2011)
V.K. Rao, M.M. Kumar, A. Kumar, Indian J. Chem. B 50, 1128 (2011)
A. Hasaninejad, N. Golzar, M. Beyrati, A. Zare, M.M. Doroodmand, J. Mole. Catal. A: Chem. 372, 137 (2013)
F. Darviche, S. Balalaie, F. Chadegani, P. Salehi, Synth. Commun. 37, 1059 (2007)
E.C. Horning, M.G. Horning, J. Org. Chem. 11, 95 (1946)
D.B. Ramachary, M. Kishor, J. Org. Chem. 72, 5056 (2007)
S. Khodabakhshi, M. Baghernejad, Iran. J. Catal. 3, 67 (2013)
M. Kidwai, V. Bansal, P. Mothsra, S. Saxena, R.K. Somvanshi, S. Dey, T.P. Singh, J. Mol. Catal. A: Chem. 268, 76 (2007)
I. Manolov, C. Maichle-Moessmer, N. Danchev, Eur. J. Med. Chem. 41, 882 (2006)
S. Hasanzadeh Banakar, M.G. Dekamin, A. Yaghoubi, New J. Chem. 42, 14246 (2018)
F. Nemati, M.M. Heravi, R. Saeedi Rad. Chin. J. Catal. 33, 1825 (2012)
Acknowledgements
We are grateful to University of Mazandaran for partial support of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lasemi, Z., Tajbakhsh, M., Alinezhad, H. et al. 1,8-Diazabicyclo [5.4.0] undec-7-ene functionalized cellulose nanofibers as an efficient and reusable nanocatalyst for the synthesis of tetraketones in aqueous medium. Res Chem Intermed 46, 3667–3682 (2020). https://doi.org/10.1007/s11164-020-04167-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04167-y