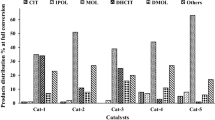
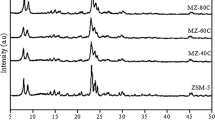
Abstract
Citronellal cyclisation to isopulegol is an important intermediate step in the production of menthol. Several heteropoly acids (PTA) supported on modified montmorillonite (MM) catalysts were synthesized and then tested in cyclisation reactions. The prepared samples were characterized by XRD, ICP-OES, FTIR, N2 sorption, NH3-TPD, pyridine adsorption, amine titration and FE-SEM techniques. Effects of post-treatment were studied on montmorillonite pore structure, acidity and catalytic activity. The catalytic activity and isopulegol selectivity improved with acid-treatment and PTA loading. The amount of Lewis acidity of montmorillonite was enhanced with acid-treatment and PTA impregnation. In cyclisation, highest catalytic activity (31.87 mmol cat g−1 min−1) was achieved with 96% isopulegol yield in the use of 20% PTA-MM catalyst. The highest catalytic activity and selectivity were obtained in the presence of higher acidity and strong Lewis acidic sites, whereas effects of pore structure blockage seemed minor. The catalytic activity further decreased with the loss of active acidic sites (L and B) due to PTA decomposition with calcination at a higher temperature.










Similar content being viewed by others
Abbreviations
- S BET :
-
Specific surface area
- V total :
-
Total pore volume
- V meso :
-
Mesopore volume
- Ds:
-
Stereo-selectivity of (±) isopulegol
- MT:
-
Montmorillonite
- MM:
-
Acid-treated montmorillonite
- PTA:
-
Phosphotungstic acid
- PTA-MM:
-
Heteropoly acid supported acid-treated montmorillonite
- L.A:
-
Lewis acid sites
- B.A:
-
Brønsted acid sites
- BET:
-
Brunauer–Emmet–Teller
- FTIR:
-
Fourier transforms infrared spectroscopy
- ICP-OES:
-
Inductively coupled plasma optical emission spectrometry
- XRD:
-
X-ray diffraction
References
G.K. Chuah, S.H. Liu, S. Jaenicke, L.J. Harrison, J. Catal. 200, 2 (2001)
H. Oertling, A. Reckziegel, H. Surburg, H.-J. Bertram, Chem. Rev. 107, 5 (2007)
T. Patel, Y. Ishiuji, G. Yosipovitch, J. Am. Acad. Dermatol. 57, 5 (2007)
P. Kočovský, G. Ahmed, J. Šrogl, A.V. Malkov, J. Steele, J. Org. Chem. 64, 8 (1999)
V.K. Aggarwal, G.P. Vennall, P.N. Davey, C. Newman, Tetrahedron Lett. 39, 1997 (1998)
G.D. Yadav, J.J. Nair, Chem. Commun. 21, 2369 (1998)
P. Mäki-Arvela, N. Kumar, V. Nieminen, R. Sjöholm, T. Salmi, D.Y. Murzin, J. Catal. 225, 1 (2004)
M. Vandichel, F. Vermoortele, S. Cottenie, D.E. De Vos, M. Waroquier, V. Van Speybroeck, J. Catal. 305, 118 (2013)
K.A. da Silva, P.A. Robles-Dutenhefner, E.M.B. Sousa, E.F. Kozhevnikova, I.V. Kozhevnikov, E.V. Gusevskaya, Catal. Commun. 5, 8 (2004)
S.M. Coman, P. Patil, S. Wuttke, E. Kemnitz, Chem. Commun. 4, 460 (2009)
P.R.S. Braga, A.A. Costa, E.F. de Freitas, R.O. Rocha, J.L. de Macedo, A.S. Araujo, J.A. Dias, S.C.L. Dias, J. Mol. Catal. A Chem. 358, 99–105 (2012)
Z. Yongzhong, N. Yuntong, S. Jaenicke, G.-K. Chuah, J. Catal. 229, 2 (2005)
S.K. Bhorodwaj, D.K. Dutta, Appl. Catal. A 378, 2 (2010)
P.J. Kropp, G.W. Breton, S.L. Craig, S.D. Crawford, W.F. Durland, J.E. Jones, J.S. Raleigh, J. Org. Chem. 60, 13 (1995)
M. Fuentes, J. Magraner, C. De Las Pozas, R. Roque-Malherbe, J.P. Pariente, A. Corma, Appl. Catal. 47, 2 (1989)
J.-I. Tateiwa, A. Kimura, M. Takasuka, S. Uemura, J. Chem. Soc. Perkin Trans. 1, 15 (1997)
G.D. Yadav, Catal. Surv. Asia 9, 2 (2005)
S.V. Awate, S.B. Waghmode, K.R. Patil, M.S. Agashe, P.N. Joshi, Korean J. Chem. Eng. 18, 2 (2001)
M. Misono, Korean J. Chem. Eng. 14, 6 (1997)
M. He, A. Pan, J. Xie, H. Li, X. Yuan, X. Cheng, M. Chen, Korean J. Chem. Eng. 29, 10 (2012)
E. Vyskočilová, M. Krátká, M. Veselý, E. Vrbková, L. Červený, Res. Chem. Intermed. 42, 9 (2016)
D.R. Park, U.G. Hong, S.H. Song, J.G. Seo, S.-H. Baeck, J.S. Chung, I.K. Song, Korean J. Chem. Eng. 27, 2 (2010)
E. Rafiee, M. Khodayari, Res. Chem. Intermed. 42, 4 (2016)
Y. Izumi, Res. Chem. Intermed. 24, 4 (1998)
G. Bai, H. Zhang, T. Li, H. Dong, J. Han, Res. Chem. Intermed. 41, 8 (2015)
M.N. Timofeeva, Appl. Catal. A 256, 1–2 (2003)
A. Samzadeh-Kermani, Chem. Mon. 147, 761–765 (2016)
E. Rafiee, N. Nobakht, L. Behbood, Res. Chem. Intermed. 42, 5573–5585 (2016)
J. Safari, M. Sadeghi, Res. Chem. Intermed. 42, 12 (2016)
G.F. Chen, N. Xiao, J.S. Yang, H.Y. Li, B.H. Chen, L.F. Han, Res. Chem. Intermed. 41, 8 (2015)
F.H.J. Al-Shemmari, A.A. Rabah, E.A.J. Al-Mulla, N.O.M.A. Alrahman, Res. Chem. Intermed. 39, 9 (2013)
K. Ravi, B. Krishnakumar, M. Swaminathan, Res. Chem. Intermed. 41, 8 (2015)
M. Kurian, R. Babu, J. Environ. Chem. Eng. 1, 1–2 (2013)
P. Ravindranathan, P.B. Malla, S. Komarneni, R. Roy, Catal. Lett. 6, 3 (1990)
B. Vijayakumar, G. Ranga Rao, J. Porous Mater. 19, 2 (2012)
S. Rostamizadeh, A.M. Amani, R. Aryan, H.R. Ghaieni, L. Norouzi, Chem. Mon. 140, 5 (2009)
Y.-S. Shin, S.-G. Oh, B.-H. Ha, Korean J. Chem. Eng. 20, 1 (2003)
D.-H. Shin, J.-J. Kim, B.-S. Yu, M.-H. Lee, D.-W. Park, Korean J. Chem. Eng. 20, 1 (2003)
A. Samzadeh-Kermani, S. Miri, Korean J. Chem. Eng. 32, 6 (2015)
C.-Y. Ryu, S.-D. Yeo, Korean J. Chem. Eng. 27, 2 (2010)
E. Rafiee, M. Kahrizi, M. Joshaghani, P. Ghaderi-Sheikhi Abadi, Res. Chem. Intermed. 42, 6 (2016)
P. Kar, B.G. Mishra, J. Environ. Chem. Eng. 4, 2 (2016)
S.K. Bhorodwaj, D.K. Dutta, Appl. Clay Sci. 53, 2 (2011)
V.V. Bokade, G.D. Yadav, Appl. Clay Sci. 53, 2 (2011)
V.N. Shetti, J. Kim, R. Srivastava, M. Choi, R. Ryoo, J. Catal. 254, 2 (2008)
M.W. Anderson, J. Klinowski, Zeolites 6, 3 (1986)
E.P. Parry, J. Catal. 2, 5 (1963)
K. Siddhartha, D. Bhorodwaj, D.D. Kumar, Appl. Catal. A 378, 2 (2010)
L. Bieseki, F. Bertell, H. Treichel, F.G. Penha, S.B.C. Pergher, Mater. Res. 16, 1122–1127 (2013)
X. Han, W. Yan, C.-T. Hung, Y. He, P.-H. Wu, L.-L. Liu, S.-J. Huang, S.-B. Liu, Korean J. Chem. Eng. 33, 2063–2072 (2016)
I.V. Kozhevnikov, J. Mol. Catal. A Chem. 305, 1–2 (2009)
Acknowledgements
This research was conducted through a mutual collaboration of Higher Education Commission Pakistan and Hanyang University, South Korea.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shah, A.K., Park, S., Khan, H.A. et al. Citronellal cyclisation over heteropoly acid supported on modified montmorillonite catalyst: effects of acidity and pore structure on catalytic activity. Res Chem Intermed 44, 2405–2423 (2018). https://doi.org/10.1007/s11164-017-3237-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-017-3237-4