Abstract
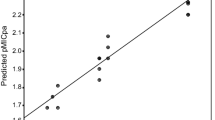
A new series of diorganotin(IV) complexes of 4-((2-hydroxybenzylidene)amino)-[1,1′-biphenyl]-3-ol (H2L1), 4-((2-hydroxy-5-nitrobenzylidene)amino)-[1,1′-biphenyl]-3-ol (H2L2), 4-((3-bromo-2-hydroxy-5-nitrobenzylidene)amino)-[1,1′-biphenyl]-3-ol (H2L3) and 4-((3,5-dibromo-2-hydroxybenzylidene)amino)-[1,1′-biphenyl]-3-ol (H2L4) Schiff base ligands with general formula R2SnL (where R = Me, Et, Bu and Ph) were synthesized. Structural aspect of these synthesized compounds have been described with the help of elemental analysis and spectroscopic techniques like FT-IR, UV–Vis, NMR, Mass and fluorescence. Schiff bases coordinated to tin metal as dibasic tridentate ligand through imine nitrogen and two phenolic oxygen atoms forming pentacoordinated complexes. Metal complexes gave low molar conductance value describing their non-electrolytic nature. Thermal decomposition of the complexes resulted in the formation of SnO2 as end product. The in vitro antimicrobial activities have been evaluated against Gram positive bacteria, Gram-negative bacteria and two fungal strains. The screening evaluation showed that organotin(IV) complexes have better antimicrobial activity. The free radical scavenging ability of the compounds was invest by in vitro antioxidant assay involving DPPH radical and was found to be moderately good. The anti-inflammatory activity was done by egg albumin method and percentage inhibition of protein denaturation was calculated. By comparing the biological activities of the synthesized compounds it was found that metal complexes were more potent than the free ligands and compounds 16, 20 were having more potential as drugs.
Graphical abstract













Similar content being viewed by others
References
N. Galic, Z. Cimerman, V. Tomisic, Spectrochim. Acta A 71, 1274 (2008)
M.H.S.A. Hamid, A.N.A.H. Said, A.H. Mirza, M.R. Karim, Md Arifuzzaman, MdA Ali, P.V. Bernhardt, Inorg. Chim. Acta 453, 742 (2016)
T.S.B. Baul, P. Kehie, A. Duthie, N. Guchhait, N. Raviprakash, R.B. Mokhamatam, S.K. Manna, N. Armata, M. Scopelliti, R. Wang, U. Englert, J. Inorg. Biochem. 168, 76 (2017)
S. Shujah, N. Khalid, S. Ali, Russ. J. Gen. Chem. 87(3), 515 (2017)
S. Nazneen, S. Ali, S. Shahzadi, S. Shujah, Russ. J. Gen. Chem. 87(12), 2970 (2017)
P. Bhatra, J. Sharma, R.A. Sharma, Y. Singh, Appl. Organomet. Chem. 31(7), e3639 (2017)
N. Neelofar, N. Ali, A. Khan, S. Amir, N.A. Khan, M. Bilal, Bull. Chem. Soc. Ethiopia 31(3), 445 (2017)
M. Jain, V. Singh, R.V. Singh, J. Iran. Chem. Soc. 1, 20 (2004)
Z. Asadi, Int. J. Chem. Kin. 43, 247 (2011)
L. Pellerito, L. Nagy, Coord. Chem. Rev. 224, 111 (2002)
S.K. Bharti, S.K. Patel, G. Nath, R. Tilak, S.K. Singh, Trans. Met. Chem. 35, 917 (2010)
N.A. Oztas, G. Yenisehirli, N. Ancınc, S.G. Oztas, Y. Ozcand, Spectrochim. Acta A 72, 929 (2009)
J. Devi, J. Yadav, Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry—Anti-Cancer Agents), 18(3), 335 (2018)
J. Devi, S. Pachwania, Inorg. Chem. Commun. 91, 44 (2018)
R. Joshi, N. Pandey, R. Tilak, S.K. Yadav, H. Mishra, S. Pokharia, Appl. Organomet. Chem. 32(5), e4324 (2018)
F. Shaheen, M. Sirajuddin, S. Ali, Z. Rehman, P.J. Dyson, N. Ali Shah, M.N. Tahir, J. Organomet. Chem. 186, 13 (2018)
J.O. Adeyemi, D.C. Onwudiwe, Molecule 23(10), 2571 (2018)
M. Sankarganesh, J.D. Raja, K. Sakthikumar, R.V. Solomon, J. Rajesh, S. Athimoolam, V. Vijayakumar, Bioorg. Chem. 81, 144 (2018)
M.S. Agiorgiti, A. Evangelou, P. Vezyraki, S.K. Hadjikakou, V. Kalfakakou, I.T. Sanaktsidi, A. Batistatou, J. Zelovitis, Y.V. Simos, V. Ragos, S. Karkabounas, D. Peschos, Med. Chem. Res. 27(4), 1122 (2018)
P.G. Avaji, C.V. Kumar, S.A. Patil, K. Shivananda, C. Nagaraju, Eur. J. Med. Chem. 44, 3552 (2009)
J.O. Adeyemi, D.C. Onwudiwe, A.C. Ekennia, C.P. Anokwuru, N. Nundkumar, M. Singh, E.C. Hosten, Inorg. Chim. Acta 485, 64 (2019)
Q.K. Panhwar, S. Memon, Inorg. Chim. Acta 407, 252 (2013)
M. Carcelli, G. Corazzari, S. Ianelli, G. Pelizzi, C. Solinas, Inorg. Chim. Acta 353, 310 (2003)
M. Nardelli, C. Pelizzi, G. Pelizzi, P. Tarasconi, J. Chem. Soc., Dalton Trans. 0(2), 321 (1985)
M.A. Girasolo, A. Attanzio, P. Sabatino, L. Tesoriere, S. Rubino, G. Stocco, Inorg. Chim. Acta 423(B), 168 (2014)
J. Devi, S. Devi, A. Kumar, Med. Chem. Comm. 5 (2016)
J. Devi, S. Devi, A. Kumar, Heterocyc. Chem. 27(6), 361 (2016)
J. Devi, S. Kumari, S. Devi, R. Malhotra, P. Kumar, B. Narasimhan, Monatsh. für Chem. 146(12), 1995 (2014)
A.I. Vogel, Text Book of Quantitative Chemical Analysis, 5th edn. (Longmans, Edison, 1999)
J. Devi, N. Batra, Spectrochim. Acta A Mol. Biomol. Spectrosc. 135, 710 (2015)
J. Devi, M. Yadav, D. Kumar, L.S. Naik, D.K. Jindal, J. App. Organomet. Chem. 33(2), e4693 (2019)
A. Braca, N.D. Tommasi, L.D. Bari, C. Pizza, M. Politi, I. Morelli, J. Nat. Prod. 64, 892 (2001)
B. Bozin, N.M. Dukic, I. Samojlik, A. Goran, R. Igic, Food Chem. 111, 925 (2008)
S. Chandra, P. Chatterjee, P. Dey, S. Bhattacharya, Asian Pacific J. Trop. Biomed. S178 (2012)
S. Chandra, P. Chatterjee, P. Dey, S. Bhattacharya, J. Adv. Pharm. Edu. Res. 2, 25 (2012)
H.L. Singh, J.B. Singh, K.P. Sharma, Res. Chem. Intermed. 38, 53 (2012)
J. Devi, M. Yadav, A. Kumar, A. Kumar, Chem. pap. (2018)
H.L. Singh, J.B. Singh, S. Bhanuka, Res. Chem. Intermed. 42, 997 (2016)
M. Hong, H.D. Yin, X.Y. Zhang, C. Li, C.H. Yue, S. Cheng, J. Organomet. Chem. 724, 23 (2013)
S. Shujah, Z. Urrehman, S. Ali, N. Khalid, N. Muhammad, M.N. Tahir, J. Organomet. Chem. 696, 2772 (2011)
S. Shujah, S. Ali, N. Khalid, M.J. Alam, S. Ahmad, A. Meetsma, Chem. Pap. 72, 903 (2017)
Yu-Xing Tan, Zhi-Jian Zhang, Yang Liu, Yu. Jiang-Xi, Xiao-Ming Zhu, Dai-Zhi Kuang, Wu-Jiu Jiang, J. Mol. Struct. 1149, 874 (2017)
T.P. Lockhart, W.F. Manders, Inorg. Chem. 25, 892 (1986)
T.P. Lockhart, W.F. Manders, J. Am. Chem. Soc. 109, 7015 (1987)
H.D. Yin, S.W. Chen, L.W. Li, D.Q. Wang, Inorg. Chim. Acta 360, 2215 (2007)
L.S. Mun, M.A. Hapipah, S.K. Shin, A.M. Sri Nurestri, L.K. Mun, Appl. Organomet. Chem. 26, 310–311 (2012)
K. SinghDharampal, Dharampal, V. Parkash, Phosphorus, Sulfur Silicon Relat. Elem. 183, 2784 (2008)
J. Wagler, U. Bohme, E. Brendler, B. Thomas, S. Goutal, H. Mayr, B. Kempf, G.Y. Remennikov, G. Roewer, Inorg. Chim. Acta 358, 4270 (2005)
G.G. Mohamed, C.M. Sharaby, Spectrochim. Acta A 66, 949 (2007)
M. Gulcan, Y. Karatas, S. Isik, G. Ozturk, E. Akbas, E. Sahin, J. Fluoresc. 24, 1679 (2014)
Y. Han-dong, C. Ji-Chun, Q. Yan-Ling, Polyhedron 27, 2157 (2008)
A. Chilwal, P. Malhotra, A.K. Narula, Phosphorus, Sulfur Silicon Relat. Elem. 189, 410 (2014)
S. Mun Lee, H.M.A. Kae, S. Sim, S.N.A. Malek, K.M. Lo, Inorg. Chim. Acta 406, 272 (2013)
K.S. Prasad, L.S. Kumar, M. Prasad, H.D. Revanasiddappa, Bioinorg. Chem. Appl. 2010, 1 (2010)
J. Devi, S. Kumari, S. Asijaa, R. Malhotra, Phosphorus, Sulfur Silicon Relat. Elem. 187, 1409 (2012)
Y. Anjaneyula, R.P. Rao, Synth. React. Inorg. Met. Org. Chem. 16, 257 (1986)
Z.H. Chohan, A. Scozzafava, C.T. Supuran, J. Enzy. Inhib. Med. Chem. 18, 259 (2003)
R. Raman, A. Selvan, J. Coord. Chem. 64, 534 (2011)
A. Chilwal, P. Malhotra, A.K. Narula, Phosphorus, Sulfur Silicon Relat. Elem. 189, 410 (2014)
M.A. Affan, M.A. Salam, F.B. Ahmad, R.B. Hitam, F. White, Polyhedron 33, 19 (2012)
D.B. Shpakovsky, C.N. Banti, E.M. Mukhatova, YuA Gracheva, V.P. Osipova, N.T. Berberova, D.V. Albov, T.A. Antonenko, L.A. Aslanov, E.R. Milaev, S.K. Hadjikakoub, Dalton Trans. 43, 6880 (2014)
S. Jabbar, I. Shahzadi, R. Rehman, H. Iqbal, Q. Ul-Ain, A. Jamil, R. Kousar, S. Ali, S. Shahzadi, M.A. Choudhary, M. Shahid, Q.M. Khan, S.K. Sharma, K. Qanungo, J. Coord. Chem. 65(4), 572 (2012)
A. Ramírez-Jiménez, R. Luna-García, A. Cortés-Lozada, S. Hernández, T.R. írez-Apan, A. Nieto-Camacho, E. Gómez, J. Organomet. Chem. 738, 10 (2013)
M. Sirajuddin, S. Ali, F.A. Shah, M. Ahmad, M.N. Tahir, J. Iran. Chem. Soc. 11, 297 (2014)
F. Diaz-Gonzalez, I. Gonzalez-Alvaro, M.R. Campanero, F. Mollinedo, M.A. del Pozo, C. Munoz, J.P. Pivel, F. Sinchez-Madrid, J. Clin. Invest. 95, 1756 (1995)
M. Nath, P.K. Saini, A. Kumar, J. Organomet. Chem. 695, 1353 (2010)
M. Nath, R. Yadav, G. Eng, T. Nguyen, A. Kumar, J. Organomet. Chem. 577, 1 (1999)
G. A. Kaysen, J. A. Dubin, Hans-Georg M¨Uller, L. Rosales, N. W. Levin, W. E. Mitch, The Hemo Study Group, Kidney International, 65, 1408 (2004)
Acknowledgements
One of the author Ms. Jyoti Yadav is highly thankful to the HSCST Panchkula for the financial support and also thankful to A. P. J. Abdul Kalam Central Instrumentation Laboratory GJUS&T, Hisar for carrying the spectral data and Department of Chemistry, GJUS&T, Hisar for financial support in the form of research project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Devi, J., Yadav, J. & Singh, N. Synthesis, characterisation, in vitro antimicrobial, antioxidant and anti-inflammatory activities of diorganotin(IV) complexes derived from salicylaldehyde Schiff bases. Res Chem Intermed 45, 3943–3968 (2019). https://doi.org/10.1007/s11164-019-03830-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03830-3