Abstract
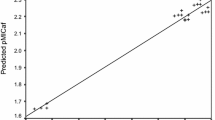
A series of some new diorganotin(IV) complexes [R2SnLCl] was synthesized by the reaction of 2-(3-methylbutanoyl)-1H-indene-1,3(2H)-dione and 4-substituted anilines (p-OCH3, p-NO2, p-CH3, p-Cl) with R2SnCl2, (R=Me, Et, n-Bu, Ph) in 1:1 molar ratio. The structure of the Schiff bases and their complexes were characterized by IR, 13C, 1H, 119Sn NMR, and mass spectral techniques. The synthesized ligands and derived organotin complexes were evaluated in vitro against some bacterial strains, viz., Escherichia coli, Pseudomonas aeruginosa, Bacillus cereus, Staphylococcus aureus and fungal strains, viz., Aspergillus flavus, Aspergillus niger, and Candida albicans by serial dilution method. The antimicrobial results revealed that organotin complexes showed a distinct escalation in biocidal activity. Phenyl and butyl complexes were found to be more intoxicating. Furthermore, we performed QSAR studies which explained the different factors affecting the enhancement in the bioactivity of the complexes.
Graphical abstract




Similar content being viewed by others
References
Thumar NJ, Patel MP (2012) Med Chem Res 21:1751
Peto J (2011) Nature 411:390
Chopra I, Schofield C, Everett M, O’Neill A, Miller K, Wilcox M, Frere JM, Dawson M, Czapleavski L, Courvalin P (2008) Lancet Infect Dis 8:133
Zamudio-Rivera LS, George-Tellez R, López-Mendoza G, Morales-Pacheco A, Flores E, Höpfl H, Barba V, Fernandez FJ, Cabirol N, Beltrán HI (2005) Inorg Chem 44:5370
Gaur S, Fahmi N, Singh RV (2007) Phosphorus. Sulfur Silicon Relat Elem 182:853
Khan RA, Yadav S, Hussain Z, Arjmand F, Tabassum S (2014) Dalton Trans 43:2534
Shpakovsky DB, Banti CN, Mukhatova EM, Gracheva YA, Osipova VP, Berberova NT, Albov DV, Antonenko TA, Aslanov LA, Milaeva ER, Hadjikakou SK (2014) Dalton Trans 43:6880
Bekhit AA, Fahmy HT, Rostom SA, Baraka AM (2003) Eur J Med Chem 38:27
Rathelot P, Azas N, El-Kashef H, Delmas F, Di Giorgio C, Timon-David P, Maldonado J, Vanelle P (2002) Eur J Med Chem 37:671
Chavan SP, Sivappa R (2004) Tetrahedron Lett 45:3941
de Oliveira KN, Andermark V, Onambele LA, Dahl G, Prokop A, Ott I (2014) Eur J Med Chem 87:794
Ge R, Wang YJ, Tang L, Cheng JM, Han LG, Li YL, Li QS (2014) Appl Organomet Chem 28:204
Iqbal H, Ali S, Shahzadi S (2015) Cogent Chem 1:1029039
Dokorou V, Kovala-Demertzi D, Jasinski JP, Galani A, Demertzis MA (2004) Helv Chim Acta 87:1940
Kovala-Demertzi D, Dokorou V, Primikiri A, Vargas R, Silvestru C, Russo U, Demertzis MA (2009) J Inorg Biochem 103:738
Singh RV, Chaudhary P, Chauhan S, Swami M (2009) Spectrochim Acta Part A 72:260
Chaudhary A, Agarwal M, Singh RV (2006) Appl Organomet Chem 20:295
Sharma A, Jain A, Saxena S (2015) Appl Organomet Chem 29:499
Chauhan HPS, Shaik NM (2005) J Inorg Biochem 99:538
Bhatra P, Sharma J, Sharma RA, Singh Y (2017) Appl Organomet Chem 31:e3639
Shah FA, Sabir S, Fatima K, Ali S, Qadri I, Rizzoli C (2015) Dalton Trans 44:10467
Jain M, Gaur S, Diwedi SC, Joshi SC, Singh RV, Bansal A (2004) Phosphorus. Sulfur Silicon Relat Elem 179:1517
Sirajuddin M, Ali S, McKee V, Sohail M, Pasha H (2014) Eur J Med Chem 84:343
Nath M, Vats M, Roy P (2013) Eur J Med Chem 59:310
Watanabe M, Watanabe K (2002) Vet Hum Toxicol 44:140
Jankovics H, Pettinari C, Marchetti F, Kamu E, Nagy L, Troyanov S, Pellerito L (2003) J Inorg Biochem 97:370
Danish M, Raza MA, Ilyas T, Sharif A, Anjum N (2015) Med Chem 5:373
Awang N, Kamaludin NF, Hamid A, Mokhtar NWN, Rajab NF (2012) Pak J Biol Sci 15:833
Yang Y, Hong M, Xu L, Cui J, Chang G, Li D, Li C (2016) J Organomet Chem 804:48
Nath M, Yadav R, Gielen M, Dalil H, Vos DD, Eng G (1997) Appl Organomet Chem 11:727
Yin HD, Hong M, Li G, Wang DQ (2005) J Organomet Chem 690:3714
Prasad KS, Kumar LS, Prasad M, Revanasiddappa HD (2010) Bioinorg Chem Appl 2010:1
Tabassuma S, Khan RA, Arjmand F, Sen S, Kayal J, Juvekar AS, Zingde SM (2011) J Organomet Chem 696:1600
Kumar D, Singh VK, Khiwar SS, Saxena N (2014) J Drug Deliv Ther 4:73
Kumar D, Singh VK, Khiwar SS, Saxena N (2014) J Biomed Pharm Res 3:30
Dawara L, Singh RV (2011) App Organomet Chem 25:643
Jayabalakrishnan C, Natarajan K (2002) Trans Met Chem 27:75
Dhawan SN, Dasgupta S, Mor S, Gupta SC (1993) Ind J Heterocycl Chem 2:155
Mor S, Nagoria S (2015) Chem Biol Interface 5:389
Malhotra R, Mehta J, Puri JK (2007) Cent Eur J Chem 5:858
Srinivas P, Suresh T, Revanasiddappa M, Khasim S (2008) J Chem 5:627
Nath M, Yadav R (1997) Bull Chem Soc Jpn 70:1331
Kapoor R, Gupta A, Kapoor P, Venugopalan P (2003) Appl Organomet Chem 17:607
Buntine MA, Kosovel FJ, Tiekink ER (2003) Cryst Eng Commun 5:331
Dakternieks D, Duthie A, Smyth DR, Stapleton CP, Tiekink ER (2003) Organometallics 22:4599
Dubey RK, Baranwal P, Jha AK (2012) J Coord Chem 65:2645
Czympiel L, Lekeu JM, Hegemann C, Mathur S (2017) Inorg Chim Acta 455:197
Kashar TI (2014) Eur Chem Bull 3:878
Sallam SA, Orabi AS, El-Shetary BA, Lentz A (2002) Trans Metal Chem 27:447
Hansch C, Leo A, Unger SH, Kim KH, Nikaitani D, Lien EJ (1973) J Med Chem 16:1207
Kier LB, Hall LH (1976) Molecular connectivity in chemistry and drug research. Academic Press, New York
Kumar A, Narasimhan B, Kumar D (2007) Bioorg Med Chem 15:4113
Hyperchem 6.0 (1993) Hypercube Inc, Gainesville
TSAR 3D Version 3.3 (2000) Oxford Molecular Limited
SPSS for Windows, version 10.05 (1999) SPSS Inc, Bangalore
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khatkar, P., Asija, S., Ahlawat, A. et al. Synthesis, characterization, in vitro antimicrobial and QSAR studies of diorganotin(IV) complexes of Schiff bases derived from 2-(3-methylbutanoyl)-1H-indene-1,3(2H)-dione and 4-substituted anilines. Monatsh Chem 150, 207–218 (2019). https://doi.org/10.1007/s00706-018-2308-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-018-2308-6