Abstract
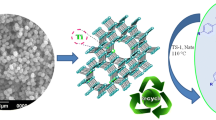
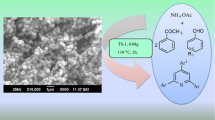
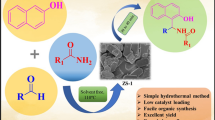
TS-1 zeolite was prepared for the synthesis of 5-arylidene-2,4-thiazolidinediones/Rhodanines in aqueous medium by incorporating titanium(IV) cations in a silicate-1 framework using hydrothermal treatment and characterized by using XRD, EDX, BET, FT-IR and SEM techniques. The catalytic activity of the catalyst was tested for Knoevenagel condensation reaction. The condensation of active ethylene 2,4-thiazolidinedione with substituted aryl aldehydes under aqueous medium at 90 °C afforded the corresponding product in excellent yield up to 92% within 30 min. The present method offers several advantages over the reported methods such as easy separation of catalyst, simple work-up procedure, and an excellent yield of desired product. Furthermore, the catalyst could be reused without significant loss in activity.
Graphical abstract





Similar content being viewed by others
References
P.A. Datar, S.B. Aher, J. Saudi Chem. Soc. 20, S196 (2016)
K.D. Patel, C.N. Patel, G.M. Patel, Med. Chem. (Los Angeles) 6(10), 647 (2016)
R.S. Bhatti, S. Shah, Suresh, P. Krishan, J.S. Sandhu, Int. J. Med. Chem. 793260 (2013)
K.R. Alagawadi, S.G. Alegaon, Arab. J. Chem. 4, 465 (2011)
B. Bozic, J. Rogan, D. Poleti, M. Rancic, N. Trisovic, B. Bozic, G. Uscumlic, Arab. J. Chem. 10, S2637 (2017)
N. Trotsko, A. Przekora, J. Zalewska, G. Ginalska, A. Paneth, M. Wujec, J. Enzyme Inhibid. Med. Chem. 33, 17 (2017)
O. Zvarec, S.W. Polyak, W. Tieu, K. Kuan, H. Dai, D.S. Pedersen, R. Morona, L. Zhang, G.W. Booker, A.D. Abell, Bioorg. Med. Chem. Lett. 22, 2720 (2012)
E. Valadbeigi, S. Ghodsi, Med. Chem. (Los Angeles) 7(6), 178 (2017)
Sucheta, S. Tahlan, P.K. Verma, Chem. Cent. J. 11, 130 (2017)
L.F. Junior, M.J.B. Rego, M.B. Cavalcanti, M.C. Pereira, M.G. Pitta, P.S.S. Oliveira, S.M.C. Gonçalves, A.L.B.P. Duarte, M.C.A. Lima, I. Pitta, M.G. Pitta, BioMed Res. Int. 926060 (2013)
A.C. Tripathi, S. Gupta, G. Fatima, P.K. Sonar, A. Verma, S.K. Saraf, Eur. J. Med. Chem. 72, 52e77 (2014)
D.D. Subhedar, M.H. Shaikh, L. Nawale, A. Yeware, D. Sarkar, B.B. Shingate, Res. Chem. Intermed. 42, 6607 (2016)
M. Azizmohammadi, M. Khoobi, A. Ramazani, S. Emami, A. Zarrin, O. Firuzi, R. Miri, A. Shafiee, Eur. J. Med. Chem. 59, 15e22 (2013)
D. Glintborg, M. Andersen, Gynecol. Endocrinol. 26(11), 791 (2010)
A.P. Rauter, M. Padilha, J.A. Figueiredo, M.I. Ismael, J. Justino, H. Ferreira, M.J. Ferreira, C. Rajendran, R. Wilkins, P.D. Vaz, M.J. Calhorda, J. Carbohydr. Chem. 24, 275 (2005)
S.V. Laxmi, P. Anil, G. Rajitha, A.J. Rao, P.A. Crooks, B. Rajitha, J. Chem. Biol. 9, 97 (2016)
S. Ozkirimli, F. Kazan, Y. Tunali, J. Enzyme Inhibid. Med. Chem. 24(2), 447 (2009)
R.S. Bahare, S. Ganguly, K. Choowongkomon, S. Seetaha, DARU J. Pharm. Sci. 23(6), 1 (2015)
S. Chandrappa, S.B. Benaka Prasad, K. Vinaya, C.S. Ananda Kumar, N.R. Thimmegowda, K.S. Rangappa, Investig. New Drugs 26, 437 (2008)
J. Dolezel, P. Hirsova, V. Opletalova, J. Dohnal, V. Marcela, J. Kunes, J. Jampilek, Molecules 14, 4197 (2009)
N. Ergenc, G. Capan, N.S. Gunay, S. Ozkirimli, M. Gungor, S. Ozbey, E. Kendi, Arch. Pharm. 332(10), 343 (1999)
H. Sachdeva, D. Dwivedi, P. Goyal, Org. Chem. Int. 976032 (2013)
Y. Wu, H. Tai, H. Cho, Bioorg. Med. Chem. 18, 1428 (2010)
S. Chandrappa, C.V. Kavitha, M.S. Shahabuddin, K. Vinaya, C.S.A. Kumar, S.R. Ranganatha, S.C. Raghavan, K.S. Rangappa, Bioorg. Med. Chem. 17, 2576 (2009)
Y. Mi Ha, Y.J. Park, J. Kim, D. Park, J.Y. Park, H. Jin Lee, J.Y. Lee, H.R. Moon, H.Y. Chung, Eur. J. Med. Chem. 49, 245 (2012)
Y. Zhang, Z. Zhou, Org. Chem. Int. 194784 (2012)
S. Rathod, M. Navgire, B. Arbad, M. Lande, S. Afr. J. Chem. 65, 196 (2012)
G. Bruno, L. Costantino, C. Curinga, R. Maccari, F. Monforte, F. Nicolo, R. Ottana, M.G. Vigorita, Bioorg. Med. Chem. 10(4), 1077 (2002)
J. Zhang, Y. Zhang, Z. Zhou, Green Chem. Lett. Rev. 7(1), 90 (2014)
A. Khazaei, H. Veisi, M. Safaei, H. Ahmadian, J. Sulfur Chem. 35(3), 270 (2014)
K.F. Shelke, S.B. Sapkal, G.K. Kakade, S.A. Sadaphal, B.B. Shingate, M.S. Shingare, Green Chem. Lett. Rev. 3, 17 (2010)
D. Kumar, S. Narwal, J.S. Sandhu, Int. J. Med. Chem. 273534 (2013)
Suresh, J.S. Sandhu, Org. Med. Chem. Lett. 3, 2 (2013)
K.F. Shelke, A.D. Badar, J.B. Devhade, Chem. Biol. Interface 6(6), 410 (2016)
Z. Zhuo, L. Wu, L. Wang, Y. Ding, X. Zhang, Y. Liu, M. He, RSC Adv. 4, 55685 (2014)
S.B. Shin, D.W. Lee, D. Chadwick, J. Mol. Catal. A Chem. 423, 478 (2016)
A.S. Albero, A.G. Atienza, E. Serrano, J.G. Martínez, J.S. Albero, Catal. Commun. 44, 35 (2014)
D. Vitvarov, L. Lupínkov, M. Kub, Microporous Mesoporous Mater. 210, 133e141 (2015)
P. Kumar, R.K. Pandey, Green Chem. 2, 29 (2000)
S. P. Gadekar, G. T. Pawar, R. R. Magar, M. K. Lande, Polycycl. Aromat. Compd. (2017). https://doi.org/10.1080/10406638.2017.1363060
N. Sachan, S.S. Kadam, V.M. Kulkarni, Indian J. Heterocycl. Chem. 17, 57 (2007)
L.S. Gadekar, B.R. Arbad, M.K. Lande, Org. Chem. Indian J. 4, 458 (2008)
R.S. Mahalle, P.D. Netankar, S.P. Bondge, R.A. Mane, Green Chem. Lett. Rev. 1, 103 (2008)
Acknowledgements
We are grateful to the Head, Department of Chemistry, Dr. Babasaheb Ambedkar Marathwada University, Aurangabad-431004(MS), India for providing the laboratory facility. The corresponding author MKL is thankful to UGC New Delhi (INDIA) for providing a grant under major research project F. No. 43-184/2014 (SR). The authors are thankful to IIT Chennai and Mumbai, STIC Cochin and SAIF Chandigarh for providing characterization facilities.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gadekar, S.P., Dipake, S.S., Gaikwad, S.T. et al. Solid acid TS-1 catalyst: an efficient catalyst in Knoevenagel condensation for the synthesis of 5-arylidene-2,4-thiazolidinediones/Rhodanines in aqueous medium. Res Chem Intermed 44, 7509–7518 (2018). https://doi.org/10.1007/s11164-018-3570-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3570-2