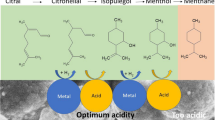
Abstract
Menthol synthesis is possible through citral and citronellal hydrogenations via following multistage chemical reactions such as hydrogenation and cyclization. This research mainly focuses on a design of selective, active and cost-effective metal-acid (bifunctional) catalysts for menthol production via citral and citronellal hydrogenations. More specifically, Pd and Ni metals were impregnated over acidic support (e.g., hetero-poly acid supported montmorillonite, HPA_MM). The prepared catalysts were characterized by BET, pyridine adsorption and amine titration methods. Some of the most important parameters such as metal type and loading; applied pressure and reaction time have been investigated throughout this work. The obtained results reveals that the 8 wt% Ni-HPA-MM catalyst (Cat-5) has produced 63% menthols (initial reaction rate 0.126 mmol/min) from citral hydrogenation (80 °C, 1.0 MPa) within 24 h. Similarly, during lower applied pressure (0.5 MPa), the production of menthol was significantly improved (approximately 98% of menthol, initial reaction rate ~ 0.138 mmol/min) with the application of 8 wt% Ni-HPA-MM catalyst (Cat-5) in citronellal hydrogenation. Higher menthol selectivity was achieved from both reactions (citral and citronellal hydrogenation) which might be due to the presence of strong Lewis and medium Brønsted acid sites.





Similar content being viewed by others
Abbreviations
- MMT:
-
Montmorillonite
- HPA:
-
Heteropoly acid (20% loading of Phosphotungstic acid)
- MM:
-
Acid-treated montmorillonite
- HPA-MM:
-
Heteropoly acid (20% loading of Phosphotungstic acid) supported acid-treated montmorillonite
- Pd-HPA-MM:
-
Pd-loaded heteropoly acid supported acid-treated montmorillonite
- Ni-HPA-MM:
-
Nickel loaded heteropoly acid supported acid-treated montmorillonite
- Cat-1:
-
5 wt% Pd-MM
- Cat-2:
-
2.5 wt% Pd-HPA-MM
- Cat-3:
-
5 wt% Pd-HPA-MM
- Cat-4:
-
5 wt% Ni-HPA-MM
- Cat-5:
-
8 wt% Ni-HPA-MM
- CIT:
-
Citronellal
- IPOL:
-
Isopulegols
- MOL:
-
Menthols
- DHCIT:
-
Dihydrocitronellal
- DMOL:
-
3, 7-Dimethyl-1-octanol
References
Mertens P, Verpoort F, Parvulescu A-N, De Vos D (2006) Pt/H-beta zeolites as productive bifunctional catalysts for the one-step citronellal-to-menthol conversion. J Catal 243(1):7–13. https://doi.org/10.1016/j.jcat.2006.06.017
Trasarti AF, Marchi AJ, Apesteguía CR (2007) Design of catalyst systems for the one-pot synthesis of menthols from citral. J Catal 247(2):155–165
Negoi A, Teinz K, Kemnitz E, Wuttke S, Parvulescu V, Coman S (2012) Bifunctional nanoscopic catalysts for the one-pot synthesis of (±)-menthol from citral. Top Catal 55(7–10):680–687. https://doi.org/10.1007/s11244-012-9850-y
Mäki-Arvela P, Kumar N, Kubička D, Nasir A, Heikkilä T, Lehto V-P, Sjöholm R, Salmi T, Murzin DY (2005) One-pot citral transformation to menthol over bifunctional micro- and mesoporous metal modified catalysts: effect of catalyst support and metal. J Mol Catal A 240(1):72–81. https://doi.org/10.1016/j.molcata.2005.06.023
Salminen E, Virtanen P, Kordás K, Mikkola J-P (2012) Alkaline modifiers as performance boosters in citral hydrogenation over supported ionic liquid catalysts (SILCAs). Catal Today 196(1):126–131
Mäki-Arvela P, Kumar N, Nieminen V, Sjöholm R, Salmi T, Murzin DY (2004) Cyclization of citronellal over zeolites and mesoporous materials for production of isopulegol. J Catal 225(1):155–169. https://doi.org/10.1016/j.jcat.2004.03.043
Solodar J (1978) Asymmetric and regioselective hydrogenation of piperitenone by homogeneous rhodium complexes. J Org Chem 43(9):1787–1789. https://doi.org/10.1021/jo00403a035
Negoi A, Teinz K, Kemnitz E, Wuttke S, Parvulescu VI, Coman SM (2012) Bifunctional nanoscopic catalysts for the one-pot synthesis of (±)-menthol from citral. Top Catal 55(7):680–687. https://doi.org/10.1007/s11244-012-9850-y
Rojas H, Martínez JJ, Mancípe S, Borda G, Reyes P (2012) Citral hydrogenation over novel niobia and titania supported Au, Ir–Au and Ir catalysts. Reac Kinet, Mech Catal 106(2):445–455
Mukherjee S, Vannice MA (2006) Solvent effects in liquid-phase reactions: I. Activity and selectivity during citral hydrogenation on Pt/SiO2 and evaluation of mass transfer effects. J Catal 243(1):108–130. https://doi.org/10.1016/j.jcat.2006.06.021
Plößer J, Lucas M, Claus P (2014) Highly selective menthol synthesis by one-pot transformation of citronellal using Ru/H-BEA catalysts. J Catal 320:189–197. https://doi.org/10.1016/j.jcat.2014.10.007
Milone C, Gangemi C, Neri G, Pistone A, Galvagno S (2000) Selective one step synthesis of (−)menthol from (+)citronellal on Ru supported on modified SiO2. Appl Catal A 199(2):239–244. https://doi.org/10.1016/S0926-860X(99)00560-8
Ravasio N, Poli N, Psaro R, Saba M, Zaccheria F (2000) Bifunctional copper catalysts. Part II. Stereoselective synthesis of (-)-menthol starting from (+)-citronellal. Top Catal 13(3):195–199. https://doi.org/10.1023/a:1009086317432
Nie Y, Niah W, Jaenicke S, Chuah G-K (2007) A tandem cyclization and hydrogenation of (±)-citronellal to menthol over bifunctional Ni/Zr-beta and mixed Zr-beta and Ni/MCM-41. J Catal 248(1):1–10. https://doi.org/10.1016/j.jcat.2007.02.018
Cortés CB, Galván VT, Pedro SS, García TV (2011) One pot synthesis of menthol from (±)-citronellal on nickel sulfated zirconia catalysts. Catal Today 172(1):21–26. https://doi.org/10.1016/j.cattod.2011.05.005
Cirujano FG, Llabrés-i-Xamena FX, Corma A (2012) MOFs as multifunctional catalysts: one-pot synthesis of menthol from citronellal over a bifunctional MIL-101 catalyst. Dalton Trans 41(14):4249–4254. https://doi.org/10.1039/c2dt12480g
da Silva Rocha KA, Robles-Dutenhefner PA, Sousa EMB, Kozhevnikova EF, Kozhevnikov IV, Gusevskaya EV (2007) Pd–heteropoly acid as a bifunctional heterogeneous catalyst for one-pot conversion of citronellal to menthol. Appl Catal A 317(2):171–174. https://doi.org/10.1016/j.apcata.2006.10.019
Virtanen P, Karhu H, Toth G, Kordas K, Mikkola J-P (2009) Towards one-pot synthesis of menthols from citral: modifying supported ionic liquid catalysts (SILCAs) with Lewis and Brønsted acids. J Catal 263(2):209–219
Trasarti AF, Marchi AJ, Apesteguía CR (2013) Synthesis of menthols from citral on Ni/SiO2–Al2O3 catalysts. Catal Commun 32:62–66. https://doi.org/10.1016/j.catcom.2012.11.030
Ambrosetti A, Silvestrelli PL (2016) Cohesive properties of noble metals by van der Waals–corrected density functional theory: Au, Ag, and Cu as case studies. Phys Rev B 94(4):045124
Yongzhong Z, Yuntong N, Jaenicke S, Chuah G-K (2005) Cyclisation of citronellal over zirconium zeolite beta—a highly diastereoselective catalyst to (±)-isopulegol. J Catal 229(2):404–413. https://doi.org/10.1016/j.jcat.2004.11.015
Chuah GK, Liu SH, Jaenicke S, Harrison LJ (2001) Cyclisation of citronellal to isopulegol catalysed by hydrous zirconia and other solid acids. J Catal 200(2):352–359. https://doi.org/10.1006/jcat.2001.3208
Cirujano F, i Xamena FL, Corma A (2012) MOFs as multifunctional catalysts: one-pot synthesis of menthol from citronellal over a bifunctional MIL-101 catalyst. Dalton Trans 41(14):4249–4254
Negoi A, Wuttke S, Kemnitz E, Macovei D, Parvulescu VI, Teodorescu C, Coman SM (2010) One-pot synthesis of menthol catalyzed by a highly diastereoselective Au/MgF2 catalyst. Angew Chem Int Ed 49(44):8134–8138
Negoi A, Teinz K, Kemnitz E, Wuttke S, Parvulescu VI, Coman SM (2012) Bifunctional nanoscopic catalysts for the one-pot synthesis of (±)-menthol from citral. Top Catal 55(7–10):680–687
Weingarten R, Tompsett GA, Conner WC, Huber GW (2011) Design of solid acid catalysts for aqueous-phase dehydration of carbohydrates: the role of Lewis and Brønsted acid sites. J Catal 279(1):174–182. https://doi.org/10.1016/j.jcat.2011.01.013
Corma A, Hamid SBA, Iborra S, Velty A (2005) Lewis and Brønsted basic active sites on solid catalysts and their role in the synthesis of monoglycerides. J Catal 234(2):340–347. https://doi.org/10.1016/j.jcat.2005.06.023
Zabeti M, Wan Daud WMA, Aroua MK (2009) Activity of solid catalysts for biodiesel production: a review. Fuel Process Technol 90(6):770–777. https://doi.org/10.1016/j.fuproc.2009.03.010
Tanabe K, Hölderich WF (1999) Industrial application of solid acid–base catalysts. Appl Catal A 181(2):399–434. https://doi.org/10.1016/S0926-860X(98)00397-4
Yadav GD (2005) Synergism of clay and heteropoly acids as nano-catalysts for the development of green processes with potential industrial applications. Catal Surv Asia 9(2):117–137. https://doi.org/10.1007/s10563-005-5997-x
Shah AK, Maitlo G, Korai RM, Unar IN, Shah AA, Khan HA, Shah SFA, Ismail U, Park YH (2019) Citronellal cyclisation to isopulegol over micro-mesoporous zsm-5 zeolite: effects of desilication temperature on textural and catalytic properties. Reac Kinet, Mech Catal 128:1–16
Bhorodwaj SK, Dutta DK (2010) Heteropoly acid supported modified Montmorillonite clay: an effective catalyst for the esterification of acetic acid with sec-butanol. Appl Catal A 378(2):221–226. https://doi.org/10.1016/j.apcata.2010.02.026
Timofeeva MN (2003) Acid catalysis by heteropoly acids. Appl Catal A 256(1–2):19–35. https://doi.org/10.1016/S0926-860X(03)00386-7
Farfan-Torres EM, Sham E, Grange P (1992) Pillared clays: preparation and characterization of zirconium pillared montmorillonite. Catal Today 15(3):515–526. https://doi.org/10.1016/0920-5861(92)85016-F
Ming-li C, Yong-fu Y, Ji-zu Y, Ming-he C (2002) Preparation and properties of pillared montmorillonite by polyhydroxyl-aluminum-manganese cations. J Wuhan Univ Technol-Mater Sci Ed 17(2):43–46. https://doi.org/10.1007/BF02832620
Shah AK, Park S, Khan HA, Bhatti UH, Kumar P, Bhutto AW, Park YH (2018) Citronellal cyclisation over heteropoly acid supported on modified montmorillonite catalyst: effects of acidity and pore structure on catalytic activity. Res Chem Intermed 44(4):2405–2423. https://doi.org/10.1007/s11164-017-3237-4
Flessner U, Jones DJ, Rozière J, Zajac J, Storaro L, Lenarda M, Pavan M, Jiménez-López A, Rodrı́guez-Castellón E, Trombetta M, Busca G (2001) A study of the surface acidity of acid-treated montmorillonite clay catalysts. J Mol Catal A 168(1–2):247–256. https://doi.org/10.1016/S1381-1169(00)00540-9
Bieseki L, Bertell F, Treichel H, Penha FG, Pergher SBC (2013) Acid treatments of montmorillonite-rich clay for Fe removal using a factorial design method. Mater Res 16:1122–1127
Adams JM (1987) Synthetic organic chemistry using pillared, cation-exchanged and acid-treated montmorillonite catalysts—a review. Appl Clay Sci 2(4):309–342. https://doi.org/10.1016/0169-1317(87)90039-1
Mishra T, Parida KM, Rao SB (1996) Transition metal oxide pillared clay: 1. A comparative study of textural and acidic properties of Fe(III) pillared montmorillonite and pillared acid activated montmorillonite. J Colloid Interface Sci 183(1):176–183. https://doi.org/10.1006/jcis.1996.0532
Bolognini M, Cavani F, Cimini M, Pozzo LD, Maselli L, Venerito D, Pizzoli F, Veronesi G (2004) An environmentally friendly synthesis of 2,4-dihydroxybenzophenone by the single-step O-mono-benzoylation of 1,3-dihydroxybenzene (resorcinol) and fries rearrangement of intermediate resorcinol monobenzoate: the activity of acid-treated montmorillonite clay catalysts. C R Chim 7(2):143–150. https://doi.org/10.1016/j.crci.2003.12.005
Bokade VV, Yadav GD (2011) Heteropolyacid supported on montmorillonite catalyst for dehydration of dilute bio-ethanol. Appl Clay Sci 53(2):263–271. https://doi.org/10.1016/j.clay.2011.03.006
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60(2):309–319. https://doi.org/10.1021/ja01269a023
Emeis CA (1993) Determination of integrated molar extinction coefficients for infrared absorption bands of pyridine adsorbed on solid acid catalysts. J Catal 141(2):347–354. https://doi.org/10.1006/jcat.1993.1145
Virtanen P, Mikkola J-P, Salmi T (2007) Kinetics of citral hydrogenation by supported ionic liquid catalysts (SILCA) for fine chemicals. Ind Eng Chem Res 46(26):9022–9031. https://doi.org/10.1021/ie0707467
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shah, A.K., Maitlo, G., Shah, A.A. et al. One pot menthol synthesis via hydrogenations of citral and citronellal over montmorillonite-supported Pd/Ni-heteropoly acid bifunctional catalysts. Reac Kinet Mech Cat 128, 917–934 (2019). https://doi.org/10.1007/s11144-019-01679-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-019-01679-6