Abstract
Passive acoustic monitoring (PAM) is useful for monitoring vocal fish but has had so far limited application in fisheries management. Here, four years (2016–2019) of concurrent daily catch and effort fishery data in Portugal and species-specific vocal activity in the Tagus estuary are compared to describe biological and small-scale fishery dynamics for a large sciaenid fish, the meagre (Argyrosomus regius), that aggregates to spawn. Consistent patterns in seasonality of acoustic and fisheries variables indicate that most fishing takes place within the Tagus estuary in spring and summer months, when higher vocal activity related to spawning aggregations is detected in the PAM station. Good fit of statistical models shows that PAM (sound pressure level in the third-octave band with centre frequency at 500 kHz during dusk) and PAM-supported variables (mean weekly catch per first sale transaction) can provide useful surveillance indicators to improve local management. Signs of overexploitation and hyperstability are detected and communicated to the estuarine fishing communities with the aim to initiate an adaptive local management cycle. The approach can be relevant for fisheries targeting other vocal fish that seasonally aggregate and face similar threats of overexploitation.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The family of sciaenid fish (corvinas, croakers and drums) includes some of the biggest teleosts (maximum size can exceed 170 cm in the genus Argyrosomus, Griffiths 1996; Potts et al. 2010; González-Quirós et al. 2011) and many species are targeted by commercial and recreational fisheries worldwide (Gannon 2008; Morales-Nin et al. 2012; Erisman et al. 2012; Ferguson et al. 2014). Sciaenids are also known for being vocal fish, producing some of the loudest biological sounds in the marine realm, audible to the human ear (Smith 1905; Erisman and Rowell 2017). The family includes several dozen genera and more than two hundred taxa, with species-specific sound repertoires and features (Ramcharitar et al. 2006; Luczkovich et al. 2008a; Parmentier et al. 2018; Bolgan et al. 2020), including choruses that can vary in intensity depending on water temperature (Monczak et al. 2019; Vieira et al. 2022). Most vocal sciaenids show temporal periodicity in sound production, often associated with a marked daily spawning synchronicity, mainly towards dusk (Holt et al. 1985; Saucier and Baltz 1993; Locascio and Mann 2011; Aalbers and Sepulveda 2012; Montie et al. 2016; Vieira et al. 2022). In the process, some species create ephemeral sex-biased groupings (Fitzhugh et al. 1993; Aalbers and Drawbridge 2008), sometimes leading to spectacular concentrations (Sadovy de Mitcheson 2016; Erisman and Rowell 2017). Several require estuarine and coastal salt-marsh habitats to complete their life cycle (Griffiths 1996; Ferguson et al. 2008; Luczkovich et al. 2008b; González-Quirós et al. 2011; Morales-Nin et al. 2012; Lowerre-Barbieri et al. 2016).
Sciaenids of the genus Argyrosomus that use estuaries to complete their life cycle also tend to have late maturation, high longevity and few predators in their adult phase, other than humans (Griffiths 1996; Potts et al. 2010; González-Quirós et al. 2011). Predictability of aggregation close to land, large size and piscivory, turn Argyrosomus species attractive and accessible to small-scale fisheries along the eastern Atlantic and the Indo-Pacific. For example, meagre (A. regius) sounds, audible during estuarine spawning aggregations, have been used for centuries by fishers to improve catchability in the Gironde estuary (Lagardére and Mariani 2006). Estuarine aggregations can attract commercial, subsistence and recreational activities, both legal and illegal (Griffiths 1996; Silberschneider and Gray 2008; Mota 2018; Sadovy de Mitcheson et al. 2019). Large fishing effort and high catchability during aggregation also turn such populations vulnerable to overexploitation (Sadovy de Mitcheson 2016; Heyman et al. 2019). Complex mating systems (Connaughton and Taylor 1996; Aalbers and Drawbridge 2008; Lowerre-Barbieri et al. 2016) can further increase vulnerability at low population levels through Allee effects (Rowe and Hutchins 2003). Sciaenid overexploitation is usually related to insufficient management due to limited monitoring investment, unaccounted fishing mortality, scant regulation or inadequate compliance (Potts et al. 2010; Prista et al. 2011; Mirimin et al. 2016; Erisman and Rowell 2017; Heyman et al. 2019).
In Portugal, meagre heavier than 50 kg can be occasionally caught (maximum register by research team: 67 kg), corresponding to fish with more than 20 years of age (maximum known register: 43 years) and, arguably, more than 15 years of recurrent seasonal migrations to the Tagus estuary for reproduction. The species is known to have been present in the Tagus and Sado estuaries since the Mesolithic (6000 BP – 4500 BP), with signs of exploitation by collector settlement-subsistence systems (Gabriel et al. 2012). In the late nineteenth century important fisheries existed in these estuaries during summer months (April to August), when adult meagre entered the rivers to spawn (Baldaque da Silva 1892). Industrial and urban pollution deteriorated the Tagus estuarine habitat in the mid-twentieth century (Raimundo et al. 2011; Rodrigues et al. 2020; Batista et al. 2022), possibly contributing to the disappearance of meagre for decades. Meagre return to the Tagus estuary was reported in the late 1990s, initially as juvenile concentrations detected and targeted by fishers (Costa and Cabral 1999). In the past two decades there has been regular presence of spawners and recruits in the estuary (Prista 2013), supporting an important small-scale seasonal commercial fishery, mainly for adults, and year-long recreational fishing, mainly for juveniles (Mota 2018).
Passive acoustic monitoring (PAM) is a useful monitoring technique for vocal fish that has so far been little used for fisheries and environmental management purposes (Luczkovich et al. 2008a; Gannon 2008; Aalbers and Sepulveda 2012; Parmentier et al. 2018; Di Iorio et al. 2020). Recent automations in species identification and sound variables detection and quantification, associated to increasing use of continuous PAM recordings (Vieira et al. 2015, 2019; Monczak et al. 2019), have made available longer series of species-specific acoustic data. Here, we review sciaenid literature in which PAM has been used concurrently with other sampling techniques and extend the remit of comparisons to a context of reduced spatial overlap between methods. We use four consecutive years (2016–2019) of concurrent daily commercial fishery data for meagre in Portugal (catch per unit effort, CPUE) and species-specific PAM variables from a station in the Tagus estuary (Vieira et al. 2022) to explore the potential relevance of PAM to enhance the utilization of aggregate CPUE data in the management of this small-scale fishery. Knowing PAM to be reliable for delimiting the temporal dynamics of meagre reproduction in Portugal (Vieira et al. 2019, 2022), we use the statistical relationships of this analysis to increase the confidence in the use of longer term indices based on the catch data. We further hypothesize that stationary PAM can be an unbiased, albeit imprecise, sampler of adult meagre estuarine abundance and delineate the additional research steps necessary to test it. The use of sound-based or PAM-supported (catch-based) surveillance indicators (Shephard et al. 2015) within a participatory management framework (Heyman et al. 2019) can be relevant for other fisheries that target vocal fish and face similar threats.
Material and Methods
The Study System
The Tagus estuary (Portugal) is one of only five known large concentration areas of meagre reproduction within the Northeast Atlantic and the Mediterranean Sea (Haffrey et al. 2012; Almeida et al. 2022). Sexually mature fish (4–5 years old onwards) perform seasonal migrations to the natal estuary, where they stay on average 2–3 months during the summer spawning season. In the late nineteenth century similar concentrations were reported during summer months in the Sado estuary (Baldaque da Silva 1892). Nowadays, some adult fish also enter other Portuguese estuaries, such as the Sado, Mira and Guadiana rivers, in summer months (Prista 2013), but without forming spawning aggregations. In the remaining months of the year adult fish disperse in coastal waters and are occasionally caught as bycatch (Prista 2013; Castro et al. 2021). Meagre´s current distribution in Portugal partially overlaps with that of weakfish (Cynoscion regalis), a non-native sciaenid originally from the western Atlantic and recently introduced to the Iberian Peninsula (Morais et al. 2017; Gomes et al. 2017). However, although weakfish currently forms regular part of commercial landings in Portugal and Spain, its value and consumer preference are still low in comparison to meagre (Bañon et al. 2017; Cerveira et al. 2022).
Tagus Estuary
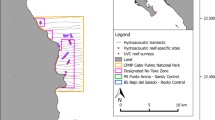
The Tagus River estuary is among the largest in Europe (> 300 km2), although a large fraction of the inner estuary is within the intertidal zone, becoming exposed at low tide. Regular environmental monitoring currently takes place (Rodrigues et al. 2020), including the CoastNet research infrastructure (Castellanos et al. 2021) of in-situ sensors with sound (PAM) recorders and an array of biotelemetry receivers to track tagged fish (Fig. 1), such as meagre, and a land-based observatory for marine mammals (Batista et al. 2022). Adult meagre start to enter the Tagus estuary in February/March where they mainly reproduce in May and June. During this period adult meagre will mainly use the deepest channels of the middle and lower section of the Tagus estuary (Marques and Quintella unpublished). Ongoing tracking studies indicate that adults can stay in the estuary until August/September and that young juveniles may overwinter in and around it. Juveniles mostly feed on crustaceans, mainly mysidacians and brown shrimp (Crangon crangon) in the Tagus (Cabral and Ohmert 2001), with an ontogenetic transition to piscivory that also leads to some cannibalism (Hurbans et al. 2017). Immediately after entering the estuary adult meagre start feeding on cuttlefish (Sepia officinalis), changing latter in the spawning season to thinlip grey mullet (Chelon ramada). The latter is a catadromous species that forms large aggregations in the beginning of its trophic migration in several Portuguese estuaries, including the Tagus river (Pereira et al. 2021).
Map of the study area, including: the meagre geographic distribution range; the intensity of fishing per area by Tagus estuary vessels with port of register in Lisbon, V. F. Xira, Trafaria, and Barreiro (targeting meagre); and the CoastNet Monitoring Infrastructure (Acoustic Biotelemetry and Passive Acoustic monitoring)
The Tagus Fishing Communities
Close to 300 small fishing vessels (< 9 m) with a port of register within the Tagus estuary (Fig. 1) reported some landing of meagre between 2016 and 2019 (Table 1). Other seasonal fisheries in the Tagus estuary include octopus (Octopus vulgaris), sea bass (Dicentrarchus labrax) and sea bream (Sparus aurata) and a year-round activity of harvesting the invasive clam Ruditapes philippinarum (Coelho et al. 2021; Batista et al. 2022). During the summer months, meagre becomes the main target species within the estuary practically for all fishers that are not dedicated to bivalve harvesting. Annual landed biomass by vessels registered in Tagus ports on average exceeds 200 tonnes and is worth more than 1 million € (in the four years of the study the average first sale price for meagre was around 6–7 € per kg). Fishing for meagre takes place with nets, longlines and lines. Around 100 fishers participated so far in meetings promoted by the co-authors with three fisher associations since late 2021. Also, 43 fishers replied individually a questionnaire dedicated to the meagre fishery during 2022 (Marques, unpublished and Table 1). The latter extends earlier inquiries by Mota (2018) in the same communities, covering issues related to the description of the fishing practice, perceptions on state of the resource and opinions on management measures and related governance (see also Stratoudakis et al. 2023 for equivalent inquiries to other estuarine fisheries). Although survey results and methodology will be reported in a separate publication, some auxiliary information is used here to characterise the communities and identify the main fishing areas (shade map within Fig. 1). Two answers are also used in the results to demonstrate the current perception of higher exploitation rate in comparison to 10 years ago (in Fig. 2b), reinforcing a perception that was already detected by Mota (2018) in relation to the period 2013–2017.
Annual catch (biomass, panel a) and fishing effort (number of first sale transactions, Panel c) for meagre in Portugal during 2002–2020 based on fisheries data, separately for vessels registered in ports within the Tagus estuary (solid line) and remaining (broken line). Panel b reports boxplots of Tagus fishers´ perceptions on the evolution of juvenile meagre abundance and fishing effort in the past decade (5-level ordinal scale response in 2022 questionnaire survey)
The fishery is regulated by a limited entry licensing system. The minimum landing size is 42 cm and has also to be respected by recreational fishers. Recreational fishing has a bag limit of 10 kg per day plus the largest individual (trophy) and can only take place during daytime. Within the Tagus estuary nets and longlines have specific requirements and limits for minimum mesh, height and size of nets, and number of hooks and nets per licensed vessel. Juvenile meagre and, in more recent years, weakfish can be caught as bycatch in gillnets for seabass. There are no temporal limits to meagre fishing. Nevertheless, net deployment is limited in practice by the presence of the local jellyfish (Catostylus tagi) that clog fishing nets from June onwards, until the first rain outflows in autumn (Gueroun et al. 2021). Finally, fishing is prohibited near the commercial port of Lisbon and within the extensive navigation corridors of the river Tagus. The prohibition is valid at the river mouth and towards the cargo terminals, as well as along the busy passenger ferry lines linking the two margins (Alves et al. 2021).
Monitoring
Passive acoustics has been shown to be a non-invasive and cost-effective method for monitoring vocal fish (Connaughton and Taylor 1995; Luczkovich et al. 1999; Ramcharitar et al. 2006; Parmentier et al. 2018). There have also been several appeals to extend PAM into fisheries monitoring and regulation (Gannon 2008; Luczkovich et al. 2008a) and into marine conservation (Aalbers and Sepulveda 2012; Howe et al. 2019; Di Iorio et al. 2020), but without known management applications so far (Heyman et al. 2019). PAM has been used for estimating animal population size and density, mainly for terrestrial but also for marine mammals. This occurs when the more traditional methods of visual surveys or physical trapping are less viable and when probability of sound detection can be estimated and density of recorded vocalization can be associated to a vocalization rate (Marques et al. 2013). Such requisites are not usually met in fish PAM, but concurrent sampling with other marine survey methods show that PAM variables correlate with several fish population parameters (Table 2).
In the case of the meagre fishery in Portugal there is no dedicated monitoring program and no stock assessment (Prista 2013). The species is not detected in routine fish monitoring surveys of the Portuguese continental shelf directed to pelagic (Daily Egg Production Method—DEPM; and active acoustic surveys, led by IPMA since the 1980s) or demersal species (scientific trawling, led by IPMA since the 1970s). Further, the small-scale fleet segments that mainly land meagre (Prista 2013; Castro et al. 2021) have little representation in the standard biological fish market sampling, with limited length frequency data and no other biological information regularly collected. Finally, the reliability of commercial landing data for meagre is unknown due to generic concerns with possible underreporting or misreporting of valuable target species (Maunder et al. 2006). Nevertheless, earlier studies with regional catch data segments (Prista et al. 2011; Prista 2013) showed internal consistency in the meagre data series, with consistent auto-correlation patterns at a monthly scale in each segment. At the time, time-series analysis revealed the system to be stable during the first decade of the twenty-first century, not detecting evidence of changes in the exploitation patterns (Prista et al. 2011). The rationale used by Prista et al. (2011) for the parsimonious detection of alert signals in fisheries exploitation is similar to that proposed by Shephard et al. (2015) for surveillance indicators. The latter aim to monitor key aspects of the ecosystem when existing information is insufficient to define targets and support formal state assessment and specific management advice (Shephard et al. 2015).
Fisheries Data
Daily first sale meagre landings data for mainland Portugal are available since 1995, but more reliable from 2002 onwards (Prista et al. 2011). Landings (hereafter catch) correspond to catch because discards are negligible for this valuable species (Castro et al. 2021). Data are made available at IPMA by the Directorate General of Fisheries (DGRM), duly anonymised to prevent identification of individual vessels. First sale transaction data correspond to weight and value of species sale in auction (or contract) for each daily transaction per vessel. Each fishing trip with landings of a species can have more than one transaction per day (i.e., multiple buyers), depending on the size of the catch and the respective buyer needs. Register of sale takes place at the port of landing, but each vessel also has a port of vessel origin/registration. Here we use the port of vessel origin to distinguish small-scale vessels registered within the Tagus estuary from remaining fishing vessels in Portugal, as this in practice corresponds to two distinct phases of meagre fishing, estuarine and oceanic (Prista 2013). For the analysis we use two data sets:
-
(i)
the shorter series 2016–2019 to compare fisheries data from the Tagus-registered vessels with meagre PAM variables (period selected to avoid presence of weakfish in acoustic data);
-
(ii)
the longer series 2002–2022 of fisheries data to provide a first estimate of temporal trends in meagre CPUE and the proportion of national catch obtained within the Tagus estuary.
Daily and weekly fishery data are aggregated to provide estimates of catch (kg) and fishing effort (number of first sale transactions). The latter is used as a proxy for the number of fishing days with meagre landings, itself a proxy of fishing effort. Exploratory analysis showed that the number of daily transactions was highly correlated with the number of distinct vessels declaring landings in that day (Pearson correlation coefficient between 0.78 and 0.9 for estuarine fleet components by port of register). Most inner estuary vessels operate exclusively within the Tagus estuary, some outer estuary vessels occasionally operate in the ocean under good weather conditions (see Table 1). For a few of these vessels, meagre is a target species all year round, but for the majority meagre targeting is seasonal. Entry of adult fish to the estuary increases fishing effort due to seasonal meagre fishers and the intensification of regular activity. Number of transactions in first sale may also increase per landing event in relation to higher yield and larger fish size.
Passive Acoustic Monitoring
Continuous acoustic recordings were obtained in the Tagus estuary from April 2016 to September 2019 (Air Force Base 6, Montijo, Portugal; 38º42'N, 8º58'W). At this location, distinct sounds from two native vocal fish species are usually detected: the meagre and the Lusitanian toadfish (Halobatrachus didactylus—Vieira et al. 2015). More recently weakfish have also been detected at this site. We therefore discarded data from 2020 onwards to minimize hypothetical confounding with weakfish vocalizations. It should be noted that the remaining PAM recording stations shown in Fig. 1 were only deployed after 2019, when weakfish sounds started becoming conspicuous in the estuary.
Recordings were made using a High Tech 94 SSQ hydrophone (sensitivity – 165 dB re. 1 V/μPa, frequency response within ± 1 dB up to 6 kHz) anchored near the bottom and connected to a stand-alone 16 channel data-logger (LGR-5325, Measurement Computing Corp, Norton, MA, USA; 4 kHz sampling rate, 16 bit). Close to the pier, where the hydrophone is placed, the water column varies with tide from 3 to 6.5 m. The region in front of the pier is deeper (8 to 10 m), with the bottom mostly composed of sandy/silty substrate. Meagre tend to produce choruses in the area in front of the pier, a region crossed by ferryboats connecting Lisbon to Montijo and by small fishing boats operating in the estuary. Lusitanian toadfish nest close to the pier and at the banks, where shelters are made available by members of the research team.
An automatic sound recognition system with hidden Markov model (HMM)-based supervised machine learning was used to annotate the continuous recordings. This was applied to detect and identify the presence of meagre sounds, assuming that the time series of an observed sound can be represented by a linear Markov model (for details on the machine-learning algorithm see Young et al. 2006). Vieira et al. (2021, 2022) demonstrated this sound recognition system to be highly effective for recordings in the Tagus estuary. For the purposes of the present study, we used four PAM variables that can be attributed to meagre sounds: counts of isolated grunts, presence and duration (in hours) of chorusing and one-third octave band sound pressure levels centred at 500 Hz (hereafter named TOL500). According to Vieira et al. (2021), TOL500 changes mostly due to the presence of meagre choruses. Because meagre chorusing activity usually starts at late afternoon and continues to the early hours of the next day (Vieira et al. 2022), we considered 24 h periods starting at noon to facilitate data viewing and interpretation.
HMM models were trained using sounds from choruses containing different degrees of call overlap and dominated by either long grunts or isolated pulses (see Vieira et al. 2022). An additional model was used to recognise long grunts with a high signal-to-noise ratio, either isolated or in a chorus. In this study we only considered isolated grunts. Although the detection of isolated grunts might be restricted to sounds with high signal-to-noise ratio, they provide additional information about the presence outside the periods of major activity. TOL500 was computed as a 20 min averaged value, centred around sunset and calibrated adapting the code available by Merchant et al. (2015) (FFT 1024, Hann window, 50% overlap, averaged for each second). The correction factor for calibration was calculated using the hydrophone sensitivity, system gain and zero-to-peak voltage of the analog-to-digital converter. To reduce the subjectivity of measuring the chorus duration, we combined the labels of the automatic recognition system with a 20-min moving average of TOL500. Chorus duration was defined as the sum of the chorus labelled minutes per day where the 20-min averaged TOL500 was above 90 dB re. 1 µPa.
The TOL centred at 500 Hz encompasses a frequency band from 447 to 562 Hz. In the Tagus estuary, the other main components of biophony are the Lusitanian toadfish, with most energy below this frequency band, and the weakfish, whose calls overlap the frequencies of the meagre. Other sources contributing with noise to this frequency band are ferryboats and small fishing boats. Nevertheless, during 2016–2019, the soundscape within the TOL500 frequencies is largely dominated by meagre, since the other relevant sound sources are intermittent (for instance, at rush hour ferryboats can be recorded at the hydrophone less than 15% of the time, Vieira et al. 2021).
Data Analysis
Generalized Additive Models (GAMs) were used to model weekly data of three meagre catch variables: proportion of catch within the Tagus estuary, catch by inner estuary vessels and, as a CPUE index, mean biomass per transaction. A binomial error distribution with a logit link was used for the proportion of catch in the estuary and a negative binomial error distribution with a log link function for the others. In the CPUE model effort was introduced as an offset of the logarithm of weekly meagre transactions by inner estuary vessels. Analysis was performed separately for the shorter data set (2016–2019) with concurrent PAM data and the longer data set (2002–2022) of national catch. In the former, explanatory variables were four PAM variables: number of isolated grunts (square rooted), presence of chorus, total duration of chorusing and mean TOL500 in a week. Year was added as a categorical variable, testing also the significance of its interaction with sound variables. In the longer data set, only the effect of year (as a smooth continuous variable) and the effect of season (as a smooth effect over 52 weeks) were considered, the latter in interaction with a categorical variable indicating whether landings were from Tagus estuary vessels or not.
Exploratory analysis of mean/variance relationships of the daily and weekly data (not shown) revealed variance to be higher than the mean and to increase non-linearly, both for Tagus vessels and the remaining oceanic fishing vessels in Portugal. Dispersion parameter was iterated in the daily data of the short data set (starting with values close to 1 and comparing quality of fit with models considering higher and lower dispersion) and subsequently adjusted to higher values for weekly aggregates. Model selection was based on the three steps described by Wood and Augustin (2002). Full models were initially built, and backwards stepwise elimination used statistics of significance of partial effects, plots of partial effects with error bands and comparisons of nested models with Akaike´s Information Criterion (AIC) to reach the most parsimonious model. First order interactions between year and significant sound variables in the final model were then tested. Selected final models were inspected with residual plots. Modelling was performed in R 4.2.0 (R Core Team 2022) using the libraries mgcv (Wood and Augustin 2002) and MASS (Venables and Ripley 2002).
Results
Meagre catch and first sale market transactions in Portugal have increased during the first decade of the twenty-first century, mainly due to the recovery of the spawning population in the Tagus estuary (Fig. 2). However, in the second decade catch and effort have initially plateaued and subsequently started to recede. Decline starts earlier and is more marked in meagre caught outside the river Tagus, especially in the last 5 years. These patterns are corroborated by the perception of Tagus´ fishers that, in the summer of 2022, considered fishing effort to have increased severalfold in the most recent decade. Similarly, they considered abundance of young meagre (< 6 kg) to have declined a lot in the same period. The weekly temporal dynamics in the data set of 2016–2019 (Fig. 3) show that most fishing for meagre took place within the Tagus estuary from March–April to August–September. During that period meagre chorusing progressively became a regular daily feature within the estuary. Local abundance, approximated by CPUE of inner estuary vessels, peaked around the mid-spawning season, when TOL500 also reached a maximum. Overall, Fig. 3 shows a very good match between fishing and PAM variables´ seasonality during the four years of concurrent observations. This includes synchronicity in some fine-scale patterns, as for example the rapid oscillation of values between weeks 68 and 71 in 2017. The same general seasonality patterns, but with higher variability, were observed when daily data were considered instead of weekly (results not shown).
Weekly register (from January 2016 to December 2019) of PAM variables from Montijo and corresponding commercial fishing activity by Tagus estuary vessels. Top panels: number of days with chorus (a) and number of isolated grunts recorded (b); Mid panels: proportion of national meagre landings by Tagus vessels (c), mean CPUE for inner estuary vessels (d); Bottom panels: total duration of chorusing (hours, e), mean TOL500 (f) per week. Dark bar at the top of graphs indicates weeks 24 to 36 (from mid-April to mid-June) in each year, the approximate peak of meagre spawning activity
GAM results indicate that a large proportion of variation in meagre catch data was explained by PAM variables (Table 3 and Fig. 4). Variation in the proportion of meagre caught by Tagus estuary vessels per week was only significantly related to the presence of chorusing in the given week (46% of deviance explained). TOL500 had a marginal effect, with proportion of catch in the estuary increasing with increasing sound level. Weekly total catch of meagre by vessels registered in the inner Tagus estuary were significantly related with total duration of chorusing and TOL500, but these relationships varied significantly across years (89% of deviance explained). Relationship with isolated grunts was marginal and also had a more complex relationship: for small values of catch it was positive, becoming subsequently negative. Finally, mean weekly CPUE by inner Tagus vessels was significantly related to three PAM variables (80% of deviance explained), positively in the case of chorusing duration and TOL500 and negatively in the case of isolated grunts. Importantly, relationships of PAM variables with CPUE were time-invariant, as their interaction with year did not improve the model fit (Table 3). The same general relationships, but with a smaller proportion of deviance explained, were obtained when daily data were modelled instead of weekly (results not shown).
GAM partial effects of relationships between weekly meagre fisheries data (upper row: proportion of national catch by Tagus vessels; middle row: Tagus meagre catch; bottom row: Tagus CPUE) and PAM variables (second column: presence of days with chorus within a week; third column: number of isolated grunts, square root; fourth column: duration of chorusing, hours; fifth column: mean TOL500) from the Montijo station daily recorded during 2016–2019 (first column). Non-significant sound variables are shown in relation to the residuals of the selected model (top right panel is marginally non-significant but shown to demonstrate overall trend)
The good correspondence between fishery data and PAM variables in the shorter data set increased confidence in the quality of meagre catch reporting and permitted to extend the analysis to the longer series (PAM-supported indicator). The observed changes in CPUE over the past two decades suggests that there was a large increase in fishing effort around 2009–2010, with a corresponding reduction in CPUE (Fig. 2). Reduction in oceanic relative abundance occurred a year earlier than in the Tagus estuary. CPUE since then has been approximately half of earlier values in both systems (Fig. 5). The same model also confirmed that highest CPUEs in the estuary are observed around peak spawning (weeks 18–26), while in oceanic waters the peak occurs much later in the year (Fig. 5 b and d respectively). In addition, analysis of the temporal dynamics of the proportion of meagre catch fished in the ocean (Fig. 6) revealed a continuous decline along the past two decades, with current values of meagre biomass fished outside the estuary below 30% of the total annual catch, as opposed to more than 70% in the start of the series. Some preliminary indication of decline can also be detected within the 2016–2019 PAM series (Fig. 3), although this was not the primary purpose of this analysis. For example, this is apparent in Fig. 3f, if we look at the number of weeks (or the aggregate TOL500) that had values above 100 (the lower limit of the change in the steepness of the relationship with CPUE in Fig. 4o). Further, if we consider the number of days (between Julian day 105 and 165) with TOL500 > 110 (based on the inflection in the relationship with CPUE shown in Fig. 4o), we get 30 days for 2016, 13 for 2017, 16 for 2018 and 4 for 2019.
Decadal (left) and seasonal (right) patterns in meagre CPUE commercial data from Portugal during 2002–2022: (a) raw data, separately for Tagus vessels (solid line) and remaining (broken line) vessels; (b) and (d) Seasonal (weekly) partial effect of CPUE GAM in the Tagus estuary and outside respectively; (c) Annual trend partial effect of meagre CPUE GAM. Dark thick line at the top of seasonal graphs corresponds to weeks 24 to 36 (from mid-April to mid-June) in each year
Decadal patterns of proportion of meagre catch outside the Tagus estuary during 2002–2022 (left panel, GAM partial effect) and weekly distribution of catch outside the Tagus estuary along this period (right panel, size of circles proportional to landings). Partial effect of week (seasonal effect) is not shown, as it mirrors the seasonal effect for Tagus presented in Fig. 5
Discussion
The present analysis revealed consistent patterns between PAM variables of meagre vocal activity in a fixed location within the Tagus estuary and meagre fishery variables based on aggregate commercial catch data across the estuary. Analysis was presented on weekly intervals over four years, but similar results were obtained with daily data. This is in line with PAM literature focusing on the comparison of relative indices of local abundance concurrently obtained (Table 2). The literature review showed that PAM for sciaenids provided congruent results with active acoustics (Rowell et al. 2017) and experimental CPUE (Gannon and Gannon 2010; Monczak et al. 2022; Souza Jr et al. 2023). However, the above studies were designed to test PAM as a method to monitor fish relative abundance and relied on a tight spatial overlap with the other sampling method. This was not the case here, as the comparison was between a fixed-point acoustic recorder in a shallow part of the estuary and the aggregate catch by small boats (< 9 m) from the inner estuary. The congruence between PAM and catch variables, despite the mismatch in the spatial resolution of the two methods, deserves attention, as it may contribute to the extension of the rationale and the potential use of PAM tested so far.
Unlike PAM validation studies, the main objective here was to evaluate the potential of standard fisheries data to be used for management advice and communication with fishers. PAM in shallow estuaries is known to have a limited spatial range of detection due to transmission loss of the acoustic signal (Biggs and Erisman 2021) and meagre detection range in the Tagus estuary will be similar to that reported in other studies. Nevertheless, previous studies in Portugal and in the Tagus estuary have demonstrated the ability of PAM to delimit the seasonal and diel dynamics of meagre reproduction reliably (Vieira et al. 2019, 2022). A difference of the present study with those reported by Gannon and Gannon (2010), Monczak et al. (2022) and Souza Jr et al. (2023) is that the CPUE here is estimated over a larger area based on opportunistic sampling by commercial rather than scientific catch data. Individual catch data are known to be very variable (i.e., low precision) due to differences between fisher performances and fine scale unresolved spatiotemporal variation. Nevertheless, if there are no problems with reporting quality (Maunder et al. 2006), meagre catch data in aggregate provide a daily and seasonal census of the fishing activity in the estuary. The observed temporal synchronicity between PAM and catch data (Fig. 3) indicate that the latter provide a reliable biological signal, increasing confidence to use the longer series (2002–2022) to complement the exploratory analysis on meagre exploitation dynamics (Prista et al. 2011) for another decade.
The PAM station in Montijo provided support for the exploration of the longer CPUE series, but may also contribute directly to cost-effective long-term monitoring for meagre fisheries management. Assuming CPUE as a proxy of relative abundance, the GAM relationships with each PAM variable (Fig. 4) can be analysed to describe the relationship between sound production and fish density, along the lines of the hypotheses proposed by Rowell et al. (2017). Isolated grunts indicate the onset of adult presence (initial positive relation with catch) but are inversely related with subsequent increase in catch and are negatively related with CPUE (Fig. 4h and m respectively). This is possibly due to a decreased detection of isolated grunts when chorusing occurs (peak density/reproductive season), but it may also be an artefact of the much higher values for 2016 (Fig. 3b) that are difficult to explain at this stage. Presence of chorusing within a week provides the best descriptor of adult fish presence in the estuary (Figs. 3c and 4b), but days with chorus per week are insensitive to changes in relative abundance, as a plateau can be easily reached (Fig. 3a). Nevertheless, the variable shows sensitivity to abrupt changes in water temperature (Vieira et al. 2022), as for example the hiatus observed during the spring of 2017. Total chorus duration per week correlates with CPUE, but the relationship is weak and also seems to reach a plateau (Fig. 4n). TOL500 is the most promissing PAM variable to use as a long-term indicator for the meagre fishery, since it is the easiest to estimate, shows the strongest linear relationship with CPUE (Fig. 4o) and is time-invariant (i.e. no significant modification of the relationship between years). The relationship of TOL500 with CPUE seems to become steeper for sound pressure levels above 100 dB, possibly due to the logarithmic nature of the variable.
For TOL500 to serve independently as an indicator of local abundance, we must hypothesize that stationary PAM can be an unbiased, albeit imprecise, sampler of meagre adult density within the Tagus estuary during the reproductive season. Sound pressure level is known to decline rapidly with distance from the source, and the rate of decline is habitat-dependent in shallow estuaries (Biggs and Erisman 2021). Based on the position of the PAM station and the surrounding bathymetry and habitat characteristics, we consider that PAM recordings in this station can reliably capture meagre sounds only up to a few hundred meters away. For TOL500 to serve as an indicator of local abundance, evidence of random presence of adult meagre within the range of detection and absence of territoriality must be proven. Existing information on the general distribution and dynamics of adult meagre within the Tagus estuary, based on telemetry (Quintella and Marques unpublished), and the respective distribution of small-scale fishing activity for meagre, based on the questionnaire survey (Fig. 1), provide some initial support for this hypothesis. The area adjacent to the PAM station is recognised as one of the most important fishing areas and biotelemetry indicates regular movements of adult fish in the lower and mid-estuary, without the establishment of territories. Under these conditions, PAM can be considered an unbiased sampler of adult fish abundance in the estuary, as proximity of imminent spawners to the recorder (and hence the probability of high TOL500) can be seen to be a function of the number of estuarine presences of adult fish (season-dependent, Prista et al. 2011) and the respective fish activity levels (temperature-dependent, Vieira et al. 2022).
For PAM to become a robust long-term instrument to support the adaptive management of meagre in the Tagus estuary, additional steps of research will be needed. One important question relates to species identification in PAM records (Gannon 2008), since calls of the invasive weakfish have also become regular after 2019 within the Tagus estuary (Amorim et al. 2023). Both weakfish and meagre produce grunts during the breeding season that mainly differ in the time interval between pulses in a grunt (weakfish: 58 ± 7 ms, meagre: 17 ± 1 ms; Amorim et al. 2023). Although their sounds are easily distinguished, additional effort will be needed to guarantee automatic separation in annotations and, especially, to separate their contribution in TOL500, given the overlap in the frequency niche of their grunts. Other challenges relate to the hypothesis of unbiased PAM sampling that will need to be addressed with the assistance of the multi-sensor infrastructure of CoastNet (Fig. 1). This refers to sound transmission limits (Biggs and Erisman 2021) and masking by background noise (Gannon 2008) in the context of increasing anthropogenic noise within a busy urban estuary (Alves et al. 2021; Vieira et al. 2021). Sound transmission limits will have to considered in relation to the distribution and dynamics of adult meagre within the estuary to evaluate whether PAM can be considered an unbiased sampler. These relationships can be tested, after the separation of weakfish sounds, by comparing PAM records among stations with contrasting levels of noise and local presence of adult meagre (Fig. 1 shows the full array of sensors currently within the estuary). The latter can be based on the analysis of daily movements of tagged fish as detected and spatially resolved by the biotelemetry array of acoustic receivers in the estuary (MIGRACORV project results, under analysis). Finally, direct methods of estimating meagre abundance and biomass, based on active fisheries acoustics (Rowell et al. 2017) or egg production methods (Holt et al. 1985; Fitzhugh et al. 1993; Stratoudakis et al. 2006), will need to be coupled with concurrent PAM to develop reference points (Shephard et al. 2015) based on sound variables.
PAM variables and PAM-supported results also facilitate communication and increases the credibility of findings, since meagre acoustic behaviour is well known among fishers in the Tagus estuary. A similar confirmatory role of PAM was found for the grouper family, where diver-based visual censuses compared well with PAM in the short-term (Rowell et al. 2012) and the long-term (Koenig et al. 2017). Such congruent results facilitate, in turn, recursive feedbacks between the state of meagre in the estuary and knowledge about the system (Blythe et al. 2017) and increases the legitimacy of potential measures to protect reproduction (Stratoudakis 2021). Temporary cessations of fishing to protect the main period of reproduction is understood and respected by fishers, when supporting evidence indicates population declines. Beyond the communicative value of PAM variables in the contact with fishers, fisheries-independent monitoring with PAM-based reference points can avoid the illusion of plenty associated with hyperstability in the CPUE (Maunder et al. 2006; Erisman et al. 2011).
The CPUE series analysed here indicates a transition in exploitation around 2010, with higher effort and lower relative abundance in more recent years, and this is in line with fisher perceptions in the Tagus estuary (Fig. 2b) and the results reported in an earlier questionnaire survey (Mota 2018). Mean weekly CPUE remains apparently stable since then at a higher exploitation rate, but there are several signs of overexploitation that deserve attention. The continuous erosion of the oceanic catch component (Fig. 6a) and the recent decline in total yield and effort (Fig. 2a and c) indicate that the overexploitation signs are possibly retarded and masked by hyperstability in CPUE (Maunder et al. 2006; Erisman et al. 2011; Sadovy de Mitcheson 2016). Hyperstability can result from fisher behaviour in relation to biological processes operating at different scales. Earlier erosion of the oceanic component in the catch can be related to the shift from bycatch to target during the seasonal migration of adult meagre to the estuary where they concentrate. Apparent stability in CPUE within the estuary, despite declining total catch and effort, can be related with an increasing frequency of the illegal practice to target ephemeral reproductive aggregations. Meagre are repeated batch spawners that create temporary aggregations in the few hours prior to the reproductive event between females with hydrated oocytes and ripe males. In recent years of declining abundance, it is known that some fishers have modified their practices to trace and illegally trap such aggregations (Mota 2018).
In conclusion, the present study provided indications that the meagre fishery in the Tagus estuary needs to move beyond the current phase of minimal technical management measures and consider additional protection of the reproduction in the Tagus estuary. It is also suggested that PAM may serve as a long-term cost-effective instrument towards adaptive fishery management in the Tagus estuary and can facilitate communication with fishers. For that, additional research steps were delimited. Increased participation and communication can help to legitimise tighter input control measures (Oyanedel et al. 2020; Stratoudakis 2021) and improve compliance in a fishery that has grown in the past two decades under limited surveillance and control. Stepping-up fishery monitoring through PAM and introducing effective regulatory measures to protect reproduction through participatory processes is a way to manage other small-scale fisheries for vocal fish that face similar problems of overexploitation and underinvestment.
Change history
01 February 2024
A Correction to this paper has been published: https://doi.org/10.1007/s11160-024-09833-7
References
Aalbers SA, Drawbridge MA (2008) White seabass spawning behavior and sound production. Trans Am Fish Soc 137:542–550. https://doi.org/10.1577/T04-058.1
Aalbers SA, Sepulveda CA (2012) The utility of a long-term acoustic recording system for detecting white seabass Atractoscion nobilis spawning sounds. J Fish Biol 81(6):1859–1870
Almeida R, Mateus CS, Alves MJ, Marques JP, Pereira J, Prista N, Cabral H, Almeida PR, Quintella BR (2022) Evidence of genetic segregation among meagre (Argyrosomus regius) Atlantic spawning areas. J Mar Sci Eng 10:1843. https://doi.org/10.3390/jmse10121843
Alves D, Vieira M, Amorim MCP, Fonseca PJ (2021) Boat noise interferes with Lusitanian toadfish acoustic communication. J Exp Biol 224 (11):jeb234849. https://doi.org/10.1242/jeb.243046
Amorim MCP, Wanjala JA, Vieira M, Bolgan M, Connaughton MA, Pereira BP, Fonseca PJ, Ribeiro F (2023) Detection of invasive fish species with passive acoustics: discriminating between native and non-native sciaenids. Mar Environ Res 188:106017. https://doi.org/10.1016/j.marenvres.2023.106017
Baldaque da Silva AA (1892) Estado actual das pescas em Portugal: comprehendendo a pesca maritima, fluvial e lacustre em todo o continente do reino, referido ao anno de 1886. Imprensa nacional, Lisboa.
Bañon R, Arias A, Arana D, Cuesta JA (2017) Identification of a non-native Cynoscion species (Perciformes: Sciaenidae) from the Gulf of Cádiz (southwestern Spain) and data on its current status. Sci Mar 81(1):19–26. https://doi.org/10.3989/scimar.04494.21A
Batista M, França S, Luís AR, Henriques A, Sá R, Grilo C (2022) Golfinhos no Tejo: por um estuário saudável. ANP|WWF. Online access: https://wwfeu.awsassets.panda.org/downloads/relatorio_golfinhos_no_tejo___por_um_estuario_mais_saudavel_2022.pdf
Biggs RB, Erisman BE (2021) Transmission loss of fish spawning vocalizations and the detection range of passive acoustic sampling in very shallow estuarine environments. Estuaries Coasts 44:2026–2038. https://doi.org/10.1007/s12237-021-00914-5
Blythe J, Nash K, Yates J, Cumming G (2017) Feedbacks as a bridging concept for advancing transdisciplinary sustainability work. Curr Opin Environ Sustain 26–27:114–119. https://doi.org/10.1016/j.cosust.2017.05.004
Bolgan M, Pereira BP, Cruciane A, Mylonas KC, Pousão-Ferreira P, Parmentier E, Fonseca PJ, Amorim MCP (2020) Vocal repertoire and consistency of call features in the meagre Argyrosomous regius (Asso, 1801). PLoS ONE 15(11):e0241792. https://doi.org/10.1371/journal.pone.0241792
Cabral HN, Ohmert B (2001) Diet of juvenile meagre, Argyrosomus regius, within the Tagus estuary. Cah Biol Mar 42:289–293
Castellanos P, Brito AC, Chainho P, Quintella BR, da Costa L, França S, Cabral H, Costa JL (2021) CoastNet dataset from Mondego, Tejo and Mira estuaries: multiparametric measurements during 2020. Front Mar Sci 8:707089. https://doi.org/10.3389/fmars.2021.707089
Castro N, Romão F, Castro JJ, Pereira TJ, Pedro S, Viegas V, Costa JL (2021) Catches, sales and discards: small-scale fisheries in a Portuguese marine park. Reg Stud Mar Sci 42:101643. https://doi.org/10.1016/j.rsma.2021.101643
Cerveira I, Baptista V, Teodósio MA, Morais P (2022) What’s for dinner? Assessing the value of an edible invasive species and outreach actions to promote its consumption. Biol Invasions 24:815–829. https://doi.org/10.1007/s10530-021-02685-3
Coelho P, Carvalho F, Goulding T, Chainho P, Guerreiro J (2021) Management models of the Manila clam (Ruditapes philippinarum) fisheries in invaded European coastal systems. Front Mar Sci 8:685307. https://doi.org/10.3389/fmars.2021.685307
Colla S, Pravoni F, Fiorin R, Malavesi S, Picciulin M (2018) Using passive acoustics to assess habitat selection by the brown meagre Sciaena umbra in a northern Adriatic Sea mussel farm. J Fish Biol 92:1627–1634. https://doi.org/10.1111/jfb.13589
Connaughton MA, Taylor MH (1995) Seasonal and daily cycles in sound production associated with spawning in weakfish, Cynoscion regalis. Environ Biol Fishes 42:233–240
Connaughton MA, Taylor MH (1996) Drumming, courtship, and spawning behaviour in captive weakfish, Cynoscion Regalis. Copeia 1:195–199
Costa MJ, Cabral HN (1999) Changes in the Tagus nursery function for commercial fish species: some perspectives for management. Aquat Ecol 33:287–292
Di Iorio L, Gervaise C, Bonhomme P, Charbonnel E, Michez N, Fouchy K (2020) A guide for the use of passive acoustics to monitor the brown meagre: from measurement to management. MedPAN Collection, 42 p.
Erisman BE, Allen LG, Claisse JT, Pondela DJ II, Miller EF, Murray JH (2011) The illusion of plenty: hyperstability masks collapses in two recreational fisheries that target fish spawning aggregations. Can J Fish Aquat Sci 68:1705–1716. https://doi.org/10.1139/F2011-090
Erisman BE, Aburto-Oropeza O, Gonzalez-Abraham C, Mascareñas-Osorio I, Moreno-Báez M, Hastings PA (2012) Spatio-temporal dynamics of a fish spawning aggregation and its fishery in the Gulf of California. Sci Rep 2:284. https://doi.org/10.1038/srep00284
Erisman BE, Rowell TJ (2017) A sound worth saving: acoustic characteristics of a massive fish spawning aggregation. Biol Let 13:20170656. https://doi.org/10.1098/rsbl.2017.0656
Ferguson GJ, Ward TM, Geddes MC (2008) Do recent age structures and historical catches of mulloway, Argyrosomus japonicus (Temminck & Schlegel, 1843), reflect freshwater inflows in the remnant estuary of the Murray River, South Australia? Aquat Living Resour 21:145–152. https://doi.org/10.1051/alr:2008034
Ferguson GJ, Ward TM, Ivey A, Barnes T (2014) Life history of Argyrosomus japonicus, a large sciaenid at the southern part of its global distribution: implications for fisheries management. Fish Res 151:148–157. https://doi.org/10.1016/j.fishres.2013.11.002
Fitzhugh GR, Thompson BA, Snider TG III (1993) Ovarian development, fecundity and spawning frequency of the red drum Pogonias chromis in Louisiana. Fish Bull 91:244–253
Gabriel S, Prista N, Costa MJ (2012) Estimating meagre (Argyrosomus regius) size from otoliths and vertebrae. Journal of Archaeological Sciences 39:2859–2865. https://doi.org/10.1016/j.jas.2012.04.046
Gannon DP (2008) Passive acoustic techniques in fisheries sciences: a review and prospectus. Trans Am Fish Soc 137:638–656. https://doi.org/10.1577/T04-142.1
Gannon DP, Gannon JG (2010) Assessing trends in the density of Atlantic croaker (Micropogonias undulatus): a comparison of passive acoustic and trawl methods. Fish Bull 108:106–116
Gomes P, Vieira AR, Oliveira R, Silva H, Martins R, Carneiro M (2017) First record of Cynoscion regalis (Pisces, Sciaenidae) in Portuguese continental waters. J Fish Biol 90:2470–2474. https://doi.org/10.1111/jfb.13318
González-Quirós R, del Árbol J, del Mar G-P, Silva-García AJ, Naranjo JM, Morales-Nin B (2011) Life-history of the meagre Argyrosomus regius in the Gulf of Cádiz (SW Iberian Peninsula). Fish Res 109:140–149. https://doi.org/10.1016/j.fishres.2011.01.031
Griffiths MH (1996) Life history of the Dusky kob Argyrosomus japonicus (Sciaenidae) off the east coast of South Africa. S Afr J Mar Sci 17:135–154
Gueroun SKM, Torres TM, Dos Santos A, Rodrigues NV, Canning-Clode J, Andrade C (2021) Catostylus tagi (Class: Scyphozoa, Order: Discomedusae, Suborder: Rhizostomida, Family: Catostylidae) life cycle and first insight into its ecology. PeerJ 9:e12056. https://doi.org/10.7717/peerj.12056
Haffray P, Malha R, Sidi MOT, Prista N, Hassan M, Castelnaud G et al (2012) Very high genetic fragmentation in a large marine fish, the meagre Argyrosomus regius (Sciaenidae, Perciformes): impact of reproductive migration, oceanographic barriers and ecological factors. Aquat Living Resour 25(2):173–183. https://doi.org/10.1051/alr/2012016
Heyman WD, Grüss A, Biggs CR, Kobara S, Farmer NA, Karnauskas M, Lowerre-Barbieri S, Erisman B (2019) Cooperative monitoring, assessment, and management of fish spawning aggregations and associated fisheries in the U.S. Gulf of Mexico. Mar Policy 109:103689. https://doi.org/10.1016/j.marpol.2019.103689
Holt GL, Holt SA, Arnold CR (1985) Diel periodicity of spawning in sciaenids. Mar Ecol Prog Ser 27:1–7
Howe BM, Miksis-Olds J, Rehm E, Sagen H, Worcester PF, Haralabus G (2019) Observing the oceans acoustically. Front Mar Sci 6:426. https://doi.org/10.3389/fmars.2019.00426
Hurbans B, Chouvelon T, Begout ML, Biais G, Bustamante P, Ducci L, Mornet F, Boiron A, Coupeau Y, Spitz J (2017) Tropic ecology of commercial-size meagre, Argyrosomous regius, in the Bay of Biscay (NE Atlantic). Aquat Living Resour 30:9. https://doi.org/10.1051/alr/2017004
Koenig CC, Bueno LS, Coleman FC, Cusick JA, Ellis RD, Kingon K et al (2017) (2017) Diel, lunar, and seasonal spawning patterns of the Atlantic goliath grouper, Epinephelus itajara, off Florida United States. Bull Mar Sci 93(2):391–406. https://doi.org/10.5343/bms.2016.1013
Lagardére JP, Mariani A (2006) Spawning sounds in meagre Argyrosomus regius recorded in the Gironde estuary, France. J Fish Biol 69:1697–1708. https://doi.org/10.1111/j.1095-8649.2006.01237.x
Locascio JV, Mann DA (2011) Diel and seasonal timing of sound production by black drum (Pogonias cromis). Fish Bull 109(3):327–338
Locascio JV, Burghart S, Mann DA (2012) Quantitative and temporal relationships of egg production and sound production by black drum Pogonias cromis. J Fish Biol 81:1175–1191. https://doi.org/10.1111/j.1095-8649.2012.03376.x
Lowerre-Barbieri S, Barbieri L, Flanders JR, Woodward A, Cotton C, Knowlton K (2008) Using passive acoustics to determine red drum spawning in Georgia Waters. Trans Am Fish Soc 137:562–575
Lowerre-Barbieri S, Walters Burnsed SL, Bickford JW (2016) Assessing reproductive behavior important to fisheries management: a case study with red drum, Sciaenops Ocellatus. Ecol Appl 26(4):979–995. https://doi.org/10.5061/dryad.47bs5
Luczkovich JJ, Sprague MW, Johnson SE, Pullinger RC (1999) Delimiting spawning areas of weakfish, Cynoscion regalis (family Sciaenidae) in Pamlico Sound, North Carolina using passive hydroacoustic surveys. Bioacoustics 10:143–160. https://doi.org/10.1080/09524622.1999.9753427
Luczkovich JJ, Mann DA, Rentree RA (2008a) Passive acoustics as a tool in fisheries science. Trans Am Fish Soc 137:533–541. https://doi.org/10.1577/T06-258.1
Luczkovich JJ, Pullinger RC, Johnson SE, Sprague MW (2008b) Identifying sciaenid critical spawning habitats by the use of passive acoustics. Trans Am Fish Soc 137:576–605. https://doi.org/10.1577/T05-290.1
Macchi GJ, Acha EM, Lasta CA (2002) Reproduction of black drum (Pogonias cromis) in the Rio de la Plata estuary, Argentina. Fish Res 59:83–92
Mann DA, Grothues TM (2009) Short-term upwelling events modulate fish sound production at a mid-Atlantic ocean observatory. Mar Ecol Prog Ser 375:65–71. https://doi.org/10.3354/meps07720
Marques TA, Thomas L, Martin SW, Mellinger DK, Ward JA, Moretti DJ, Harris D, Tyack PL (2013) Estimating animal population density using passive acoustics. Biol Rev Cambridge Philos Soc 88:287–309. https://doi.org/10.1111/brv.12001
Maunder MN, Sibert JR, Fonteneau A, Hampton J, Kleiber P, Harley SJ (2006) Interpreting catch per unit effort data to assess the status of individual stocks and communities. ICES J Mar Sci 63:1363–1385
Merchant ND, Fristrup KM, Johnson MP, Tyack PL, Witt MJ, Blondel P, Parks SE (2015) Measuring acoustic habitats. Methods Ecol Evol 6(3):257–265. https://doi.org/10.1111/2041-210X.12330
Mirimin L, Macey B, Kerwath S, Laberth S, Bester-van der Merwe A, Cowley P et al (2016) Genetic analyses reveal declining trends and low effective population size in an overfished South African sciaenid species, the dusky kob (Argyrosomus japonicus). Mar Freshw Res 67:266–276. https://doi.org/10.1071/MF14345
Monczak A, Berry A, Kehrer C, Montie EW (2017) Long-term acoustic monitoring of fish calling provides baseline estimates of reproductive timelines in the May River estuary, southeastern USA. Mar Ecol Prog Ser 581:1–19
Monczak A, Ji Y, Soueidan J, Montie EW (2019) Automatic detection, classification, and quantification of sciaenid fish calls in an estuarine soundscape in the Southeast United States. PLoS ONE 14:e0209914. https://doi.org/10.1371/journal.pone.0209914
Monczak A, McKinney B, Souiedan J, Marian AD, Seder A, May E, Morgenstern T, Roumillat W, Montie EW (2022) Sciaenid courtship sounds correlate with juvenile appearance and abundance in the May River, South Carolina, USA. Mar Ecol Prog Ser 693:1–17. https://doi.org/10.3354/meps14109
Montie EW, Vega S, Powell M (2015) Seasonal and spatial patterns of fish sound production in the May River, South Carolina. Trans Am Fish Soc 144:705–716. https://doi.org/10.1080/00028487.2015.1037014
Montie EW, Kehrer C, Yost J, Brenkert K, O’Donnell T, Denson MR (2016) Long-term monitoring of captive red drum Sciaenops ocellatus reveals that calling incidence and structure correlate with egg deposition. J Fish Biol 88:1776–1795. https://doi.org/10.1111/jfb.12938
Montie EW, Hoover M, Kehrer C, Yost J, Brenkert K, O’Donnell T, Denson MR (2017) Acoustic monitoring indicates a correlation between calling and spawning in captive spotted seatrout (Cynoscion nebulosus). PeerJ 5:e2944. https://doi.org/10.7717/peerj.2944
Morais P, Cerveira I, Teodósio MA (2017) An update on the invasion of weakfish Cynoscion regalis (Bloch & Schneider, 1801)(Actinopterygii: Sciaenidae) into Europe. Diversity 9(4):47. https://doi.org/10.3390/d9040047
Morales-Nin B, Geffen AJ, Pérez-Mayol S, Palmer M, González-Quirós R, Grau A (2012) Seasonal and ontogenic migrations of meagre (Argyrosomus regius) determined by otolith geochemical signatures. Fish Res 127–128:154–165. https://doi.org/10.1016/j.fishres.2012.02.012
Mota A (2018) Pescarias de corvina-legítima na região do estuário do Tejo: caraterização das componentes lúdica e comercial. MSc Thesis in Marine Ecology, University of Lisbon. http://hdl.handle.net/10451/36284
Mueller C, Monczak A, Soueidan J, McKinney B, Smott S, Mills T et al (2020) Sound characterization and fine-scale spatial mapping of an estuarine soundscape in the southeastern USA. Mar Ecol Prog Ser 645:1–23. https://doi.org/10.3354/meps13373
Oyanedel R, Gelchich S, Milner-Gulland EJ (2020) Motivations for (non-)compliance with conservation rules by small-scale resource users. Conserv Lett 2020:e12725. https://doi.org/10.1111/conl.12725
Parmentier E, Di Iorio L, Picciulin M, Malavasi S, Lagardère JP, Bertucci F (2018) Consistency of spatiotemporal sound features supports the use of passive acoustics for long-term monitoring. Anim Conserv 21:211–220. https://doi.org/10.1111/acv.12362
Pereira E, Quintella BR, Lança MJ, Alexandre CM, Mateus CS, Pedro S et al (2021) Temporal patterns of the catadromous thinlp grey mullet migration in freshwater. Ecohydrology 14:e2345. https://doi.org/10.1002/eco.2345
Potts WM, Sauer WHH, Henriques R, Sequesseque S, Santos CV, Shaw PW (2010) The biology, life history and management needs of a large sciaenid fish, Argyrosomus coronus, in Angola. Afr J Mar Sci 32:247–258. https://doi.org/10.2989/1814232X.2010.501567
Prista N (2013) Argyrosomus regius (Asso, 1801) fishery and ecology in Portuguese waters, with reference to its relationships to other European and African populations. PhD Thesis, University of Lisbon, Lisbon. https://repositorio.ul.pt/handle/10451/10817
Prista N, Diawara N, Costa MJ, Jones CM (2011) Use of SARIMA models to assess data-poor fisheries: a case study with a sciaenid fishery off Portugal. Fish Bull 109:170–185
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Raimundo J, Pereira P, Caetano M, Cabrita T, Vale C (2011) Decrease of Zn, Cd and Pb concentrations in marine fish species over a decade as response to reduction of anthropogenic inputs: the example of Tagus estuary. Mar Pollut Bull 62:2854–2858. https://doi.org/10.1016/j.marpolbul.2011.09.020
Ramcharitar J, Gannon DP, Popper AN (2006) Bioacoustics of fishes of the family Sciaenidae (croakers and drums). Trans Am Fish Soc 135:1409–1431. https://doi.org/10.1577/T05-207.1
Ricci SW, Eggleston DB, Bohnenstiehl DWR (2017) Use of passive acoustic monitoring to characterize fish spawning behavior and habitat use within a complex mosaic of estuarine habitats. Bull Mar Sci 93(2):439–453. https://doi.org/10.5343/bms.2016.1037
Rodrigues M, Cravo A, Freire P, Rosa A, Santos D (2020) Temporal assessment of the water quality along an urban estuary (Tagus estuary, Portugal). Mar Chem 223:103826. https://doi.org/10.1016/j.marchem.2020.103824
Rowe S, Hutchins J (2003) Mating systems and the conservation of commercially exploited marine fish. Trends Ecol Evol 18(11):567–572
Rowell TJ, Schärer MT, Appeldoorn RS, Nemeth MI, Mann DA, Rivera JA (2012) Sound production as an indicator of red hind density at a spawning aggregation. Mar Ecol Prog Ser 462:241–250. https://doi.org/10.3354/meps09839
Rowell TJ, Demer DA, Aburto-Oropeza O, Cota-Nieto JJ, Hyde JR, Erisman BE (2017) Estimating fish abundance at spawning aggregations from courtship sound levels. Sci Rep 7(1):1–14. https://doi.org/10.1038/s41598-017-03383-8
Sadovy de Mitcheson Y (2016) Mainstreaming fish spawning aggregations into fisheries management calls for a precautionary approach. Bioscience 66:295–306. https://doi.org/10.1093/biosci/biw013
Sadovy de Mitcheson Y, To AW, Wong NW, Kwan HY, Bud WS (2019) Emerging from the murk: threats, challenges and opportunities from the global swim-bladder trade. Rev Fish Biol Fisheries 29:809–835. https://doi.org/10.1007/s11160-019-09585-9
Saucier M, Baltz D (1993) Spawning site selection by spotted seatrout, Cynoscion nebulosus, and black drum, Pogonias cromis, in Louisiana. Environ Biol Fishes 36:257–272
Silberschneider V, Gray CA (2008) Synopsis of biological, fisheries and aquaculture-related information on mulloway Argyrosomus japonicus (Pisces: Sciaenidae), with particular reference to Australia. J Appl Ichthyol 24:7–17. https://doi.org/10.1111/j.1439-0426.2007.00913.x
Smith HM (1905) The drumming of the drum-fishes (Sciaenidae). Science 376–378. https://doi.org/10.1126/science.22.560.376
Souza PM Jr, Olsen Z, Brandl SJ (2023) Paired passive acoustic and gillnet sampling reveal the utility of bioacoustics for monitoring fish populations in a turbid estuary. ICES J Mar Sci 80:1240–1255. https://doi.org/10.1093/icesjms/fsad085
Stratoudakis Y, Bernal M, Ganias K, Uriarte A (2006) The daily egg production method: recent advances, current applications and future challenges. Fish Fish 7:35–57
Stratoudakis Y (2021) Legitimacy objects, agents, and evaluators: a framing to strengthen planning and performance of fisheries co-management. Environ Sci Policy 115:230–238. https://doi.org/10.1016/j.envsci.2020.11.01
Stratoudakis Y, Antunes C, Correia C, Belo F, Almeida PR (2023) Riverine communities and management systems for anadromous fisheries in the Iberian Peninsula: global strategy, local realities. Rev Fish Biol Fisheries 33:875–892. https://doi.org/10.1007/s11160-022-09742-7
Shephard S, Greenstreet SPR, Piet GJ, Rindorf A, Dickey-Collas M (2015) Surveillance indicators and their use in implementation of the Marine Strategy Framework Directive. ICES J Mar Sci 72(8):2269–2277. https://doi.org/10.1093/icesjms/fsv131
Venables WN, Ripley BD (2002) Modern applied statistics with S, 4th edn. Springer, New York
Vieira M, Fonseca PJ, Amorim MCP, Teixeira CJ (2015) Call recognition and individual identification of fish vocalizations based on automatic speech recognition: an example with the Lusitanian toadfish. J Acoust Soc Am 138(6):3941–3950. https://doi.org/10.1121/1.4936858
Vieira M, Pereira BP, Pousão-Ferreira P, Fonseca PJ, Amorim M (2019) Seasonal variation of captive meagre acoustic signalling: a manual and automatic recognition approach. Fishes 4(2):28. https://doi.org/10.3390/fishes4020028
Vieira M, Fonseca PJ, Amorim MCP (2021) Fish sounds and boat noise are prominent soundscape contributors in an urban European estuary. Mar Pollut Bull 172:112845. https://doi.org/10.1016/j.marpolbul.2021.112845
Vieira M, Amorim MCP, Marques TA, Fonseca PJ (2022) Temperature mediates chorusing behaviour associated with spawning in the sciaenid Argyrosomus regius. Mar Ecol Prog Ser 697:109–124. https://doi.org/10.3354/meps14128
Walters S, Lowerre-Barbieri S, Bickford J, Mann D (2009) Using a passive acoustic survey to identify spotted seatrout spawning sites and associated habitat in Tampa Bay, Florida. Trans Am Fish Soc 138:88–98. https://doi.org/10.1577/T07-106.1
Wood SN, Augustin N (2002) GAMs with integrated model selection using penalized regression splines and applications to environmental modelling. Ecol Model 157:157–177. https://doi.org/10.1016/S0304-3800(02)00193-X
Young S, Evermann G, Gales M (2006) The HTK Book Version 3.4. Cambridge University Press, Cambridge, UK.
Acknowledgements
We thank DGRM for providing daily commercial landing data in mainland Portugal, Air Force Base No 6 (Montijo, Portugal) for allowing the field study of PAM in their military establishment and Tagus´ Fisher Associations and fishers for participating in meetings and the questionnaire survey. We also thank Monica Mesquita (FCT/UNL and MARE/OLO) for creating initial conditions to establish contacts with local fishers and Ivone Figueiredo (IPMA) and the project PPCENTRO for contributing to the rationale of the approach. This study was supported by National Funds through the Foundation for Science and Technology (FCT) via the project “MIGRACORV-Integrated approach to study the movement dynamics of the meagre Argyrosomus regius” (PTDC/BIA-BMA/30517/2017), the strategic projects UIDB/04292/2020 awarded to MARE and UID/BIA/00329/2020 to cE3c and the project LA/P/0069/2020 granted to the Associate Laboratory ARNET. FCT individually funded BRQ through the Scientific Employment Stimulus Program (2020.02413.CEECIND) and MV and JPM through Doctoral grants (SFRH/BD/115562/2016; 2020/06801.BD).
Funding
Open access funding provided by FCT|FCCN (b-on).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No financial or other interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
“The original online version of this article was revised”: the author name “Stratoudakis” has been corrected in XML.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stratoudakis, Y., Vieira, M., Marques, J.P. et al. Long-Term Passive Acoustic Monitoring to Support Adaptive Management in a Sciaenid Fishery (Tagus Estuary, Portugal). Rev Fish Biol Fisheries 34, 491–510 (2024). https://doi.org/10.1007/s11160-023-09825-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11160-023-09825-z