Abstract
The use of underwater video techniques has expanded rapidly in ecological studies and is particularly desirable in protected areas since the method does not impact the habitat or remove fish. The Mediterranean Sea is a biodiversity hotspot under high anthropogenic pressure and consequently, non-destructive and non-extractive techniques for fish monitoring are advantageous. Here, we review 110 publications that used underwater video in fish-related studies in the Mediterranean basin. The most common technique used in the Mediterranean Sea was Remotely Operated Vehicles (ROV) (33%), followed by Remote Underwater Video (RUV) systems (20%), Diver Operated Video (DOV) systems (20%) and Baited Remote Underwater Video (BRUV) systems (19%). Approximately one third of the studies used an additional sampling method, such as fisheries-based or molecular methods with the aim to compare the surveying effectiveness or produce complementary data. The most frequent objectives of the reviewed studies were related to fish community structure, i.e., focusing on community wide metrics such as abundance and biodiversity, or behavioral analyses, while the most commonly studied environments were those of the western Mediterranean and shallow waters, usually involving sandy or rocky reef habitats. Sampling protocols differed widely among studies with transect lengths, soak times and baits all varying. Future research should focus on the least studied parts of the region, such as the eastern and southern Mediterranean Sea and deep-sea habitats. Finally, the development of standardized sampling protocols is recommended to ensure that data are comparable among studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fish monitoring underpins conservation and management and is particularly critical given the declining biomass trends that characterize many commercial fish species worldwide (Costello et al. 2012; Palomares et al. 2020). Fish monitoring can utilize either fishery-dependent (commercial or recreational) or fishery-independent data. Fishery-independent techniques include experimental fishing (Priester et al. 2021), remote sensing (Santos et al. 2000), acoustic surveys (Rountree et al. 2006), telemetry (Hammerschlag et al. 2011), underwater visual census (Samoilys and Carlos 2000), genetics (Blower et al. 2012) and underwater video (Mallet and Pelletier 2014). The use of non-destructive and non-extractive techniques, such as those based on video, is particularly desirable in Marine Protected Areas (MPAs) in order to maintain the integrity of the ecosystem and avoid placing further pressure on fish populations (Collie et al. 2000; Sciberras et al. 2018; Murphy and Jenkins 2010). Underwater video use in ecological studies mainly aims to monitor fish populations (Marra et al. 2016), study the behavior of fish (Ajemian et al. 2016) or develop and compare methodologies (Stobart et al. 2007).
The first underwater video system for biological studies was described in 1952 (Barnes 1952) and since then, underwater video has become an increasingly popular ecological tool due to its versatility and the technological advances during the last two decades (Mallet and Pelletier 2014). In the Mediterranean Sea, the first underwater video study was conducted in late 1970s (Fedra and Machan 1979) with a rapid increase in the use of underwater video after 2000. Most underwater video techniques, in contrast to Underwater Visual Census (UVC), enable scientists to sample without time and depth limitations and reach locations inaccessible to divers (Cappo et al. 2007; Unsworth et al. 2014). Video-based methods provide a permanent record of standardized replicates with detailed optical view, which minimizes observer bias regarding species identification, fish length estimates and sample unit area (Langlois et al. 2010; Unsworth et al. 2014). Moreover, sampling can be performed by non-taxonomy experts and video footage can be examined several times and by different observers in the lab (Langlois et al. 2010). Contrary to fisheries-based methods and experimental fishing, video techniques also provide information on habitat (Collins et al. 2017) and animal behavior (Ayma et al. 2016) and are less selective in terms of species and sizes (Murphy and Jenkins 2010). The use of video techniques is however constrained under poor visibility conditions (Sward et al. 2019) and the presence of bait in baited camera systems may introduce unnatural behaviours (Harvey et al. 2007). When bait is used, the area extent from which fish are drawn is unknown and thus, abundance is relative rather than a real estimate (Heagney et al. 2007).
Underwater video-based fish studies can utilize a range of methodologies, depending on the study objectives and available resources. Underwater video equipment can be permanently or temporarily deployed, in shallow water or abyssal depths, powered by batteries or cable, stationary or deployed along transects, and target pelagic or benthic habitats. Systems may be deployed with single cameras or in stereo-configuration to allow length measurements. Worldwide, commonly used methods include the remote underwater video technique (RUV), baited remote underwater video (BRUV) systems, remotely operated vehicle (ROV), diver operated video (DOV) systems and towed video (TOWV) Techniques.
Remote underwater video (RUV) systems consist of a video recording device being placed underwater, on the seafloor or in the water column, and do not require human presence to operate. The systems can be set by a diver or deployed from a vessel (Mallet and Pelletier 2014) and can be either linked to a monitoring station by cable (Aguzzi et al. 2011), usually used for long term monitoring, or deployed autonomously operating by battery power (Galasso et al. 2015). Bait can be placed optionally in front of the mounted camera(s) creating an odor plume in order to attract organisms from a broader area, in which case, the systems are referred to as Baited Remote Underwater Video (BRUV) systems (Cappo et al. 2003). Fish monitoring studies increasingly use BRUV systems (Bailey et al. 2007), especially in Australia (Mallet & Pelletier 2014; Whitmarsh et al. 2017; Harvey et al. 2021) and their use is strongly recommended for mobile and predatory species (Cappo et al. 2003). Similar to RUV, BRUV systems can be either linked to an onboard personal computer to provide real time underwater footage (D’Onghia et al. 2015a) or autonomously function underwater without cables (Torres et al. 2020).
Remotely Operated Vehicle (ROV) is an unmanned underwater submersible operated from the surface. ROVs are available in various sizes and can perform at different depths (Sward et al. 2019). They can carry a large range of tools, such as jaw grabbers, multiple cameras, laser beams and multiparametric probes (Bo et al. 2012a; D’Onghia et al. 2015a). The main advantage of ROVs is that they can survey at deep waters and cover large areas (Sward et al. 2019). However, careful consideration is needed as to the appropriateness of the technique as certain fish species can react negatively to ROV presence (Ayma et al. 2016). Another limiting factor for ROVs use in fish studies is their high cost (Sward et al. 2019) but during the last decade, market demand has led to the creation of mini-ROVs, tools that are low-cost, reliable and easy to operate (Rubin 2013).
Diver Operated Video (DOV) systems is a form of underwater visual census where the transect is filmed by a diver. DOV surveys can be conducted along predesigned transects (Marra et al. 2016), by stationary point count (Mascolino et al. 2019) or by following the focal organisms (Goverts et al. 2021). The main advantage of DOV systems is that the diver-operated camera can be maneuvered within inaccessible areas such as overhangs, and thus record cryptic species (Watson et al. 2005). Limitations of DOV techniques include their constraint to relatively shallow depths and the impacts of the diver’s presence on fish (Langlois et al. 2010).
Towed video (TOWV) systems are survey tools consisting of an underwater camera attached on a frame linked to a vessel such that the frame is drawn behind the vessel, either in the form of a sled (Spencer et al. 2005) or a structure hovering above the seafloor (Barker et al. 1999). The main advantage of a TOWV system is its suitability for high current areas as it is driven by the vessel’s horsepower (Barker et al. 1999). A disadvantage is that a sled’s operation is challenging on rocky and irregular substrates (Rooper 2008).
In the Mediterranean Sea, all these methods have been used. In addition to applications to fish ecology and monitoring, video recording techniques have also been used for commercial purposes in aquaculture and fisheries. Aquaculture studies have used such techniques to estimate abundance and biomass of bluefin tuna (Costa et al. 2009; Mariani et al. 2014) or behavioral changes of captive fish under different conditions (Sarà et al. 2007; 2010). Fisheries studies use video techniques to estimate the catchability of fishing gears (Dremière et al. 1999; Papadopoulou et al. 2015) and record the effect fish aggregating devices can have on fish community structure (Addis et al. 2016) or fish behavior (Sinopoli et al. 2015). During the last years, sea users are also contributing to ecological fish knowledge by producing public amateur underwater videos. Fish videos by recreational fishermen or scuba divers often help scientists identify the appearance of non-indigenous fish for the first time (Stern et al. 2019), track their expanding distribution in the Mediterranean (Deidun et al. 2011) or provide information about the spatio-temporal distribution of important species, such as groupers (Sbragaglia et al. 2021).
This review focuses on underwater video techniques used in the Mediterranean Sea, which have been either primarily or secondarily used to study marine fishes. Underwater video techniques have a promising future in the study of fish given their ongoing evolution in performance and their non-destructive and non-extractive nature, making their review timely. The Mediterranean Sea hosts over 17,000 marine species (Coll et al. 2010), 750 of which are fishes (Dimarchopoulou et al. 2017), and has been highly impacted by anthropogenic activities over the last decades (Halpern et al. 2008; Micheli et al. 2013). Fisheries (Piroddi et al. 2020), climate change (Barnett et al. 2001) and invasive species (Katsanevakis et al. 2014) have all contributed to the declining health of the Mediterranean Sea by leading directly and indirectly to substantial declines in species richness, diversity, density and biomass levels of both vertebrates and invertebrates (Claudet & Fraschetti 2010; Coll et al. 2010; Prato et al. 2013). This is particularly the case for species targeted by fisheries, the majority of them being overexploited (Tsikliras et al. 2015). Reflecting the need to better understand the fish of the Mediterranean, this review also provides recommendations for fish monitoring.
Materials and methods
Literature survey and data selection
Searches in Google Scholar and Scopus were conducted for all articles published prior to 31 December 2021. We used the keyword string “ROV or RUV or VIDEO or DOV or TOWV or BRUV or RECORDING and MEDITERRANEAN and FISH”. The first 1000 findings from Google Scholar were viewed, while all results from the extended field of natural sciences in Scopus were viewed, including the results “Agricultural and Biological Sciences”, “Biochemistry”, “Genetics and Molecular Biology”, “Environmental Science”, “Earth and Planetary Sciences”, “Engineering”, “Multidisciplinary”, “Computer Science”, “Physics and Astronomy”, “Chemistry”, “Mathematics”, “Chemical Engineering”, “Energy”, “Veterinary”. We included only publications in peer reviewed journals and conference proceedings in the English language. Materials in the grey literature (i.e., technical reports, theses etc.) were excluded from the analysis. With the above selection criteria, 110 publications were gathered (102 peer reviewed journal articles and 8 conference papers) and were further reviewed. Included studies were strictly limited to marine waters, the use of underwater video recording techniques and pertained to fish, both as their main researched organisms and as unintentional occurrences in another research framework. The reviewed studies were also carried out exclusively by scientists and were held in field environments. Therefore, citizen science studies and those in the fields of aquaculture and fisheries were not included.
Consistent information was recorded for each included study, including the year, the study objective, the studied organisms, the sampling site and technique and the data analysis. Publications were assigned to categories according to the study objective: (1) studies related to fish behavior (“behavioral”); (2) studies focusing on community analysis and community wide metrics such as abundance and biodiversity (“community related”); (3) studies making comparisons between different techniques and describing new methods (“methodological”); and (4) studies with main focus usually on benthos and not fish, but mentioning fish species in the results as species associated with the studied system (“auxiliary”). Category 3 included only studies that described a new technique rather than studies using a modified technique. For each publication, the focal study organisms were recorded and assigned to one of the following categories: (1) single fish species; (2) groups of fish species; (3) the entire fish community; or (4) other organisms such as benthic invertebrates. The information extracted from the videos was noted, including, for instance, abundance, biomass and fish size. The habitat type was also noted based on the substrates included in the sampling regions. Habitat types included coralligenous habitat, coral habitat (alive, dead or rubble), rocky reef or algae covered rock, seagrass, artificial substrate and grains. Grains were further categorized by increasing grain sizes and are hereafter referred in the text as “sand” (clay/silt/mud/sand), “gravel” (gravel/pebble/cobble) and “bare rock” (boulder/bare rock). Artificial substrates included artificial reef, hard substrates on pillars of gas platforms or power stations and shipwrecks. The depths at which the video recording systems were deployed in each publication were recorded as well.
The country, division, and geographical subarea (GSA) were recorded for each study according to the sampling locations. The divisions included the ecoregions western Mediterranean, central Mediterranean, Adriatic Sea and eastern Mediterranean (ICES, www.ices.dk). The GSAs were assigned according to Food and Agriculture Organization (FAO, fao.org) areas: northern Alboran Sea (GSA 1), Alboran island (GSA 2), southern Alboran Sea (GSA 3), Algeria (GSA 4), Balearic islands (GSA 5), northern Spain (GSA 6), Gulf of Lion (GSA 7), Corsica (GSA 8), Ligurian Sea and Northern Tyrrhenian Sea (GSA 9), southern and central Tyrrhenian Sea (GSA 10), Sardinia (GSA 11), northern Tunisia (GSA 12), Gulf of Hammamet (GSA 13), Gulf of Gabes (GSA 14), Malta (GSA 15), southern Sicily (GSA 16), northern Adriatic Sea (GSA 17), southern Adriatic Sea (GSA 18), western Ionian Sea (GSA 19), eastern Ionian Sea (GSA 20), southern Ionian Sea (GSA 21), Aegean Sea (GSA 22), Crete (GSA 23), northern Levant Sea (GSA 24), Cyprus (GSA 25), southern Levant Sea (GSA 26), and eastern Levant Sea (GSA 27).
Sampling technique was assigned as one of 5 categories: (1) “RUV” included all studies in which a recording device was fixed underwater; (2) “BRUV” was assigned to all baited RUV studies; (3) “ROV” included studies using remotely operated video; (4) “DOV” included those studies that used diver-held underwater cameras to record; and (5) “Other” category consisted of TOWV, manned submersible, techniques that did not fall in any of the other descriptions e.g. camera fixed on a kayak recording in transects or camera fixed on a trawl, and all other sampling methods that were not clearly described. RUV and BRUV techniques ranged from custom-made structures equipped with cameras (Torres et al. 2020) to benthic landers (Capezzuto et al. 2012). Lastly, information on additional sampling methods apart from the video techniques, such as nets and longline was recorded.
Percentages in the results section do not sum up to 100% since most studies were conducted in multiple areas and countries, covered multiple habitats, used more than one sampling gear etc.
Results
Year and geographical characteristics
The first underwater video study in the Mediterranean was published in 1979 (Fig. 1). A 20-year research gap followed with little to no use of video-based methods. From approximately 2010 and onwards, the use of video techniques in fish research has increased rapidly, with more than half of all studies published since 2016 (Fig. 1). Across 110 publications 115 records of video techniques were identified. Most studies used ROV (33%) while RUV, BRUV and DOV systems were almost equally used in around 20% of studies each. In a small number of studies (8%) the description of the video technique was inadequate and thus, could not be assigned to a specific group. Articles using manned submersible vehicles and TOWV techniques were published once and twice respectively.
The number of underwater video studies in the Mediterranean Sea for each of the identified techniques published prior to 31 December 2021. Underwater video techniques include Baited Remote Underwater Video (BRUV) and Remote Underwater Video (RUV) systems, Remote Operated Vehicle (ROV), Diver Operated Video (DOV) systems, and other (techniques not included in any of the above groups)
The most studied area in the Mediterranean Sea using underwater video techniques was the western part of the basin, surveyed in 63% of the reviewed publications (Fig. 2). Specifically, almost half (49%) of the studies were conducted in three GSAs, all located in the western Mediterranean: northern Spain (GSA 6), southern and central Tyrrhenian (GSA 10) and Balearic Islands (GSA 5). The central Mediterranean and Adriatic Sea were the geographic locations of 19 and 16% of the studies, respectively, while only 7% of the studies were conducted in the eastern Mediterranean Sea (Fig. 2). The least studied area was the southern part of the Mediterranean basin with no studies being conducted in the Alboran Island (GSA 2), Algeria (GSA 4), northern Tunisia (GSA 12), Gulf of Gabes (GSA 14), Malta (GSA 15) and southern Ionian Sea (GSA 21). By country, the majority of the studies included Italy (49%), Spain (28%) and France (11%).
Distribution of underwater video publications in the Mediterranean Sea per Geographical Subarea (GSAs) and frequency of underwater video techniques per Mediterranean division as indicated by the pies. Circle sizes indicate proportionally the number of studies in each Mediterranean division. Techniques include Baited Remote Underwater Video (BRUV), Remote Underwater Video (RUV), Remote Operated Vehicle (ROV), Diver Operated Video (DOV), other (techniques not included in any of the above groups). Numbers show the geographical subareas (GSAs) according to FAO (see Materials and Methods)
Approximately one third of the publications (35%) stated that they were conducted within MPAs, while four studies took place in locations that were candidates to become MPAs at the time. The rest of the publications did not specify whether they were conducted in protected areas or not, however the possibility of more studies being conducted in MPAs but failing to mention it cannot be ruled out. The prevailing sampling methods used in MPAs were RUV (37%), DOV (26%) and BRUV (21%) systems. In MPAs, many studies using underwater video explored the protection effect on fish populations and fish behavior. However, the use of MPAs as sampling sites has not been restricted to protection effect studies. Populational changes on spatial and temporal level and habitat use based on the season were also questions addressed within MPAs. Lastly, biodiversity characterization has taken place in regions candidates for MPAs (Table S1 in Supplementary Material).
Depth & habitat
The areas that were mostly covered (more than 50%) by underwater video methods were shallow waters of less than 500 m depth. The maximum depth at which video systems were deployed was greater than 5000 m depth (Fig. 3A). Studies using DOV and RUV systems were restricted to shallow depths (Fig. 3B) up to 62 and 45 m, respectively (Fig. 3B). All other video systems sampled a wider range of depths from shallow waters (0–20 m) down to the bathypelagic (ROV) or even the abyssopelagic zone (BRUV).
a Frequency of underwater video uses in the Mediterranean Sea by depth and b Depth distribution for each video technique. Underwater video techniques include Baited Remote Underwater Video (BRUV), Remote Underwater Video (RUV), Remoted Operated Vehicle (ROV), Diver Operated Video (DOV), other (techniques not included in any of the above groups)
In most studies (93%), video techniques were used to study fish close to the seafloor, as the camera systems were set on the seafloor (RUV, BRUV), operated on the seafloor (TOWV) or above it (DOV, ROV) usually at around 1.5 m from the bottom. Exceptions were specific studies in which the camera was set in the water column to monitor pelagic predators, set at a depth gradient along fish farm cages or under the bow of a kayak in a study of fish availability as prey for birds. Also, in certain DOV and ROV studies, divers and ROVs deviated from the seafloor to perform the survey by following the distribution pattern of the studied fish.
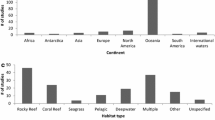
Information regarding the substrate type was also recorded. Many of the publications (45%) included multiple habitats. Overall, across 110 publications 174 habitat records were identified. The most common habitats appearing were sand (40%), rocky reef (32%), bare rock (23%) and seagrass (22%) (Fig. 4). The rest of habitats included coral ecosystems (15%), artificial substrates (14%), coralligenous habitats (8%) and gravel (5%). Studies carried out in regions consisting exclusively of a single habitat included rocky reefs (14%), seagrass meadows and artificial substrates (7% each) (Fig. 4).
Objectives
The objective of the studies utilizing video recording techniques varied widely (Table S1 in Supplementary Material) with some studies having multiple ones. In total 126 objective records were identified. Most studied topics were at fish community level (44%) focusing mainly on differences in community wide metrics, such as abundance. ROV (31%) and BRUV systems (29%) were the most popular techniques in community analysis studies, followed by DOV systems (21%). Secondary to this objective were behavioral studies (34%) focusing on feeding or mating behaviour for example. Differences in community wide metrics or behavior were tested against season, location, depth, level of protection, habitat complexity or habitat type, environmental factors, such as CO2 concentration and temperature, time of the day, tourism impact or alien algae invasion (Table S1 in Supplementary Material). Behavioral hypotheses were usually addressed using RUV and DOV systems (38 and 35%, respectively). Apart from the community related and behavioral studies, some studies (17%) were methodological and others (20%) included fish as organisms of secondary interest associated with other animals, such as corals (Table S1 in Supplementary Material). These studies were conducted almost entirely (91%) by ROV, whereas BRUV systems were frequently used in methodological studies (42%).
Most methodological publications compared exclusively the effectiveness of different techniques to study fish (72%), while some focused only on the description of a new sampling method (22%) and one study described a new methodology and also compared it to another method (Table S1 in Supplementary Material). Comparative studies were related to the effectiveness of video techniques against other fish sampling methods such as UVC, eDNA, nets, longline and trawling.
More than half of the studies (71%) that used recording devices focused exclusively on fish without references to other organisms such as invertebrates, which might have been recorded. Few studies examined both fish and benthos concurrently (9%) and some (20%) used video to mainly study benthic macrofauna while mentioning the occurrence of fish when present. RUV and BRUV systems were almost entirely used in fish studies. In contrast, more than half of the ROV surveys included fish in their results only as an incidental occurrence in benthic studies (Table S1 in Supplementary Material). Additionally, 6 ROV studies incorporated also the human impact in the form of marine litter, in addition to fish and benthos (Table S1 in Supplementary Material).
Combination of multiple techniques
More than two thirds of the publications (69%) used exclusively a single video technique, while others (26%) combined a video technique with another method, such as fisheries data. Three studies (3%) used two different video techniques and two studies (2%) combined two video techniques with additional sampling methods (Table S1 in Supplementary Material).
Generally, the use of multiple techniques aimed to combine their results and make inferences for different metrics (59%), compare methodologies (12%) or both (29%). UVC was the most common additional method (35%) and was mostly combined with DOV (in 42% of the DOV studies). The use of fisheries-based methods was also frequent and included long-lines, nets, trawls and traps (Table S1 in Supplementary Material). These techniques were usually combined with BRUV and rarely with ROV. Behavioral studies related to foraging, besides video techniques, included the collection of specimens for gut analysis, the calculation of their Body Condition Index or stable isotope and fatty acid analyses. The consumed biomass was also measured in an herbivory experiment. Further methods included theoretical modelling and eDNA analysis (Table S1 in Supplementary Material).
Technical information: sampling & video analysis
Clupeid species and more specific European pilchard (Sardina pilchardus) and round sardinella (Sardinella aurita), were the most common bait used (38%) in BRUV sampling either individually or as part of a mixture in pellets. Mackerels (Scomber spp.) was also a common bait (29%), while baits mentioned only in one study (8% each) included a mix of fish scraps, cephalopods, and/or cetacean flesh and oil, tuna pieces and phytodetritus in the form of a live diatom culture deposition. Only three studies failed to mention the type of bait used.
The sampling effort in stationary techniques (RUV and BRUV systems) was the soak time (Fig. 5A). Many publications (28%) using RUV or BRUV systems provided only the range of time analyzed, or the minimum or maximum deployment duration. It was common for BRUV systems to sample for 30 min (19%), while in RUV studies sampling lasted for even less than 30 min (27%). Five publications using stationary techniques (12%) did not specify the sampling effort, while five of the RUV studies (23%) mentioned the rate in which they extracted picture frames from their video footage.
a The soak time (min) in Baited Remote Underwater video (BRUV) and Remote Underwater Video (RUV) studies. “Range” includes studies mentioning the minimum, maximum or range of deployment duration, “unspecified” refers to non-provided information and “pictures” represents the cases in which frames were extracted from the video footage. b The transect length (m) in Diver Operated Video (DOV) and Remotely Operated Vehicle (ROV) studies. “Range” includes studies using various transect lengths, and studies in which no information was given are placed in the “unspecified” group
In transect techniques the sampling effort was estimated according to the transect length (Fig. 5B). Most of the ROV studies (56%) included no clear information regarding the transect length sampled. In some cases (22%) transects were of unequal length and only a range of the size was included. A small number of publications sampling with ROV used transects of 100 m and shorter (14%), while in three studies (8%) the ROV sampled for transects greater than 100 m. Usual transect lengths in DOV studies (Fig. 5B) were 25 or 50 m (36%), while more than half of the publications (64%) did not specify the transect length.
Depending on the study goal different data could be derived from the videos processed (Table 1). Data types included community wide metrics, behavior, such as mating and feeding behavior, and functional traits, such as trophic guilds. A common metric was species abundance, that can be estimated in multiple ways depending on the technique type used and author preferences (Table 1). In stationary techniques, besides the traditional metrics such as biodiversity, evenness and species richness, time of first arrival and the time at which the maximum number of individuals per species is seen (tMax) were measured (Table 1).
Discussion
The application of video recording techniques in fish monitoring has been increasing worldwide (Mallet and Pelletier 2014) and this trend is also followed in the Mediterranean Sea, as described in the present work. The use of video recording techniques is expected to increase in the future, particularly for monitoring fish populations and habitats in MPAs and sensitive ecosystems as they are non-destructive and fisheries independent methods (Murphy and Jenkins 2010).
The hydrography of the Mediterranean Sea, a semi-enclosed basin, contributes to the rapid warming up due to climate change, which is more intense in the south than the north (Marbà et al. 2015). At the same time, local environmental knowledge has already indicated alterations in species abundance and distribution in the northern parts of Africa, as thermophilic species are undergoing abundance rises (Azzurro et al. 2011) and other species distributions are shifting northwards (Azzurro et al. 2019). Additional stress is present in the eastern Mediterranean that is impacted by invasions of non-native species more than the western (Galil et al. 2018). Even though rapid changes are occurring in the southern and eastern parts of the Mediterranean, the northwestern regions are the most well studied areas as shown by this work and former studies (Marbà et al. 2015). Considering the above, the need to conduct new surveys in these quickly changing parts of the southern and eastern Mediterranean arises. Future research could focus on gathering data of southern Mediterranean fish to determine the current state of species inhabiting these waters, but it could also focus on comparative studies between the south and north, to identify and highlight the observed changes. Regarding the expansion of alien species, studies could be conducted not only to track their distribution, but also to explore their behavior in the new ecosystems (D’Agostino et al. 2020) and the potential altered behavior of the native species as a response to the invasion (Alós et al 2018; Vivó-Pons et al. 2020).
Many studies in the western Mediterranean were mainly conducted in Italy, Spain and France. While the prosperity of countries in the western part of the basin could explain the skewness in the distribution of publications in the Mediterranean Sea, the large number of ROV studies in Italy could also be linked to local ROV production (Global Electric Italiana). Meanwhile, Spain run a large number of RUV studies, half held at a permanent observatory system (OBSEA), a cabled system established in Barcelona, providing permanent visual material of the surroundings (Aguzzi et al. 2011).
A rather positive finding in the present review was the extended use of underwater video techniques in MPAs. Underwater video methods form an effective alternative to traditional sampling techniques, such as commercial or experimental fishing, and especially in the Mediterranean Sea, a biodiversity hotspot (Coll et al. 2010) with only 6% of the total area under protection (Claudet et al. 2020), such initiatives need to be further encouraged and become well established to secure habitat and fish population integrity.
The use of ROV has been the most common video method in the Mediterranean Sea, particularly for benthic studies, despite the high purchase and operational costs (Sward et al. 2019). Such studies usually focused on describing coral (Fabri et al. 2019) or other benthic habitats (Bo et al. 2012b) combined with ecosystem-associated fish. This method will probably become even more popular in the upcoming years with more widespread use of mini-ROV (e.g., Nalmpanti et al. 2021); due to its low-cost and easy operation (Rubin 2013). RUV and DOV systems were also common techniques, especially in behavioral studies. For example, in experimental settings a fixed camera is beneficial to allow recording the fish reaction to an altered condition (e.g. in Paxton and Smith 2018) and in studies where the fish need to be followed, a diver is a more appropriate option (e.g. in Buñuel et al 2020).
Besides the research objectives that determine the technique choice, experimental depth is also an aspect that needs to be considered when selecting the appropriate technique.
Underwater video can reach a wide range of depths (Sward et al. 2019; Whitmarsh et al. 2017), but most studies in the Mediterranean Sea have been conducted above 500 m depth. Especially DOV techniques were restricted in depths above 62 m due to diver capability limitations, as for depths extending the limits of conventional SCUBA special equipment and specialized divers are required (Soldo and Glavičić 2020). RUV systems were also limited to shallow waters, but BRUV and ROV techniques extended deeper. The choice of BRUV is appropriate in greater depths, where the occurrence of motile fauna is sporadic, since the bait plume can attract individuals from extended areas (Cappo et al. 2003). Indeed, in our review bait was always included when deploying RUVs in depths greater than 45 m. All BRUV systems deployed in depths greater than 350 m were landers (Capezzuto et al. 2012) or submersibles (Jones et al. 2003) fixed on the seafloor. These apparatuses are designed to withstand high pressure (Liang et al. 1998) and operate for long periods of time (Kononets et al. 2021). In contrast, simple apparatuses, which consist of a camera with short battery life, placed inside its housing and then fixed on a steel frame, were deployed in shallower depths (Díaz-Gil et al. 2017; Cattano et al. 2021). Similar to landers and submersibles, ROVs are also capable of reaching deeper regions (Sward et al. 2019). Indeed, their usage in the Mediterranean Sea extended in a wide range of depths, although the nature of many ROV studies, which focused on specific habitats, set them usually in deeper locations. Such environments were cold-water coral ecosystems (Taviani et al. 2017; Moccia et al. 2019), submarine canyons (Fabri et al. 2014; Ayma et al. 2016) and bathyal seamounts (Bo et al. 2020a).
With the exception of one study focusing on pelagic fish, all other publications were restricted to recording fish close to the sea floor or right above it. Fish studies carried out in the water column are scarce probably due to the challenging nature of the sampling technique, as very high sampling effort is required to record few specimens (Torres et al. 2020). The absence of pelagic video samplings does not help with enhancing data collection on large pelagic fish; their assessment is still limited despite worldwide population declines (Letessier et al. 2015).
Other sections of the Mediterranean that were understudied besides pelagic systems were the deep-sea habitats; the reviewed studies were mostly restricted in waters above 500 m. Nonetheless, fish in the deep-sea ecosystems have slowly started to be harvested and thus, their status, ecosystem function and services should be examined (Armstrong et al. 2012) despite the financial constraints of exploring these regions. Additionally, studies in coralligenous habitats, the second most important habitat in the Mediterranean Sea for species diversity after Posidonia oceanica meadows (Boudouresque 2004), were also limited. The increase of video studies in these habitats is not only essential due to their high ecological value, but also because these habitats are vulnerable to destructive sampling techniques, such as fisheries-dependent methods.
Technical considerations
The present review revealed that sampling protocols can differ widely among publications. Bait was not consistent among BRUV studies, although clupeid fishes that are fairly cheap to purchase and contain high amount of oil (Dorman et al. 2012) were a usual choice. Soak duration ranged from a few minutes to hours. RUV systems were used for short deployments (in experimental settings such as in Alós et al. 2015b) or even months in the case of the permanent observatory system OBSEA (Aguzzi et al. 2011), while BRUV systems were usually deployed for around 30 min, a duration that has been proposed in literature as the minimum time required to reach MaxN, the maximum number of individuals recorded in a single video frame (Díaz-Gil et al. 2017). At the same time, maximum deployment duration was defined by battery life capacity (Follana-Berná et al. 2020). In transect studies usual transect lengths ranged between 25 and 50 m for DOV systems, while ROVs were deployed to cover a large variety of transect lengths.
Considering that there is need for comparable data in spatial and temporal scales among different research groups, the multiple sampling settings mentioned before, form an obstacle to the homogeneity of the results. In addition, experiments producing accumulation curves (Stobart et al. 2007) to find the adequate sampling effort are time and money consuming and it is thus, common not to follow this preparatory step before sampling. The above create a need for technical papers focusing on assessing the sampling effort and procedure required to obtain “good enough” and comparable data. Therefore, it would be beneficial for future studies to focus on technical experiments related to the deployment duration (such as Stobart et al. 2007 for BRUV systems), length and width of transects, number of replicates required and type and amount of bait in different habitats, depths and target species. Publications should include reproducible and straightforward sampling protocols with detailed information regarding the methodology, such as details regarding the video system, deployment and sampling design and analysis, as suggested for BRUV studies (Whitmarsh et al. 2017).
In addition, there is need for more methodological studies aiming to standardize the sampling effort per technique, to make possible the complementary use of different techniques in the future. Studies that simultaneously use techniques of different nature, such as fisheries-dependent, sedentary or transect techniques have yielded higher fish diversity observations (Lowry et al. 2011; Schramm et al. 2020). Despite the benefits, when outputs are expressed in different units, for example in a transect and a sedentary sampling, direct comparisons are challenging.
Conclusion
The current review clearly reveals that the use of underwater video in the Mediterranean Sea has been increasing rapidly in fish ecological studies. Their use expands from community analysis to behavioral and methodological approaches. Despite that, the eastern and southern parts of the basin remain understudied. Deep-sea and pelagic habitats, as well as coralligenous ecosystems are also regions of high ecological value with large research gaps. Finally, methodological studies aiming to optimize and standardize sampling effort are generally lacking but are necessary. In the future technological progress is expected to give more opportunities to explore the uses of underwater video and fill research gaps especially in ecologically important regions that need protection.
References
Addis P, Cau A, Massutí E, Merella P, Sinopoli M, Andaloro F (2016) Spatial and temporal changes in the assemblage structure of fishes associated to fish aggregation devices in the Western Mediterranean. Aquat Living Resour 19:149–160. https://doi.org/10.1051/alr:2006018
Aglieri G, Baillie C, Mariani S, Cattano C, Calò A, Turco G, Spatafora D, Di Franco A, Di Lorenzo M, Guidetti P, Milazzo M (2021) Environmental DNA effectively captures functional diversity of coastal fish communities. Mol Ecol 30:3127–3139. https://doi.org/10.1111/mec.15661
Aguilar R, Garcia S, Perry AL, Alvarez H, Blanco J, Bitar G (2019, January) The bathymetric distribution of fish and other key benthic species and communities in Lebanese submarine canyons. 2nd Mediterranean symposium on the conservation of dark habitats. Antalya, Turkey (pp. 5–12).
Aguzzi J, Mànuel A, Condal F, Guillén J, Nogueras M, del Rio J, Costa C, Menesatti P, Puig P, Sardà F, Toma D, Palanques A (2011) The New Seafloor Observatory (OBSEA) for Remote and Long-Term Coastal Ecosystem Monitoring. Sensors 11:5850–5872. https://doi.org/10.3390/s110605850
Aguzzi J, Sbragaglia V, Santamaría G, Del Río J, Sardà F, Nogueras M, Manuel A (2013) Daily activity rhythms in temperate coastal fishes: insights from cabled observatory video monitoring. Mar Ecol Prog Ser 486:223–236. https://doi.org/10.3354/meps10399
Ajemian MJ, Kenworthy MD, Sánchez-Lizaso JL, Cebrian J (2016) Aggregation dynamics and foraging behaviour of striped red mullet Mullus surmuletus in the western Mediterranean Sea. J Fish Biol 88:2051–2059. https://doi.org/10.1111/jfb.12932
Alós J, Puiggrós A, Díaz-Gil C, Palmer M, Rosselló R, Arlinghaus R (2015) Empirical evidence for species-specific export of fish naïveté from a no-take marine protected area in a coastal recreational hook and line fishery. PLoS ONE 10(8):e0135348. https://doi.org/10.1371/journal.pone.0135348
Alós J, Palmer M, Trías P, Díaz-Gil C, Arlinghaus R (2015b) Recreational-angling intensity correlates with alteration of vulnerability to fishing in a carnivorous coastal fish species. Can J Fish Aquat 72:217–225. https://doi.org/10.1139/cjfas-2014-0183
Alós J, Bujosa-Homar E, Terrados J, Tomas F (2018) Spatial distribution shifts in two temperate fish species associated to a newly-introduced tropical seaweed invasion. Biol Invasions 20:3193–3205. https://doi.org/10.1007/s10530-018-1768-2
Alós J, Campos-Candela A, Arlinghaus R (2019) A modelling approach to evaluate the impact of fish spatial behavioural types on fisheries stock assessment. ICES J Mar Sci 76(2):489–500. https://doi.org/10.1093/icesjms/fsy172
Alós j, Palmer M, Rosselló R, Arlinghaus R, (2016) Fast and behavior-selective exploitation of a marine fish targeted by anglers. Sci Rep 6:38093. https://doi.org/10.1038/srep38093
Andaloro F, Ferraro M, Mostarda E, Romeo T, Consoli P (2013) Assessing the suitability of a remotely operated vehicle (ROV) to study the fish community associated with offshore gas platforms in the Ionian Sea: a comparative analysis with underwater visual censuses (UVCs). Helgol Mar Res 67:241–250. https://doi.org/10.1007/s10152-012-0319-y
Angeletti L, Taviani M, Canese S, Foglini F, Mastrototaro F, Argnani A, Trincardi F, Bakran-petricioli T, Ceregato A, Chimienti G, Mačić V, Poliseno A (2014) New deep-water cnidarian sites in the southern Adriatic Sea. Mediterr Mar Sci 15(2):263–273
Angiolillo M, Di Lorenzo B, Farcomeni A, Bo M, Bavestrello G, Santangelo G, Cau A, Mastascusa V, Cau A, Sacco F, Canese S (2015) Distribution and assessment of marine debris in the deep Tyrrhenian Sea (NW Mediterranean Sea, Italy). Mar Pollut Bull 92:149–159
Angiolillo M, La Mesa G, Giusti M, Salvati E, Di Lorenzo B, Rossi L, Canese S, Tunesi L (2021) New records of scleractinian cold-water coral (CWC) assemblages in the southern Tyrrhenian Sea (western Mediterranean Sea): Human impacts and conservation prospects. Prog Oceanogr 197:102656. https://doi.org/10.1016/j.pocean.2021.102656
Armstrong CW, Foley NS, Tinch R, Van den Hove S (2012) Services from the deep: Steps towards valuation of deep sea goods and services. Ecosyst Serv 2:2–13. https://doi.org/10.1016/j.ecoser.2012.07.001
Ayma A, Aguzzi J, Canals M, Lastras G, Bahamon N, Mecho A, Company JB (2016) Comparison between ROV video and Agassiz trawl methods for sampling deep water fauna of submarine canyons in the Northwestern Mediterranean Sea with observations on behavioural reactions of target species. Deep-Sea Res 114:149–159. https://doi.org/10.1016/j.dsr.2016.05.013
Azzurro E, Moschella P, Maynou F (2011) Tracking signals of change in Mediterranean fish diversity based on local ecological knowledge. PLoS ONE 6(9):e24885. https://doi.org/10.1371/journal.pone.0024885
Azzurro E, Sbragaglia V, Cerri J, Bariche M, Bolognini L, Ben Souissi J, Busoni G, Coco S, Antoniadou C, Fanelli E, Ghanem R, Garrabou J, Gianni F, Grati F, Kolitari J, Letterio G, Lipej L, Mazzoldi C, Milone N, Pannacciulli F, Pešić A, Samuel-Rhoads Y, Saponari L, Tomanic J, Topçu NE, Vargiu G, Moschella P (2019) Climate change, biological invasions, and the shifting distribution of Mediterranean fishes: A large-scale survey based on local ecological knowledge. Glob Chang Biol 25(8):2779–2792. https://doi.org/10.1111/gcb.14670
Bailey DM, King NJ, Priede IG (2007) Cameras and carcasses: historical and current methods for using artificial food falls to study deep-water animals. Mar Ecol Prog Ser 350:179–191. https://doi.org/10.3354/meps07187
Barker BAJ, Helmond I, Bax NJ, Williams A, Davenport S, Wadley VA (1999) A vessel-towed camera platform for surveying seafloor habitats of the continental shelf. Cont Shelf Res 19:1161–1170. https://doi.org/10.1016/S0278-4343(99)00017-5
Barnes H (1952) Under-water television and Marine Biology. Nature 169:477–479
Barnett TP, Pierce DW, Schnur R (2001) Detection of Anthropogenic Climate Change in the World’s Oceans. Science 292(5515):270–274. https://doi.org/10.1126/science.1058304
Battaglia P, Canese S, Ammendolia G, Romeo T, Sandulli R, Tunesi L, Andaloro F (2015) New records and underwater observation of the rare fish Scorpaenodes arenai (Osteichthyes: Scorpaenidae) from the central and western Mediterranean Sea. Ital J Zool 82(3):454–458. https://doi.org/10.1080/11250003.2015.1028485
Blower DC, Pandolfi JM, Bruce BD, Gomez-Cabrera MDC, Ovenden JR (2012) Population genetics of Australian white sharks reveals fine-scale spatial structure, transoceanic dispersal events and low effective population sizes. Mar Ecol Prog Ser 455:229–244. https://doi.org/10.3354/meps09659
Bo M, Canese S, Spaggiari C, Pusceddu A, Bertolino M, Angiolillo M, Giusti M, Loreto MF, Salvati E, Greco S, Bacestrello G (2012) Deep Coral Oases in the South Tyrrhenian Sea. PLoS ONE 7(11):e49870. https://doi.org/10.1371/journal.pone.0049870
Bo M, Bertolino M, Bavestrello G, Canese S, Giusti M, Angiolillo M, Pansini M, Taviani M (2012b) Role of deep sponge grounds in the Mediterranean Sea: a case study in southern Italy. Hydrobiologia 687:163–177. https://doi.org/10.1007/s10750-011-0964-1
Bo M, Bavestrello G, Angiolillo M, Calcagnile L, Canese S, Cannas R, Cau A, D’Elia M, D’Oriano F, Follesa MC, Quarta G, Cau A (2015) Persistence of Pristine Deep-Sea Coral Gardens in the Mediterranean Sea (SW Sardinia). PLoS ONE 10(3):e0119393. https://doi.org/10.1371/journal.pone.0119393
Bo M, Coppari M, Betti F, Enrichetti F, Bertolino M, Massa F, Bava S, Gay G, Cattaneo-Vietti R, Bavestrello G (2020a) The high biodiversity and vulnerability of two Mediterranean bathyal seamounts support the need for creating offshore protected areas. Aquat Conserv 31:543–566. https://doi.org/10.1002/aqc.3456
Bo M, Coppari M, Betti F, Massa F, Gay G, Cattaneo-Vietti R, Bavastrello G (2020) Unveiling the deep biodiversity of the Janua Seamount (Ligurian Sea): first Mediterranean sighting of the rare Atlantic bamboo coral Chelidonisis aurantiaca Studer, 1890. Deep-Sea Res 156:103186. https://doi.org/10.1016/j.dsr.2019.103186
Boada J, Arthur R, Farina S, Santana Y, Mascaró O, Romero J, Alcoverro T (2015a) Hotspots of predation persist outside marine reserves in the historically fished Mediterranean Sea. Biol Conserv 191:67–74. https://doi.org/10.1016/j.biocon.2015.06.017
Boada J, Sanmartí N, Selden RL, Lucas A, Pérez M, Alcoverro T, Romero J (2015b) Evaluating potential artifacts of tethering techniques to estimate predation on sea urchins. J Exp Mar Biol 471:17–22. https://doi.org/10.1016/j.jembe.2015.05.011
Boada J, Pagès JF, Gera A, Macpherson E, Santana Y, Romero J, Alcoverro T (2018) The richness of small pockets: Decapod species peak in small seagrass patches where fish predators are absent. Mar Environ Res 142:1–6. https://doi.org/10.1016/j.marenvres.2018.09.012
Boudouresque CF (2004) Marine biodiversity in the Mediterranean: status of species, populations and communities. Scientific Reports of Port-Cros National Park 20:97–146
Bozzano A, Sarda F (2002) Fishery discard consumption rate and scavenging activity in the northwestern Mediterranean Sea. ICES J Mar Sci 59:15–28. https://doi.org/10.1006/jmsc.2001.1142
Bracciali C, Campobello D, Giacoma C, Sarà G (2012) Effects of nautical traffic and noise on foraging patterns of Mediterranean Damselfish (Chromis chromis). PLoS ONE 7(7):e40582. https://doi.org/10.1371/journal.pone.0040582
Buñuel X, Alcoverro T, Pagès JF, Romero J, Ruiz JM, Arthur R (2020) The dominant seagrass herbivore Sarpa salpa shifts its shoaling and feeding strategies as they grow. Sci Rep 10:10622. https://doi.org/10.1038/s41598-020-67498-1
Buñuel X, Alcoverro T, Romero J, Arthur R, Ruiz JM, Pérez M, Ontoria Y, Raventós N, Macpherson E, Torrado H, Pagès JF (2021) Warming intensifies the interaction between the temperate seagrass Posidonia oceanica and its dominant fish herbivore Sarpa salpa. Mar Environ Res 165:105237. https://doi.org/10.1016/j.marenvres.2020.105237
Capezzuto F, Maiorano P, Panza M, Indennidate A, Sion L, D’Onghia G (2012) Occurrence and behaviour of Paromola cuvieri (Crustacea, Decapoda) in the Santa Maria di Leuca cold-water coral community (Mediterranean Sea). Deep-Sea Res 59:1–7. https://doi.org/10.1016/j.dsr.2011.10.006
Capezzuto F, Calculli C, Carlucci R, Carluccio A, Maiorano P, Pollice A, Sion L, Tursi A, D’Onghia G (2019) Revealing the coral habitat effect on benthopelagic fauna diversity in the Santa Maria di Leuca cold-water coral province using different devices and Bayesian hierarchical modelling. Aquat Conserv 29:1608–1622. https://doi.org/10.1002/aqc.3144
Cappo M, Death G, Speare P (2007) Inter-reef vertebrate communities of the Great Barrier Reef Marine Park determined by baited remote underwater video stations. Mar Ecol Prog Ser 350:209–221. https://doi.org/10.3354/meps07189
Cappo M, Harvey E, Malcolm H, Speare P (2003) Potential of video techniques to monitor diversity, abundance and size of fish in studies of marine protected areas. In: Beumer JP, Grant A,Smith DC (eds), Aquatic protected areas: what works bestand how do we know? Proceedings of the World Congress on Aquatic Protected Areas, August 2002, Cairns. Cairns, Austalia: Australian Society of Fish Biology, pp 455–464.
Carluccio A, Caapezzuto F, Maiorano P, Sion L, D’Onghia G (2021) Deep-water cartilaginous fishes in the central mediterranean sea: comparison between geographic areas with two low impact tools for sampling. J Mar Sci 9:686. https://doi.org/10.3390/jmse9070686
Cattano C, Turco G, Di Lorenzo M, Gristina M, Visconti G, Milazzo M (2021) Sandbar shark aggregation in the central Mediterranean Sea and potential effects of tourism. Aquat Conserv 31:1420–1428. https://doi.org/10.1002/aqc.3517
Cau A, Follesa MC, Moccia D, Bellodi A, Mulas A, Bo M, Canese S, Angiolillo M, Cannas R (2017) Leiopathes glaberrima millennial forest from SW Sardinia as nursery ground for the small spotted catshark Scyliorhinus canicula. Aquat Conserv 27:731–735. https://doi.org/10.1002/aqc.2717
Chimienti G, Angeletti L, Mastrototaro F (2018) Withdrawal behaviour of the red sea pen Pennatula rubra (Cnidaria: Pennatulacea). Eur Zool j 85(1):64–70. https://doi.org/10.1080/24750263.2018.1438530
Chimienti G, De Padova D, Mossa M, Mastrototaro F (2020) A mesophotic black coral forest in the Adriatic Sea. Sci Rep 10:8504. https://doi.org/10.1038/s41598-020-65266-9
Ciriaco S, Costantini M, Italiano C, Odorico R, Picciulin M, Verginella L, Spoto M (1998) Monitoring the miramare marine reserve: assessment of protection efficiency. Ital J Zool 65:383–386
Claudet J, Fraschetti S (2010) Human-driven impacts on marine habitats: a regional meta-analysis in the Mediterranean Sea. Biol Conserv 143:2195–2206. https://doi.org/10.1016/j.biocon.2010.06.004
Claudet J, Loiseau C, Sostres M, Zupan M (2020) Underprotected marine protected areas in a global biodiversity hotspot. One Earth 2:380–384. https://doi.org/10.1016/j.oneear.2020.03.008
Coll M, Piroddi C, Steenbeek J, Kaschner K, Ben Rais Lasram F, Aguzzi J, Ballesteros E, Bianchi CN, Corbera J, Dailianis T, Danovaro R, Estrada M, Froglia C, Galil BS, Gasol JM, Getwagen R, Gil J, Guilhaumon F, Kesner-Reyes K, Kitsos MS, Koukouras A, Lampadariou N, Laxamana E, López-Fé CM, Lotze HK, Martin D, Mouillot D, Oro D, Raicevich S, Ruis-Barile J, Saiz-Salinas JI, San Vicente C, Somot S, Templado J, Turon X, Vafidis D, Villanueva R, Voultsiadou E (2010) The Biodiversity of the Mediterranean Sea: Estimates, Patterns, and Threats. PLoS ONE 5(8):e11842. https://doi.org/10.1371/journal.pone.0011842
Collie JS, Hall SJ, Kaiser MJ, Poiner IR (2000) A quantitative analysis of fishing impacts on shelf-sea benthos. J Anim Ecol 69:785–798. https://doi.org/10.1046/j.1365-2656.2000.00434.x
Collins DL, Langlois TJ, Bond T, Holmes TH, Harvey ES, Fisher R, McLean DL (2017) A novel stereo-video method to investigate fish-habitat relationships. Methods Ecol Evol 8:116–125. https://doi.org/10.1111/2041-210X.12650
Condal F, Aguzzi J, Sardà F, Nogueras M, Cadena J, Costa C, Del Rio J, Manuel A (2012) Seasonal rhythm in a Mediterranean coastal fish community as monitored by a cabled observatory. J Mar Biol 159:2809–2817. https://doi.org/10.1007/s00227-012-2041-3
Consalvo I, La Mesa G, Canese S, Giusti M, Salvati E, Loia M, Tunesi L (2021) The demersal fish assemblages of the infra and circalittoral coastal rocky bottoms of the Aeolian Archipelago (Central Mediterranean Sea) studied by Remotely Operated Vehicle (ROV). Mediterr Mar Sci 22(1):27–39
Consoli P, Martino A, Romeo T, Sinopoli M, Perzia P, Canese S, Vivona P, Andaloro F (2015) The effect of shipwrecks on associated fish assemblages in the central Mediterranean Sea. J Mar Biolog Assoc 95(1):17–24. https://doi.org/10.1017/S0025315414000940
Consoli P, Esposito V, Battaglia P, Altobelli C, Perzia P, Romeo T, Canese S, Andaloro F (2016) Fish distribution and habitat complexity on banks of the strait of sicily (Central Mediterranean Sea) from remotely-operated vehicle (ROV) Explorations. PLoS ONE 11(12):e0167809. https://doi.org/10.1371/journal.pone.0167809
Consoli P, Altobelli C, Perzia P, Bo M, Rosso A, Alongi G, Serio D, Canese S, Romeo T, Andaloro F (2021) Species and habitats of conservation interest in the ecologically and biologically significant area of the Strait of Sicily: a contribution towards the creation of a specially protected areas of Mediterranean importance. Mediterr Mar Sci 22(2):297–316. https://doi.org/10.12681/mms.25125
Corbera G, Lo Iacono C, Gràcia E, Grinyó J, Pierdomenico M, Huvenne VAI, Aguilar R, Gili JM (2019) Ecological characterisation of a Mediterranean cold-water coral reef: Cabliers Coral Mound Province (Alboran Sea, western Mediterranean). Progr Oceanogr 175:245–262. https://doi.org/10.1016/j.pocean.2019.04.010
Costa C, Scardi M, Vitalini V, Cataudella S (2009) A dual camera system for counting and sizing Northern Bluefin Tuna (Thunnus thynnus; Linnaeus, 1758) stock, during transfer to aquaculture cages, with a semi automatic artificial neural network tool. Aquaculture 291:161–167. https://doi.org/10.1016/j.aquaculture.2009.02.013
Costello C, Ovando D, Hilborn R, Gaines SD, Deschenes O, Lester SE (2012) Status and solutions for the world’s unassessed fisheries. Science 338(6106):517–520. https://doi.org/10.1126/science.1223389
D’Agostino D, Jimenez C, Reader T, Heyworth S, Hadjioannou L, Josephides M, Feary DA (2020) Behavioural traits and feeding ecology of Mediterranean lionfish and naiveté of native species to lionfish predation. Mar Ecol Prog Ser 638:123–135. https://doi.org/10.3354/meps13256
D’Anna G, Giacalone VM, Badalamenti F, Pipitone C (2004) Releasing of hatchery-reared juveniles of the white seabream Diplodus sargus (L., 1758) in the Gulf of Castellammare artificial reef area (NW Sicily). Aquaculture 233:251–268. https://doi.org/10.1016/j.aquaculture.2003.10.024
D’Onghia G, Indennidate A, Giove A, Savini A, Capezzuto F, Sion L, Vertino A, Maiorano P (2011) Distribution and behaviour of deep-sea benthopelagic fauna observed using towed cameras in the Santa Maria di Leuca cold-water coral province. Mar Ecol Prog Ser 443:95–110. https://doi.org/10.3354/meps09432
D’Onghia G, Capezzuto F, Carluccio A, Carlucci R, Giove A, Mastrototaro F, Panza M, Sion L, Tursi A, Maiorano P (2015a) Exploring composition and behaviour of fish fauna by in situ observations in the Bari Canyon (Southern Adriatic Sea, Central Mediterranean). Mar Ecol 36:541–556. https://doi.org/10.1111/maec.12162
D’Onghia G, Capezzuto F, Cardone F, Carlucci R, Carluccio A, Chimienti G, Corriero G, Longo C, Maiorano P, Mastrototaro F, Panetta P, Rosso A, Sanfilippo R, Sion L, Tursi A (2015) Macro- and megafauna recorded in the submarine Bari Canyon (southern Adriatic, Mediterranean Sea) using different tools. Mediterr Mar Sci 16(1):180–196. https://doi.org/10.12681/mms.1082
Deidun A, Castriota L, Arrigo S (2011) A tale of two Atlantic fish migrants: records of the lesser amberjack Seriola fasciata and the African hind Cephalopholis taeniops from the Maltese Islands. J Black Sea/medit 17(3):223–233
Díaz-Gil C, Smee SL, Cotgrove L, Follana-Berná G, Hinz H, Marti-Puig P, Grau A, Palmer M, Catalán IA (2017) Using stereoscopic video cameras to evaluate seagrass meadows nursery function in the Mediterranean. Mar Biol 164:137. https://doi.org/10.1007/s00227-017-3169-y
Dimarchopoulou D, Stergiou KI, Tsikliras AC (2017) Gap analysis on the biology of Mediterranean marine fishes. PLoS ONE 12(4):e0175949. https://doi.org/10.1371/journal.pone.0175949
Dorman SR, Harvey ES, Newman SJ (2012) Bait effects in sampling coral reef fish assemblages with stereo-BRUVs. PLoS ONE 7(7):e41538. https://doi.org/10.1371/journal.pone.0041538
Dremière PY, Fiorentini L, Cosimi G, Leonori I, Sala A, Spagnolo A (1999) Escapement from the main body of the bottom trawl used for the Mediterranean international trawl survey (MEDITS). Aquat Living Resour 12(3):207–217. https://doi.org/10.1016/S0990-7440(00)88471-5
Enrichetti F, Dominguez- Carrió C, Toma M, Bavestrello G, Betti F, Canese S, Bo M (2019) Megabenthic communities of the Ligurian deep continental shelf and shelf break (NW Mediterranean Sea). PLoS ONE 14(10):e0223949. https://doi.org/10.1371/journal.pone.0223949
Enrichetti F, Bava S, Bavestrello G, Betti F, Lanteri L, Bo M (2019b) Artisanal fishing impact on deep coralligenous animal forests: A Mediterranean case study of marine vulnerability. Ocean Coastal Manag 177:112–126. https://doi.org/10.1016/j.ocecoaman.2019.04.021
Fabri MC, Pedel L, Beuck L, Galgani F, Hebbeln D, Freiwald A (2014) Megafauna of vulnerable marine ecosystems in French Mediterranean submarine canyons: Spatial distribution and anthropogenic impacts. Deep-Sea Res 104:184–207. https://doi.org/10.1016/j.dsr2.2013.06.016
Fabri MC, Vinha B, Allais AG, Bouhier ME, Dugornay O, Gaillot A, Arnaubec A (2019) Evaluating the ecological status of cold-water coral habitats using noninvasive methods: An example from Cassidaigne canyon, northwestern Mediterranean Sea. Prog Oceanogr 178:102172. https://doi.org/10.1016/j.pocean.2019.102172
Fanelli E, Sbragaglia V, Azzurro E, Marini S, Del Rio J, Toma D, Aguzzi J (2017) Seasonal changes in coastal fish assemblages by multiparametric video-observatory monitoring. IMEKO International Conference on Metrology for The Sea Naples, Italy, pp 13–16.
Fedra K, Machan R (1979) A self-contained underwater time-lapse camera for in situ long-term observations. Mar Biol 55:239–246
Follana-Berná G, Palmer M, Lekanda-Guarrotxena A, Grau A, Arechavala-Lopez P (2020) Fish density estimation using unbaited cameras: Accounting for environmental-dependent detectability. J Exp Mar Biol 527:151376. https://doi.org/10.1016/j.jembe.2020.151376
Galasso NM, Bonaviri C, Di Trapani F, Picciotto M, Gianguzza P, Agnetta D, Badalamenti F (2015) Fish-seastar facilitation leads to algal forest restoration on protected rocky reefs. Sci Rep 5:12409. https://doi.org/10.1038/srep12409
Galil BS, Marchini A, Occhipinti-Ambrogi A (2018) East is east and West is west? Management of marine bioinvasions in the Mediterranean Sea. Estuar Coast Shelf Sci 201:7–16. https://doi.org/10.1016/j.ecss.2015.12.021
Gates AR, Jones DOB, Cartes JE (2012) In situ video observations of benthic megafauna and fishes from the deep eastern Mediterranean Sea off Egypt. Afr J Mar Sci 34(2):215–222. https://doi.org/10.2989/1814232X.2012.675121
Giakoumi S, Pey A, Thiriet P, Francour P, Guidetti P (2019) Patterns of predation on native and invasive alien fish in Mediterranean protected and unprotected areas. Mar Environ Res 150:104792. https://doi.org/10.1016/j.marenvres.2019.104792
Glavičić I, Kovačić M, Paliska D, Laslo D (2020) The influence of depth on the Mediterranean reef fish assemblage revealed by the new video on underwater scooter method. Acta Adriat 61(2):145–162. https://doi.org/10.32582/aa.61.2.3
Goverts Z, Nührenberg P, Jordan A (2021) Environmental reconstruction and tracking as methods to explore social interactions in marine environments: A test case with the Mediterranean rainbow wrasse Coris julis. Front Mar Sci 8:695100. https://doi.org/10.3389/fmars.2021.695100
Grane-Feliu X, Bennett S, Hereu B, Aspillaga E, Santan-Garcon J (2019) Comparison of diver operated stereo-video and visual census to assess targeted fish species in Mediterranean marine protected areas. J Exp Mar Biol 520:151205. https://doi.org/10.1016/j.jembe.2019.151205
Grinyó J, Chevaldonné P, Schohn T, Le Bris N (2021) Comparison of diver operated stereo-video and visual census to assess targeted fish species in Mediterranean marine protected areas. Deep-Sea Res 171:103475. https://doi.org/10.1016/j.dsr.2021.103475
Halpern BS, Walbridge S, Selkoe KA, Kappel CV, Micheli F, D’Agrosa C, Bruno JF, Casey KS, Ebert C, Fox HE, Fujita R, Heinemann D, Lenihan HS, Madin EMP, Perry MT, Selig ER, Spalding M, Steneck R, Watson R (2008) A global map of human impact on marine ecosystems. Science 319(5865):948–952. https://doi.org/10.1126/science.1149345
Hammerschlag N, Gallagher AJ, Lazarre DM (2011) A review of shark satellite tagging studies. J Exp Marine Biol Ecol 398(1–2):1–8
Harvey ES, Cappo M, Butler JJ, Hall N, Kendrick GA (2007) Bait attraction affects the performance of remote underwater video stations in assessment of demersal fish community structure. Mar Ecol Prog Ser 350:245–257. https://doi.org/10.3354/meps07192
Harvey ES, McLean DL, Goetze JL, Saunders BJ, Langlois TJ, Monk J, Barrett N, Wilson SK, Holmes TH, Ierodiaconou D, Jordan AR, Meekan MG, Malcolm HA, Heupel MR, Harasti D, Huveneers C, Knott NA, Fairclough DV, Currey-Randall LM, Travers MJ, Radford BT, Rees MJ, Speed CW, Wakefield CB, Cappo M, Newman SJ (2021) The BRUVs workshop – An Australia-wide synthesis of baited remote underwater video data to answer broad-scale ecological questions about fish, sharks and rays. Mar Policy 127:104430. https://doi.org/10.1016/j.marpol.2021.104430
Heagney EC, Lynch TP, Babcock RC, Suthers IM (2007) Pelagic fish assemblages assessed using mid-water baited video: standardising fish counts using bait plume size. Mar Ecol Prog Ser 350:255–266. https://doi.org/10.3354/meps07193
Jeffreys RM, Lavaleye MSS, Bergman MJN, Duineveld GCA, Witbaard R (2011) Do abyssal scavengers use phytodetritus as a food resource? Video and biochemical evidence from the Atlantic and Mediterranean. Deep-Sea Res 58:415–428. https://doi.org/10.1016/j.dsr.2011.02.002
Jones EG, Tselepides A, Bagley PM, Collins MA, Priede IG (2003) Bathymetric distribution of some benthic and benthopelagic species attracted to baited cameras and traps in the deep eastern Mediterranean. Mar Ecol Prog Ser 251:75–86. https://doi.org/10.3354/meps251075
Katsanevakis S, Coll M, Piroddi C, Steenbeek J, Ben Rais Lasram F, Zenetos A, Cardoso AC (2014) Invading the Mediterranean Sea: biodiversity patterns shaped by human activities. Front Mar Sci 1:1–32. https://doi.org/10.3389/fmars.2014.00032
Kononets M, Tengberg A, Nilsson M, Ekeroth N, Hylen A, Robertson EK, Van de Velde S, Bonaglia S, Rütting T, Blomqvist S, Hall POJ (2021) In situ incubations with the Gothenburg benthic chamber landers: Applications and quality control. J Mar Syst 214:103475. https://doi.org/10.1016/j.jmarsys.2020.103475
Kruschel C, Ivancic T, Lakic R, Schultz ST (2016) The effects of interference interactions on fish communities as assessed by Baited Remote Underwater Videography (BRUV). Rapp Comm Int Mer Médit 41:449
La Manna G, Manghi M, Perretti F, Sarà G (2016) Behavioral response of brown meagre (Sciaena umbra) to boat noise. Mar Pollut Bull 110:324–334. https://doi.org/10.1016/j.marpolbul.2016.06.045
La Manna G, Guala I, Grech D, Perretti F, Ronchetti F, Manghi M, Ruiu A, Ceccherelli G (2021) Performance of a baited underwater video system vs. the underwater visual census technique in assessing the structure of fish assemblages in a Mediterranean marine protected area. Mediterr Mar Sci 22(3):480–495
Langlois TJ, Harvey ES, Fitzpatrick B, Meeuwig JJ, Shedrawi G, Watson DL (2010) Cost-efficient sampling of fish assemblages: comparison of baited video stations and diver video transects. Aquat Biol 9:155–168. https://doi.org/10.3354/ab00235
Letessier TB, Bouchet PJ, Meeuwig JJ (2015) Sampling mobile oceanic fishes and sharks: implications for fisheries and conservation planning. Biol Rev 92(2):627–646. https://doi.org/10.1111/brv.12246
Liang CC, Teng TL, Lai WH (1998) A study of diving depth on deep-diving submersible vehicle. Int J Press Vessels Pip 75:447–457. https://doi.org/10.1016/S0308-0161(98)00041-6
Linley TD, Lavaleye M, Maiorano P, Bergman M, Capezzuto F, Cousins NJ, D’Onghia G, Duineveld G, Shields MA, Sion L, Tursi A, Priede IG (2017) Effects of cold-water corals on fish diversity and density (European continental margin: Arctic, NE Atlantic and Mediterranean Sea): Data from three baited lander systems. Deep-Sea Res II 145:8–21. https://doi.org/10.1016/j.dsr2.2015.12.003
Linley TD, Craig J, Jamieson AJ, Priede IG (2018) Bathyal and abyssal demersal bait-attending fauna of the Eastern Mediterranean Sea. Mar Biol 165:159. https://doi.org/10.1007/s00227-018-3413-0
Lowry M, Folpp H, Gregson M, Mckenzie R (2011) A comparison of methods for estimating fish assemblages associated with estuarine artificial reefs. Braz J Oceanogr 59:119–131. https://doi.org/10.1590/S1679-87592011000300014
Macali A, Semenov A, De Mendoza FP, Dinoi A, Bergami E, Tiralongo F (2020) Relative influence of environmental factors on biodiversity and behavioural traits of a rare mesopelagic fish, Trachipterus trachypterus (Gmelin, 1789), in a continental shelf front of the Mediterranean Sea. J Mar Sci Eng 8:581. https://doi.org/10.3390/jmse8080581
Mallet D, Pelletier D (2014) Underwater video techniques for observing coastal marine biodiversity: a review of sixty years of publications (1952–2012). Fish Res 154:44–62. https://doi.org/10.1016/j.fishres.2014.01.019
Marbà N, Jordà G, Agustí S, Girard C, Duarte CM (2015) Footprints of climate change on Mediterranean Sea biota. Front Mar Sci 2:56. https://doi.org/10.3389/fmars.2015.00056
Mariani A, Dell’Aquila M, Scardi M, Costa C (2014) Feasibility study to assess the utilization of stereo-video systems during transfer of atlantic bluefin tunas (Thunnus thynnus) to evaluate their number and size. Collect Vol Sci P ICCAT 70(2):401–421
Marletta G, Lombardo A (2020) Assessment of grazing impact on deep canopy-forming species in the western Ionian Sea. Central Mediterranean. Int J Aquat Biol 8(5):365–376. https://doi.org/10.22034/ijab.v8i5.882
Marra S, Coppa S, Camedda A, Mazzoldi C, Wrachien F, Massaro G, De Lucia GA (2016) Recovery trends of commercial fish: The case of an underperforming Mediterranean marine protected area. PLoS ONE 11(1):e0146391. https://doi.org/10.1371/journal.pone.0146391
Mascolino S, Mariani S, Benvenuto C (2019) Behavioural responses in a congested sea: an observational study on a coastal nest-guarding fish. Eur Zool j 86(1):504–518. https://doi.org/10.1080/24750263.2019.1699611
Mastrototaro F, D’Onghia G, Corriero G, Matarrese A, Maiorano P, Panetta P, Gherardi M, Longo C, Rosso A, Sciuto F, Sanfilippo R, Gravili C, Boero F, Taviani M, Tursi A (2010) Biodiversity of the white coral bank off Cape Santa Maria di Leuca (Mediterranean Sea): An update. Deep-Sea Res II 57:412–430. https://doi.org/10.1016/j.dsr2.2009.08.021
Mastrototaro F, Chimienti G, Acosta J, Blanco J, Garcia S, Rivera J, Aguilar R (2017) Isidella elongata (Cnidaria: Alcyonacea) facies in the western Mediterranean Sea: visual surveys and descriptions of its ecological role. Eur Zool j 84(1):209–225. https://doi.org/10.1080/24750263.2017.1315745
Mecho A, Aguzzi J, De Mol B, Lastras G, Ramirez-Llodra E, Bahamon N, Company JB, Canals M (2018) Visual faunistic exploration of geomorphological human-impacted deep-sea areas of the north-western Mediterranean Sea. J Mar Biolog Assoc 98(6):1241–1252. https://doi.org/10.1017/S0025315417000431
Micheli F, Halpern BS, Walbridge S, Ciriaco S, Ferretti F, Fraschetti S, Lewison R, Nykjaer L, Rosenberg AA (2013) Cumulative human impacts on mediterranean and black sea marine ecosystems: assessing current pressures and opportunities. PLoS ONE 8(12):e79889. https://doi.org/10.1371/journal.pone.0079889
Milazzo M, Cattano C, Alonzo SH, Foggo A, Gristina M, Rodolfo-Metalpa R, Sinopoli M, Spatafora D, Stiver KA, Hall-Spencer JM (2016) Ocean acidification affects fish spawning but not paternity at CO2 seeps. Proc Royal Soc B 283:20161021. https://doi.org/10.1098/rspb.2016.1021
Moccia D, Cau A, Alvito A, Canese S, Cannas R, Bo M, Angiolillo M, Folesa MC (2019) New sites expanding the “Sardinian cold-water coral province” extension: a new potential cold-water coral network? Aquat Conserv 29:153–160. https://doi.org/10.1002/aqc.2975
Moccia D, Cau A, Bramanti L, Carugati L, Canese S, Follesa MC, Cannas R (2021) Spatial distribution and habitat characterization of marine animal forest assemblages along nine submarine canyons of Eastern Sardinia (central Mediterranean Sea). Deep-Sea Res 167:103422. https://doi.org/10.1016/j.dsr.2020.103422
Monti F, Duriez O, Dominici JM, Sforzi A, Robert A, Fusani L, Grémillet D (2018) The price of success: integrative long-term study reveals ecotourism impacts on a flagship species at a UNESCO site. Anim Conserv 21(6):448–458. https://doi.org/10.1111/acv.12407
Murphy HM, Jenkins GP (2010) Observational methods used in marine spatial monitoring of fishes and associated habitats: a review. Mar Freshw Res 61:236–252. https://doi.org/10.1071/MF09068
Nagelkerken I, Russell BD, Gillanders BM, Connell SD (2015) Ocean acidification alters fish populations indirectly through habitat modification. Nat Clim Chang 6(1):89–93. https://doi.org/10.1038/NCLIMATE2757
Nalmpanti M, Pardalou A, Tsikliras AC, Dimarchopoulou D (2021) Assessing fish communities in a multiple-use marine protected area using an underwater drone (Aegean Sea, Greece). J Mar Biolog Assoc 101:1061–1071. https://doi.org/10.1017/S0025315421000904
Neiße N, Santon M, Bitton PP, Michiels NK (2020) Small benthic fish strike at prey over distances that fall within theoretical predictions for active sensing using light. J Fish Biol 97:1201–1208. https://doi.org/10.1111/jfb.14502
Palomares MLD, Froese R, Derrick B, Meeuwing JJ, Nöel SL, Tsui G, Woroniak J, Zeller D, Pauly D (2020) Fishery biomass trends of exploited fish populations in marine ecoregions, climatic zones and ocean basins. Estuar Coast Shelf Sci 243:106896. https://doi.org/10.1016/j.ecss.2020.106896
Papadopoulou KN, Smith J, Apostolidis C, Karachle PK (2015) Fish pot trials in the Aegean Sea: first experimental results. 11th Panhellenic Symposium on Oceanography and Fisheries, Mytilene, Lesvos island, Greece.
Paxton AB, Smith D (2018) Visual cues from an underwater illusion increase relative abundance of highly reef-associated fish on an artificial reef. Mar Freshw Res 69:614–619. https://doi.org/10.1071/MF17179
Pelletier D, Roos D, Bouchoucha M, Schohn T, Roman W, Gonson C, Bockel T, Carpentier L, Preuss B, Powell A, Garcia J, Gaboriau M, Cadé F, Royaux C, Le Bras Y, Reecht Y (2021) A standardized workflow based on the STAVIRO Unbaited Underwater Video System for monitoring fish and habitat essential biodiversity variables in coastal areas. Front Mar Sci 8:689280. https://doi.org/10.3389/fmars.2021.689280
Picciulin M, Sebastianutto L, Codarin A, Farina A, Ferrero EA (2010) In situ behavioural responses to boat noise exposure of Gobius cruentatus (Gmelin, 1789; fam. Gobiidae) and Chromis chromis (Linnaeus, 1758; fam. Pomacentridae) living in a Marine Protected Area. J Exp Mar Biol 386:125–132. https://doi.org/10.1016/j.jembe.2010.02.012
Pierdomenico M, Martorelli E, Dominguez-Carrió C, Gili JM, Chiocci FL (2016) Seafloor characterization and benthic megafaunal distribution of an active submarine canyon and surrounding sectors: the case of Gioia Canyon (southern Tyrrhenian Sea). J Mar Syst 157:101–117. https://doi.org/10.1016/j.jmarsys.2016.01.005
Piroddi C, Colloca F, Tsikliras AC (2020) The living marine resources in the Mediterranean Sea large marine ecosystem. Environ Dev 36:100555. https://doi.org/10.1016/j.envdev.2020.100555
Prato G, Guidetti P, Bartolini F, Mangialajo L, Francour P (2013) The importance of high-level predators in marine protected area management: consequences of their decline and their potential recovery in the Mediterranean context. Adv Oceanogr Limnol 4:176–193. https://doi.org/10.4081/aiol.2013.5343
Priester CR, Martinez-Ramirez L, Erzini K, Abecasis D (2021) The impact of trammel nets as an MPA soft bottom monitoring method. Ecol Indic 120:106877. https://doi.org/10.1016/j.ecolind.2020.106877
Raventos N, Ferrari B, Planes S (2009) Differences in population parameters and behaviour of the herbivorous fish Sarpa salpa between protected and unprotected seagrass meadows in the north-western Mediterranean. J Mar Biol Assoc 89(6):1153–1159. https://doi.org/10.1017/S0025315409000423
Rizzo L, Musco L, Crovetta F (2021) Cohabiting with litter: Fish and benthic assemblages in coastal habitats of a heavily urbanized area. Mar Pollut Bull 164:112077. https://doi.org/10.1016/j.marpolbul.2021.112077
Rooper CN (2008) Underwater Video Sleds: Versatile and Cost Effective Tools for Habitat Mapping. In: Reynolds JR, Greene HG (eds) Marine Habitat Mapping Technology for Alaska. University of Alaska Fairbanks, Fairbanks, pp 100–107
Rountree RA, Gilmore RG, Goudey CA, Hawkins AD, Luczkovich JJ, Mann DA (2006) Listening to fish: applications of passive acoustics to fisheries science. Fisheries 31(9):433–446. https://doi.org/10.1577/1548-8446(2006)31[433:LTF]2.0.CO;2
Rubin S (2013) Mini-ROVs, going where no ROV has gone before. Proceedings of the 2013 OCEANS conference, San Diego, CA, USA, pp. 1–4.
Samoilys MA, Carlos G (2000) Determining methods of underwater visual census for estimating the abundance of coral reef fishes. Environ Biol Fishes 57:289–304. https://doi.org/10.1023/A:1007679109359
Santos AMP (2000) Fisheries oceanography using satellite and airborne remote sensing methods: a review. Fish Res 49(1):1–20. https://doi.org/10.1016/S0165-7836(00)00201-0
Sarà G, Oliveri A, Martino G, Serra S, Meloni G, Pais A (2007) Response of captive seabass and seabream as behavioural indicator in aquaculture. Ital J Anim Sci 6:823–825
Sarà G, Oliveri A, Martino G, Campobello D (2010) Changes in behavioural response of Mediterranean seabass (Dicentrarchus labrax L) under different feeding distributions. Ital J Anim Sci 9:e23. https://doi.org/10.4081/ijas.2010.e23
Sbragaglia V, Nuñez JD, Dominoni D, Coco S, Fanelli E, Azzurro E, Marini S, Nogueras M, Ponti M, Del Rio J, Aguzzi J (2019) Annual rhythms of temporal niche partitioning in the Sparidae family are correlated to different environmental variables. Sci Rep 9:1708. https://doi.org/10.1038/s41598-018-37954-0
Sbragaglia V, Coco S, Correia RA, Coll M, Arlinghaus R (2021) Analyzing publicly available videos about recreational fishing reveals key ecological and social insights: a case study about groupers in the Mediterranean Sea. Sci Total Environ 765:142672. https://doi.org/10.1016/j.scitotenv.2020.142672
Sbragaglia V, Aguzzi J, Santamaria G, Manuel A, Del Rio J, Nogueras M, Sardà F (2013, March) The use of coastal cabled video-observatories to monitor seasonal changes in shallow water fish community. International Symposium on Underwater Technology (UT13) Volume: Proceeding of the UT-13 IEEE-Tokyo: 1–3
Schramm KD, Harvey ES, Goetze JS, Travers MJ, Warnock B, Saunders BJ (2020) A comparison of stereo-BRUV, diver operated and remote stereo-video transects for assessing reef fish assemblages. J Exp Mar Biol 524:151273. https://doi.org/10.1016/j.jembe.2019.151273
Sciberras M, Hiddink JG, Jennings S, Szostek CL, Hughes KM, Kneafsey B, Clarke LJ, Ellis N, Rijnsdorp AD, McConnaughey RA, Hilborn R, Collie JS, Pitcher CR, Amoroso RO, Parma AM, Suuronen P, Kaiser MJ (2018) Response of benthic fauna to experimental bottom fishing: a global meta-analysis. Fish Fish 19(4):698–715. https://doi.org/10.1111/faf.12283
Shabtay A, Portman M, Carmel Y (2018) Contributions of marine infrastructures to marine planning and protected area networking. Aquat Conserv 28:830–839. https://doi.org/10.1002/aqc.2916
Sinopoli M, Consoli P, Perzia P, Romeo T, Andaloro F (2015) Distribution of fish fauna associated with a shipwreck in the southern Tyrrhenian Sea: vertical distribution and shipwreck structures. J Appl Ichthyol 31:96–101. https://doi.org/10.1111/jai.12954
Smith CJ, Marrs SJ, Atkinson RJA, Papadopoulou KN, Hills JM (2003) Underwater television for fisheries-independent stock assessment of Nephrops norvegicus from the Aegean (eastern Mediterranean) Sea. Mar Ecol Prog Ser 256:161–170. https://doi.org/10.3354/meps256161
Smith C, Papadopoulou KN, Machias A, Nikolioudakis N (2015) Consumption of trawl discards in the Ionian Sea: experimental results. 11th Panhellenic Symposium on Oceanography and Fisheries, Mytilene, Lesvos, Greece, pp 141–144.
Soldo A, Glavičić I (2013) Fish communities and their depth distribution at vertical corraligen reefs in the Adriatic. Rapp Comm Int Mer Médit 40:776
Soldo A, Glavičić I (2020) Underwater visual census of deeper vertical rocky reefs. Turkish J Fish Aquat Sci 20(11):785–794. https://doi.org/10.4194/1303-2712-v20_11_02
Soldo A, Glavičić I, Kovačić M (2021) Combining methods to better estimate total fish richness on temperate reefs: the case of a Mediterranean coralligenous cliff. J Mar Sci 9:670. https://doi.org/10.3390/jmse9060670
Soldo A, Glavičić I (2011) Underwater visual census (UVC) of vertical reefs. Tenth International workshop on methods for the development and evaluation of maritime technologies, Split, Croatia pp 3–12.
Spencer M, Stoner A, Ryer CH, Munk JE (2005) A towed camera sled for estimating abundance of juvenile flatfishes and habitat characteristics: comparison with beam trawls and divers. Estuar Coast Shelf Sci 64:497–503. https://doi.org/10.1016/j.ecss.2005.03.012
Stagličić N, Šegvić-Bubić T, Ugarković P, Talijančić I, Žužul I, Tičina V, Grubišić L (2017) Ecological role of bluefin tuna (Thunnus thynnus) fish farms for associated wild fish assemblages in the Mediterranean Sea. Mar Environ Res 132:79–93. https://doi.org/10.1016/j.marenvres.2017.10.015
Stern N, Badreddine A, Bitar G, Crocetta F, Deidun A, Dragičević B, Dulčić J, Durgham H, Galil BS, Galiya MY, Ikhtiyar S, Izquiredo-Muñoz A, Kassar A, Lombardo A, Lubinevsky H, Masalles D, Othman RM, Oussellam M, Pešić V, Pipitone C, Ramos-Esplá AA, Rilov G, Rothman SBS, Selfati M, Tiralongo F, Türker A, Ugarković P, Yapici S, Zava B (2019) New Mediterranean Biodiversity Records (July 2019). Mediterr Mar Sci 20(2):409–426
Stobart B, García-Charton JA, Espejo C, Rochel E, Goñi R, Reñones O, Herrero A, Crec’hriou R, Polti S, Marcos C, Planes S, Pérez-Ruzafa A (2007) A baited underwater video technique to assess shallow-water Mediterranean fish assemblages: methodological evaluation. J Exp Mar Biol 345:158–174. https://doi.org/10.1016/j.jembe.2007.02.009
Sward D, Monk J, Barrett N (2019) A Systematic review of remotely operated vehicle surveys for visually assessing fish assemblages. Front Mar Sci 6:134. https://doi.org/10.3389/fmars.2019.00134
Tassetti AN, Minelli A, Ferrà C, Guicciardi S, Gaetani A, Fabi G (2020) An integrated approach to assess fish spatial pattern around offshore gas platforms: A pilot study in the Adriatic Sea. Mar Environ Res 162:105100. https://doi.org/10.1016/j.marenvres.2020.105100
Taviani M, Angeletti L, Canese S, Cannas R, Cardone F, Cau A, Cau AB, Follesa MC, Marchese F, Montagna P, Tessarolo C (2017) The “Sardinian cold-water coral province” in the context of the Mediterranean coral ecosystems. Deep-Sea Res II 145:61–78. https://doi.org/10.1016/j.dsr2.2015.12.008
Tessier A, Pastor J, Francour P, Saragoni G, Crec’hriou R, Lenfant P, (2013) Video transects as a complement to underwater visual census to study reserve effect on fish assemblages. Aquat Biol 18:229–241. https://doi.org/10.3354/ab00506
Torres A, Abril AM, Clua EEG (2020) A time-extended (24 h) Baited Remote Underwater Video (BRUV) for monitoring pelagic and nocturnal marine species. J Mar Sci 8:208. https://doi.org/10.3390/jmse8030208
Tsikliras AC, Dinouli A, Tsiros V-Z, Tsalkou E (2015) The Mediterranean and Black Sea fisheries at risk from overexploitation. PLoS ONE 10(3):e0121188. https://doi.org/10.1371/journal.pone.0121188
Unsworth RKF, Peters JR, McCloskey RM, Hinder SL (2014) Optimising stereo baited underwater video for sampling fish and invertebrates in temperate coastal habitats. Estuar Coast Shelf Sci 150:281–287. https://doi.org/10.1016/j.ecss.2014.03.020
Ventura D, Lasinio GJ, Ardizzone G (2014) Temporal partitioning of microhabitat use among four juvenile fish species of the genus Diplodus (Pisces: Perciformes, Sparidae). Mar Ecol 36:1013–1032. https://doi.org/10.1111/maec.12198
Ventura D, Bonifazi A, Lasinio GJ, Gravina MF, Mancini E, Ardizzone G (2018) Can microscale habitat-related differences influence the abundance of ectoparasites? Multiple evidences from two juvenile coastal fish (Perciformes: Sparidae). Estuar Coast Shelf Sci 209:110–122. https://doi.org/10.1016/j.ecss.2018.05.020
Vergés A, Tomas F, Cebrian E, Ballesteros E, Kizilkaya Z, Dendrinos P, Karamanlidis AA, Spiegel D, Sala E (2014) Tropical rabbitfish and the deforestation of a warming temperate sea. J Ecol 102:1518–1527. https://doi.org/10.1111/1365-2745.12324
Vertino A, Savini A, Rosso A, Di Geronimo I, Mastrototaro F, Sanfilippo R, Gay G, Etiope G (2010) Benthic habitat characterization and distribution from two representative sites of the deep-water SML Coral Province (Mediterranean). Deep-Sea Res II 57:380–396. https://doi.org/10.1016/j.dsr2.2009.08.023
Vivó-Pons A, Alós J, Tomas F (2020) Invasion by an ecosystem engineer shifts the abundance and distribution of fish but does not decrease diversity. Mar Pollut Bull 160:111586. https://doi.org/10.1016/j.marpolbul.2020.111586
Watson DL, Harvey ES, Anderson MJ, Kendrick GA (2005) A comparison of temperate reef fish assemblages recorded by three underwater stereo-video techniques. Mar Biol 148:415–425. https://doi.org/10.1007/s00227-005-0090-6
Whitmarsh SK, Fairweather PG, Huveneers C (2017) What is Big BRUVver up to? Methods and uses of baited underwater videο. Rev Fish Biol and Fish 27:53–73. https://doi.org/10.1007/s11160-016-9450-1
Witkowski F, Vion A, Bouchoucha M (2016) Temporal partitioning of diurnal behavioural patterns of Coris julis and Diplodus vulgaris (Actinopterygii: Perciformes) in Mediterranean coralligenous habitats. Acta Ichthyol Piscat 46(3):171–183. https://doi.org/10.3750/AIP2016.46.3.02
Acknowledgements
We thank the reviewers for their critical comments that helped improve this manuscript.
Funding
Open access funding provided by HEAL-Link Greece. This research was funded by the European Union’s Horizon 2020 Research and Innovation. Horizon 2020 Framework Programme, 101000302, Athanassios C. Tsikliras
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Consent to participate
All authors consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nalmpanti, M., Chrysafi, A., Meeuwig, J.J. et al. Monitoring marine fishes using underwater video techniques in the Mediterranean Sea. Rev Fish Biol Fisheries 33, 1291–1310 (2023). https://doi.org/10.1007/s11160-023-09799-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11160-023-09799-y