Abstract
The study aims to systematically extract and analyse data about Quality of Life (QoL) in the transgender population. A systematic literature search and meta-analysis were conducted using the MEDLINE, EMBASE, PubMed, and PsycINFO databases, up to July 2017. Only English language quantitative studies, in adults, which reported the means for validated QoL measures were included. Random-effect meta-analysis was adopted to pool data and estimate the 95% Confidence Intervals (CI). From 94 potentially relevant articles, 29 studies were included within the review and data extraction for meta-analysis was available in 14 studies. The majority of the studies were cross-sectional, lacked controls and displayed moderate risk of bias. Findings from the systematic review suggested that transgender people display poor QoL, independent of the domain investigated. Pooling across studies showed that transgender people report poorer mental health QoL compared to the general population (−0.78, 95% CI = −1.08 to −0.48, 14 studies). However, meta-analysis in a subgroup of studies looking at QoL in participants who were exclusively post-CHT found no difference in mental health QoL between groups (−0.42, 95% CI = −1.15 to 0.31; 7 studies). There was insufficient data for a pre-treatment subgroup. Evidence suggests that transgender people have lower QoL than the general population. Some evidence suggests that QoL improves post-treatment. Better quality studies that include clearly defined transgender populations, divided by stage of gender affirming treatment and with appropriate matched control groups are needed to draw firmer conclusions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The term transgender (or trans) describes people whose gender identity differs from the sex they were assigned at birth based on their sexual characteristics, whilst the appellation cisgender refers to any individual who is not transgender and whose gender identity matches the sex assigned at birth [1]. Due to the mismatch between gender identity and sex assigned at birth, many transgender people experience severe distress, generally known as gender dysphoria, which tends to ameliorate following transition to the experienced gender [2].
The process of physical transition consists of different stages. Guidelines for the assessment and treatment of transgender and gender non-conforming people have been developed by the World Professional Association of Transgender Health (WPATH) to facilitate this process (Standards of Care, SOC) [2]. The SOC aims to describe the different treatments that transgender people might wish to undergo, known as Gender Affirming Treatments (GAT), which may include puberty suppression, Cross-sex Hormonal Treatment (CHT), Chest Reconstructive Surgery (CRS) and Gender Affirming Genital Surgeries (GAGSs) [2]. Thus, for the present review, the term ‘treatment’ is also used to describe GAT.
GAT produces bodily changes that impact and alter gender role and its expression by developing secondary sexual characteristics of the experienced gender in order for the body to become more congruent with the gender identity of the individual [2]. These changes might be sufficient to mitigate the gender dysphoric symptoms [2] and hence improve the individual’s QoL. However, not every transgender person requires gender affirming treatment and the dysphoria may improve through gender social role transition only. Thus, treatment might vary depending on the specific needs of the transgender person seeking treatment [2].
Many transgender people, particularly prior to their physical transition, face considerable challenges. These challenges can be physiological (development of some of the secondary sexual characteristics of the sex assigned at birth), social (lack of social support, rejection, discrimination, victimisation, transphobia) [3,4,5,6,7,8,9,10,11,12] and psychological (e.g. anxiety, depression, low self-esteem) [3, 13,14,15,16]. All these factors have been found to have a negative impact on the quality of life (QoL) of transgender people [17, 18].
QoL is a complex and broad concept. It has been described in different ways, such as the quality of one’s life conditions, one’s satisfaction with life conditions, and as a combination of life’s conditions and satisfaction [19]. De Vries and colleagues [20] defined QoL as the individuals’ perceptions of their life satisfaction and happiness that has an impact on objective and subjective wellbeing. Hence, QoL measures can be considered as a way of quantifying the level of functioning and perceived wellbeing of people’s lives [17]. The concept of QoL encompasses a range of different physical and psychosocial domains. Several factors have been shown to affect QoL in transgender populations, such as presence or absence of depression and psychopathology, transitional status (such as the use of cross sex hormone treatment), levels of social support and perceived discrimination [6, 21,22,23,24,25,26,27,28,29].
The literature regarding QoL in transgender people mainly focuses on four QoL dimensions: voice-related (vQoL); sex-related QoL; body image-related QoL; and general QoL. Voice-related QoL can be described as the impact that the perception of one’s own voice, in terms of femininity and masculinity, has on the QoL of the individual [30]. This dimension is very important for transgender people, as the pitch of the voice is an important aspect of gender expression and perception [31, 32]. Sex-related QoL is a state of social, physical and mental wellbeing related to sexual life [33]. This concept refers to the sexual functioning and general satisfaction with sexual life [34]. Body image-related QoL stems from the notion that experiencing a positive body image is linked with more satisfactory relationships, sexuality, improved well-being and overall general QoL [35]. Thus, transgender people’s incongruence between gender identity and bodily characteristics could potentially impact their body satisfaction and as a consequence their QoL [35,36,37,38]. Finally, general QoL describes the overall satisfaction with life not linked to specific physical health conditions and which includes subcategories linked to aspects of mental, physical, and social life [39].
There are mixed results regarding the QoL in the transgender population. While most of the literature suggests that transgender people have lower QoL compared to the general population [17], which increases once on CHT or post-GAGS [28, 40, 41], other studies have not replicated such findings [42, 43]. These mixed results may be explained by the lack of homogeneity in the population studies, as well as by the different types of QoL and measurements used. For instance, the effect of CHT and genital surgery on the QoL of transgender people when compared to the general population is unclear, as studies often use mixed samples in terms of treatment status and/or focus onto different states of transition. The review carried out by Murad and colleagues [27] suggested that CHT improves QoL, sexual and psychological functioning as well as gender dysphoria; however these findings are based on low quality evidence and the actual impact of both medical and social transitions upon QoL needs to be better understood [44].
Therefore, the primary aim of this this study is to conduct a critical systematic review and meta-analysis of studies of QoL in transgender populations and to explore the range of QoL assessed. The present research also aims to investigate the impact of CHT by exploring QoL in transgender people at different stages of gender transition.
Additionally, as there is a lack of understanding of the QoL domains most relevant to transgender people and of how demographic, psychosocial and treatment-related factors influence those domains, this review specifically aims to assess the different dimensions of QoL in transgender populations and their associated factors.
1.1 Eligibility criteria
Studies were included if they aimed to measure QoL in transgender populations using validated QoL tools. Articles were eligible for inclusion if they reported a mean QoL score for a transgender population and were either written in the English language or had an available translation into English. Both cross-sectional and longitudinal studies were included and there was no restrictions on settings. Studies were excluded from this systematic review if they investigated QoL in transgender children (<18 years) as QoL vary with age [45]. Additionally, articles were excluded if they had fewer than 20 participants as in small studies there is a high risk of selection bias and a lack of statistical power [46, 47]. Where different articles utilised the same database and same measures, the most recent article was taken into consideration and included within the meta-analysis. Qualitative studies, case studies, conference abstracts and review articles were also excluded. See Table 1 for summary of the review’s eligibility process.
1.2 Search strategy
PRISMA guidelines were followed [48] to carry out this review. Ovid (PubMed, EMBASE, PsycINFO) and Medline databases were searched from 1946 to July 2017. Terms for transgender people (Transgender, Transsexual, Gender Identity Disorders, and Gender Dysphoria) were searched using the OR function and combined with the terms related to (Quality of Life, QoL, Life Satisfaction) using the “AND” operator. Additionally, the reference lists of pertinent articles were searched to identify any further potential relevant papers.
1.3 Quality assessment
Risk of bias was assessed using an instrument adapted from Ibrahim et al. [49] as this instrument covered the most relevant criteria to assess risk of bias in descriptive studies. Criteria were [1] a clear definition of the target population, [2] adoption of either random, complete or consecutive recruitment or an attempt at recruiting every participant in the sampling frame, [20] sample as representative of the target population or the report presents evidence that results can be generalised to transgender people, acknowledging that most studies included treatment-seeking transgender people attending gender clinics [3] response rate equal or greater than 70%, [4] adequate sample size with a minimum of 300 participants as smaller sample sizes produce large confidence intervals and less precise results [50, 51] and [5] use of validated measures. The chosen criteria were evaluated as providing either a risk of bias (or unclear risk of bias) (1 point) or no risk of bias (0 point). Scores are then summed and an overall risk of bias rating is created where higher scores indicate greater risk of bias. Studies were rated as low risk of bias (++) (when all or most of the criteria were satisfied), moderate risk of bias (+) (when some of the criteria were satisfied) or high risk of bias (−) (when either a few or no criteria were satisfied), as per the NICE [52] guidelines for risk of bias assessment.
1.4 Data extraction
A data extraction table was used to record authors, date of publication, country where the study was conducted, participants’ information (sample sizes, mean age of sample at assessments), information on treatment status, study design, control group and follow-up (if applicable), QoL measures used, results, factors associated with QoL and conclusions. Separate tables were constructed differentiating depending on the QoL domain investigated.
1.5 Meta-analysis
Mental health-related QoL was used as the outcome of interest for the meta-analysis, as it was the most widely reported outcome and physical QoL is more sensitive to the effects of age [53]. The most frequently used QoL measures (e.g. SF-36, SF-12) do not calculate a total score but calculate separate composite scores for mental and physical health. Generic (i.e. not condition specific) mental health-QoL scores for all samples with means and Standard Deviations (SDs) reported were eligible for inclusion in the meta-analysis. When the means and SDs for a cisgender group were provided, these were used as the comparison in the meta-analysis. Where these were not available, normative data most applicable to the study country were obtained from the articles providing validation of the specific measures adopted and were used as comparison. Utilisation of normative data as a control might cause methodological concerns, as this might increase effect sizes; however, to not lose valuable data and to be able to carry out the meta-analyses, this method was deemed as the best approach. This approach was adopted for four studies [54,55,56,57].
In longitudinal studies, data from the first time point at which the participants met the age criterion for the review were used. Where studies reported incomplete results, values were either manually calculated (e.g. SDs from means) or authors were contacted to provide the missing data.
A second meta-analysis with a sub-group of studies reporting data for samples of participants who were exclusively post-GAGS, and therefore post-CHT, as the big majority of people undergoing gender affirming surgeries are already on hormonal treatment, was conducted. Pre-treatment-QoL was not assessed due to a lack of studies using exclusively pre-treatment samples. RevMan 5 [58] was utilised to conduct the meta-analyses.
It was hypothesised that the results would be heterogeneous because of differences between studies in the stages of transition investigated (e.g. mixed samples, pre-CHT, post-CHT, post-GAGS), in the diverse types of recruitment utilised (e.g. consecutive, snowballing), in the presence of clinical and/or non-clinical individuals within the samples as well as in the focus onto the different gender identities of the participants (e.g. transman, transwoman, both). Consequently, Random Effects Models (RAM) with 95% confidence interval was used for the analyses as it implies that the selected studies are carried out in diverse populations [59]. I2 statistics were calculated to examine heterogeneity, which is expressed in percentages suggesting different degrees of heterogeneity with 25% indicating low, 50% moderate and above 75% high [60]. Additionally, Q statistics were calculated to determine the statistical significance of heterogeneity [61].
1.6 QoL measures used in the review
See Table 2 for a description of the measures used in the studies to assess QoL. Voice-related QoL was assessed using the Voice Handicap Inventory (VHI) and the Transgender Self-Evaluation Questionnaire (TSEQ), sex-related QoL using the sexual subdomain of the WHOQOL-100 and the King’s Health Questionnaire (KHQ), body image-related QoL using the body image-related subdomain of the WHOQOL-100 as well as the Body Image Quality of Life Inventory (BIQLI), and generic (non-condition specific) QoL was measured using the Short Form 36 Health Survey (SF-36), version 2 of the Short Form 36 Health Survey (SF-36-v2), version 2 of the Short Form 12 Health Survey SF-12-v2, WHOQOL-100, WHOQOL-BREF, WHOQOL-BREF-TR or the Subjective Quality of Life Analysis (SQUALA). See Table 2 for a description of the measures.
2 Results
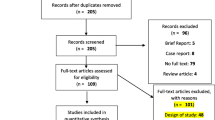
A total of 403 studies were identified through database searches, 288 through Ovid and 115 through PubMed. An additional 12 articles were selected for inclusion in the review after screening reference lists of relevant papers. After removing duplicates, 94 abstracts were screened by the first researcher (AN), which resulted in 43 studies that were read in full. Of these, fifteen were excluded due to reasons such as lack of a validated QoL measure (n = 4), of direct measurement of QoL (n = 6), of results reported specifically for transgender people (n = 4) and one study was qualitative. Finally, a sample of 29 papers was discussed, agreed with the other researchers (JA and CG) and included within this review. See Fig. 1 for description of the study’s selection process.
2.1 Study characteristics
The earliest articles included within this review were published in 2006 [17, 87] whilst the most recent papers were published in 2017 [31, 32, 55, 57, 64].
The majority of the studies were conducted in European countries (n = 20). Three studies were carried out in Spain [21, 32, 34], in France [22, 23, 66] and in Belgium [43, 65, 83]. Two studies were conducted in Italy [73, 74], UK [69, 70], the Netherlands [20, 35] and Germany [31, 55], whilst one study was carried out in Switzerland [81], one in Sweden [64] and one in Turkey [76]. With regard to non-European countries one study was from Brazil [56], one from China [54] and the remaining articles were from the USA (n = 7).
Out of the 29 included articles; a) four explored vQoL [24, 30, 31, 83], b) four looked at sex-related QoL [34, 73, 74, 81], c) three assessed body image-related QoL [35, 73, 74], and e) 22 studies measured generic (non-condition specific) QoL [2, 17, 21,22,23, 42, 43, 54,55,56,57, 64,65,66, 68,69,70,71, 73, 74, 76, 81]. With regard to vQoL, the study by Mora and colleagues [32] measured vQoL with the aid of a non-validated measure as well as general QoL with a well-validated tool, thus the article was included in the subgroup of general QoL. The study conducted by Parola and colleagues [66] was excluded from the sex-related QoL domain, as it did not employ a validated measure to assess sex-related QoL. Studies reporting generic-QoL that either separated mental and psychological subscales or provided a total QoL score (e.g. Castellano et al. – 71) were included in the systematic review. Of the four papers that measured sex-related QoL, three used the sex-related facet of the WHOQOL-100 [34, 73, 74] whilst one paper measured QoL related to incontinency in transgender women post-GAGS and was included within the sex-related QoL domain [81]. Finally, with regard to body image-related QoL, one article used a specific body image-related QoL measure (BIQLI) [35] whilst the others used the body image-facet of the WHOQOL-100 [73, 74].
In terms of study design, 22 studies were cross-sectional [17, 21,22,23,24, 30, 34, 42, 43, 54, 55, 57, 65, 66, 68,69,70,71, 73, 76, 81, 83] and seven were longitudinal [20, 31, 32, 35, 56, 64, 74], although three of the longitudinal studies [20, 31, 32] only reported cross-sectional data for QoL. Of the 29 included studies, eight compared scores of transgender people to normative data [17, 21, 34, 42, 43, 64, 65, 68], and eight compared transgender to cisgender individuals [21, 22, 30, 31, 69, 70, 74, 81] of which four studies used a matched comparison group [22, 69, 70, 74]. However for one matched study [70] the gender identity of the comparison group was unclear. Four articles compared QoL in transgender women to QoL in transgender men [23, 66, 76, 83]. The majority of studies (n = 23) recruited transgender people through clinical services [20,21,22,23, 30,31,32, 34, 35, 43, 55, 56, 64,65,66, 68,69,70, 73, 74, 76, 81, 83]. The remaining five studies recruited participants through opportunity sampling, word of mouth, flyers, advertisement and through community outreach [17, 42, 54, 57, 71].
2.2 Risk of bias
Risk of bias was evaluated for the 29 studies according to the criteria stated in Table 1. Only three studies recruited more than 300 participants [17, 42, 71] and the majority either reported response rates lower than 70% or did not mention this information (n = 21) thus increasing the risk of sampling bias [17, 20, 23, 24, 30,31,32, 34, 35, 38, 42, 43, 54, 55, 64, 65, 68, 70, 74, 81, 83]. Overall, only two studies were rated having a low risk of bias [22, 69], twenty studies had a moderate risk of bias [17, 20, 21, 23, 34, 35, 42, 54,55,56,57, 64, 65, 70, 71, 73, 74, 76, 81, 83] and seven a high risk of bias [24, 30,31,32, 43, 66, 68]. See Table 3 for details regarding studies’ quality assessment and risks of bias.
3 Results of the literature review
3.1 Voice-related QoL
Of the four papers describing vQoL, three used a cross-sectional design [24, 30, 83], whilst one article used a longitudinal design with cross-sectional data for vQoL [31]. There were no studies looking at pre-treatment transgender people. Only one study offered comparisons of transgender people post-treatment with normative data and reported worse vQoL for transgender people when compared to controls [31]. The cross-sectional studies that looked at people post-treatment (GAGS, CHT, Voice Feminisation Treatment - VFT) found transgender people to experience voice-related disability, in that they feel handicapped in everyday life because of their voice [31, 83]. This could be due to the fact that hormone therapy for transgender women had not affected on their voice. Only one study compared people according to their gender identity; this study by T’Sjoen and colleagues [83] found that transgender men report better vQoL compared to transgender women post-GAT. Overall, vQoL appears to be worse in transgender people, particularly in women, even post-GAT. See Table 4 for details.
The few studies investigating predictors of vQoL found increased age of the transgender individual, increased femininity of the voice [24] and low dihydrotestosterone as well as high Luteinising Hormone (LH) in the blood [83] to be factors predictive of a positive vQoL in populations of transgender women.
3.2 Sex-related QoL
Four studies investigated sex-related QoL by adopting a cross-sectional design [34, 73, 74, 81]; one offered comparisons with normative data [34], one compared the transgender group with a cisgender group matched for experienced gender [73], one carried out comparisons between transgender men and transgender women as well as between pre- and post-CHT [74] and the fourth study compared QoL linked to incontinence in transgender people post-GAGS to twenty members of the clinical staff who underwent at least one previous abdominal or pelvic operation [81]. This last study was included within this section as it is linked to surgery outcomes and to satisfaction with sexual life.
Only one study looked at a transgender sample pre-GAGS and found that transgender people report worse sex-related QoL than the general population [34]. Studies including people post-GAGS suggested that transgender people still experienced lower sex-related QoL than their matched controls [73, 81]. When looking at gender differences, Castellano et al. [73] suggested that, at post-GAGS, transgender women did not display significantly different sex-related QoL compared to cisgender women, whilst transgender men showed lower sex-related QoL than cisgender men. Instead, studies comparing transgender people according to gender identity reported a significantly lower sex-related QoL in transgender men when compare to transgender women, independently of the transitional status [34, 73]. Only one study described changes in sex-related QoL using a longitudinal methodology [74]. This study found a significant improvement in sex-related QoL for both transgender men and transgender women post-CHT [74]. Overall, it appears that sex-related QoL improves post-GAT. However, such appears to be poor, particularly in transgender men when compared to cisgender men. See Table 5 for details.
Regarding predictors of sex-related QoL, CHT [34], low LH in the blood [73], having a partner and experiencing less negative mood symptoms [34] have been found to be factors associated with more positive sex-related QoL.
3.3 Body image-related QoL
Three papers described body image-related QoL, however none of these studies investigated QoL pre-GAT [35, 73, 74]. The cross-sectional study conducted by Castellano et al. [73] found no difference in body image-related QoL between the transgender sample post-medical treatments and a matched cisgender sample. The two longitudinal studies reported an improvement in body image-related QoL after treatment, specifically after CHT [74] and mastectomy for transgender men [35]. This suggests that gender affirming treatments are of benefit to body image-related QoL in transgender samples. The limited research in this area shows that body image related QoL improves post-GAT. See Table 6 for details.
Only one study looked at factors associated to body image quality of life and found low levels of LH in the blood to be associated to a positive body image-related QoL [73].
3.4 General (non-condition specific) QoL
Out of the 22 studies that assessed generic (non-condition specific) QoL, there were no cross-sectional studies looking specifically at people pre-GAT. Five studies investigated post-GAT [20, 43, 66, 68, 73], twelve were mixed in term of treatment status [17, 21,22,23, 42, 54, 55, 65, 69,70,71, 76], three were longitudinal [56, 64, 74], one did not report information on treatment status [57] and one study reported only that participants were pre-facial feminisation treatment [32].
There is pre-CHT data available from the longitudinal studies showing that transgender people have lower QoL than the general population [56, 74]. Of the six cross-sectional studies looking specifically at people pre-GAGS [21,22,23, 54, 68, 71], two studies found transgender people to report poorer QoL than the general population [21, 68] and one found similar scores to their matched cisgender controls [22]. The remaining studies did not include comparisons with the general population but they suggested that transgender people report poor QoL.
Five studies investigated QoL at post-GAGS; four studies found that transgender people at this stage still report lower QoL than the general population [43, 66, 68] whilst two studies suggested transgender people to display similar QoL to the general population [20, 73].
With regard to mixed samples, one study suggested transgender people report worse QoL than their matched cisgender controls [69], two reported poorer QoL than the cisgender non-matched controls [21, 70] and one study suggested worse QoL than the general population [65]. Studies looking at people according to their gender found that some of the results regarding QoL in transgender men were contradictory; one study indicated that they suffer from worse QoL than the general population [17] whilst another study suggested the opposite [42]. Transgender men were also found to report a better QoL when undergoing CHT than when not on hormones [71]. Instead, transgender women displayed high physical health-related QoL and poor mental-health related QoL [54]. The remaining three mixed sample studies made comparisons between transgender men and transgender women, and the results are inconsistent. Two studies found that transgender women report better QoL than transgender men [23, 76] and one found no difference between transgender men and transgender women [55]. In another mixed study, Motmans and colleagues [65] found that transgender men had worse QoL than the cisgender population. However the results of the above mixed sample studies need to be interpreted carefully.
Regarding the three longitudinal studies [56, 64, 74], one found an improvement in QoL 1-year post-CHT when compared to pre-CHT levels [74]. A different study also found an improvement in transgender women 1-year post-GAGS when compare to pre-CHT values [56]. The third study compared QoL pre-GAGS (on CHT) and 1, 3 and 5-years post-GAGS. This study found that although QoL pre-GAGS was lower than the general population it improves 1-year post-GAGS. However the study also found that it reduces 3 years post-GAGS and even more 5 years after genital surgery. This could be explained as the first year post-GAGS is often known as the “honeymoon period” and people tend to report overly enhanced QoL, which are not representative of a long-term picture of patients’ psychological status and QoL [43]. When investigating longitudinal results according to gender a study found that transgender women displayed greater improvements in QoL 1-year post-CHT compared to transgender men [74].
Overall, the studies investigating general QoL in transgender people found poorer QoL pre-GAT than the general population, which improve after GAT in the short term. See Table 7 for details.
Medical and surgical treatments (i.e. CHT, CRS, GAGS) [17, 21,22,23, 56, 66, 68, 71], post-surgical well-being [20] and sexual functioning [43], presence of social and family support [21, 42, 55, 70, 77], decreased depression, anxiety and stress levels [42, 55], lack of chronic pain symptomatology [55], hope and resilience [54], high self-esteem and low levels of interpersonal issues [69], lack of identity stigma [57], having a good body image and good sleep quality [55], low levels of LH in the blood [73] as well as being employed and in a relationship, younger age, higher education, a high household income [65] and having undergone military service [57] were found to be factors predictive of a positive QoL.
4 Results of the meta-analysis
4.1 Meta-analysis – Mental health-related QoL of transgender people compared to the general population
Measurements of QoL provide information regarding physical and mental health-related QoL but only a minority of studies looked at physical health-related QoL; therefore the meta-analysis focused on mental health-related QoL compared to those of the general population. Of the 22 studies assessing general QoL in transgender populations, 14 were considered suitable for inclusion in the meta-analysis [17, 20,21,22, 43, 54,55,56,57, 64, 65, 68,69,70]. These studies include people pre-GAT, post-GAT and mixed groups at different stages of medical transition. Studies were excluded from meta-analysis due to the absence of the mean, SD and/or sample size [66, 74], mental health quality of life not reported separately [73], insufficient detail about scoring [32, 42, 71] and the lack of access to appropriate normative data [23, 76]. Data at pre-treatment were utilised for the two longitudinal studies included in the meta-analysis [56, 64]. Additionally, normative data as comparison was obtained for four studies [54,55,56,57].
The results of the meta-analysis (14 studies) showed that transgender people report a statistically significantly lower mental health-related QoL than the general population (standard mean difference − 0.78, 95% CI = −1.08 to −0.48, Z = 5.16, p < 0.00001). Heterogeneity was high (I2 = 97%, p < 0.00001) (see Fig. 2).
4.2 Meta-analysis – Subgroup analysis – Mental health-related QoL post-hormonal treatment of transgender people compared to the general population
A second meta-analysis was conducted with only the 7 studies that included exclusively post-treatment QoL scores [20, 43, 56, 64, 65, 68, 70]. The longitudinal study of Lindqvist et al. [64] investigated QoL post-GAGS but as the first time measurement was pre-GAGS and thus post-CHT, this measure was included in this analysis. Whilst for the longitudinal study of de Vries et al. [20], values at the latest time-point were used, as they measured QoL post-CHT in a sample of individuals older than 17 years of age.
The meta-analysis of 7 studies found that there was no statistically significant difference in mental –health related QoL of transgender people following CHT compared to the general population (standard mean difference = −0.42 CI 95% = −1.15 to 0.31; Z = 1.13; p = 0.26). Heterogeneity was high (I2 = 98%; p < 0.00001) (see Fig. 3).
5 Discussion
The aim of this study was to systematically and critically review the literature pertaining to quality of life in transgender people, to meta-analytically investigate mental-health related QoL compared to cisgender populations and to investigate the impact of GAT on the QoL of this population. A total of 29 studies met the inclusion criteria and were used for the systematic review and, of these, 14 studies were suitable for including in the meta-analysis. Most papers in this area investigated general QoL and only a few focused on either vQoL, sex-related QoL or body image-related QoL. The majority of these articles displayed either high or moderate risk of bias. Many studies used transgender samples that are not homogeneous in terms of gender affirming medical treatment status, which makes it difficult to draw firm conclusions about the impact of GAT.
Findings from the meta-analysis of mental health-related QoL suggest that the QoL of transgender people is significantly poorer than that of the general population, with a medium to large effect size (standard mean difference = 0.78). The subgroup meta-analysis including only the samples of transgender people who were classifiable as post-hormonal treatment found that transgender people post-CHT still had lower mental health-related QoL than the general population. This difference was not significant and the effect size was reduced (standard mean difference = 0.42). The possibility that treatment is associated with improvements in mental wellbeing is supported by the findings from the small number of longitudinal studies in this review. These found that both CHT and GAGS improve QoL [56, 64, 74]. However these results need to be treated with caution, as the only study to employ a longer term follow-up [64] reported that after an initial improvement in QoL at 1-year post-GAGS, scores tend to steadily decrease in the following years until reaching 5-years post-GAGS, when QoL is lower than at pre-treatment [64]. During the first year post-GAGS people tend to report overly enhanced QoL, which may not be representative of a long-term picture of patients’ psychological status and QoL [43]. The improvement in QoL experienced by transgender people at short-term could be attributed to relief at being able to live as the experienced gender. Additionally, as QoL in the general population has been shown to decrease with age [53], a decline in these scores is somewhat expected as time passes.
In contrast, the small number of studies that explore general physical health-related QoL suggest that at post-GAT, transgender people’s reported QoL scores either similar to [22] or better than that found in the general population [17, 43]. However, only a minority of studies report findings related to physical health-related QoL and it is difficult to draw accurate conclusions.
5.1 Condition specific QoL
When looking at condition-specific QoL, studies investigating vQoL reported that CHT has been shown to have a positive impact on transgender men. This is not surprising, as testosterone is known to affect voice by thickening vocal chords and by decreasing the pitch [87]. On the other hand, studies in transgender women, including post-voice feminising surgery, found that they still feel handicapped regarding their voice in their everyday life, irrespective of the transitional status. In fact, studies have suggested that the more feminine a transgender person perceives her own voice, the higher the experienced vQoL [30, 31]. However, these studies are limited by focusing on transgender women who transitioned post-puberty. This means that by the time they initiated physical transition, testosterone has already negatively altered their voice. Thus findings from vQoL cannot be generalised to the overall transgender population. Future studies should explore differences in vQoL between those who transitioned pre-puberty and therefore before the breaking of the voice, and those who transitioned post-puberty, when the voice has already been affected.
Regarding sex-related QoL, longitudinal studies suggest that undergoing GAT improves sex-related QoL [74] but the QoL of transgender men post-GAGS is still worse than that of cisgender men. However, the articles investigating sex-related QoL in transgender men did not distinguish whether patients underwent phalloplasty or metoidioplasty. Surgical treatments help the transgender person reach the desired physical changes and lead towards a more congruous body with their gender identity. This may lead to people feeling more comfortable with their own bodies and consequently when being intimate with others.
Longitudinal studies also supported an amelioration in body image-related QoL [35, 74]. In fact, Castellano and colleagues [73] reported no difference between the transgender population and their matched controls. This could be due to the fact that people undergoing GAGS are generally already on CHT and hormonal treatment is known to have a positive effect on body image [10, 36, 88] by aiding in the development of desired secondary sexual characteristics of the experienced gender, whilst helping to alter some of the attributes relative to the sex assigned at birth. Consequently, this leads to an improvement in body image-related QoL. Nonetheless, van de Grift et al. [35] proposed body image-related QoL to be lower for transgender people post-CRS, than for the general population. This might be caused by the fact that following CRS, some people’s genital dysphoria may increase. However, caution is still needed when generalising the findings due to the studies’ moderate risk of bias as well as their methodological limitations.
5.2 General QoL
Studies regarding general QoL have found that transgender people’s QoL is poorer than that of cisgender people, but that it improves post-GAT. The poorer QoL found in the transgender population pre-CHT [56, 74] could be explained by the high degree of mental health problems reported in this population [16, 89] as well as by the difficulties that many have in socialising and living a fulfilling life [10,11,12, 37]. However, the studies that focus only on people pre-CHT were rare and only included those seeking medical transition, which does not allow for a generalisation of these findings to the general transgender population.
When looking at general QoL post-GAT, only a small number of studies provided control data and none of them had a low risk of bias. Findings of the subgroup meta-analysis at post-treatment showed that there is no difference in general QoL between transgender people and the general population. The improvement in QoL post-GAT could be due to the effect of treatment in the reduction of dysphoria and mental health problems, such as self-harm and depression [13, 90].
Overall, findings support the idea that QoL improves following hormonal treatment [74] as well as post-genital surgeries [43, 56]. As people undergoing surgery are generally already on hormones, the exact role of genital surgery in QoL cannot be extrapolated from these studies. Often, even after an improvement in QoL post-surgical treatment, transgender people reported lower QoL compared to cisgender individuals [7, 56, 64, 70]. This could be due to the fact that, even if being happier with their own bodies, society is still not ready to accept transgender people; thus work, education or relationships can be affected by being transgender [6, 91, 92]. This is confirmed by the findings of studies investigating factors predictive of QoL in this population, as described in the section below.
Caution is needed while interpreting the results in comparison to cisgender individuals as not all studies have matched controls and sample sizes are generally small. Additionally, as the majority of the studies investigated QoL in transgender clinical populations, generalisation of findings for the general transgender population is hindered.
Studies on differences between transgender men and transgender women advance contrasting results. Two studies seemed to suggest that transgender men display lower QoL compared to transgender women [23, 76], two studies suggested the opposite [21, 66], whilst one study proposed no statistically significant differences between groups [55]. Literature also suggested that at 2-years post-treatment transgender men display higher QoL than transgender women [66], whilst still lower than cisgender people [73]. These findings might be due to baseline differences in QoL scores [65] as well as because of the utilisation of mixed samples in terms of treatment status. Results need to be interpreted with caution as none of the articles displayed a low risk of bias. A possible explanation for transgender men to report higher QoL than transgender women might be due to the wider social acceptance towards masculinity than femininity. This presents itself with transgender men reporting less marked psychopathology, getting involved more easily in society and being employed in more stable jobs, whilst feeling less limitations in daily life related to their physical and emotional state [21, 66]. Additionally, studies that reported transgender women to display higher QoL than transgender men suggested that these findings are unexpected and surprising [76] considering the low social status of and amount of discrimination faced by transgender women in some countries (e.g. Turkey).
5.3 Factors associated with QoL
QoL can be influenced by a wide array of factors, which can predict both its increment, as well as its decline. Literature looking at variables associated with a positive QoL for transgender people suggested that undergoing medical and surgical treatments (i.e. CHT, CRS, GAGS) are the main predictive factors, irrespective of the QoL domain studied [17, 21,22,23, 34, 66, 68, 71]. These findings were confirmed by longitudinal studies, which indicated an improvement in QoL from pre- to post-treatment [35, 56, 74].
Additionally, social and family support, being employed, being in a relationship, being younger, having a partner, being highly educated, having a high household income, and the presence of past military service were associated with improved scores on general and sex-related QoL [21, 34, 54, 57, 65].
Instead, anxiety, poor sleep quality, experiencing pain, reduced self-esteem and high interpersonal issues are factors that have been linked to poor QoL in both transgender populations [22, 42, 55, 69, 70, 76] as well as in the general population [93].
6 Conclusion
As all systematic literature reviews, this study is also limited by the amount and quality of the published literature available. Future studies should employ more robust methodologies, which explore QoL in a more homogeneous population and using matched control groups.
Despite the limitations of the published literature, this review concludes that overall transgender people display poorer QoL than the general population, particularly pre-GAT, and that QoL improves once people are on CHT.
When specifically looking at the different dimensions of QoL (vQoL, sex-related QoL, and body image-related QoL), findings of the systematic review suggest that transgender people display poorer QoL than the general population, independent of the QoL domain investigated. As per general QoL, all dimensions of QoL have been shown to improve post-GAT. However, as the effect of GAT is linked to gender, a more positive vQOL was found for transgender men than transgender women at post-GAT, whilst opposite findings were obtained for sex-related QoL.
As long-term follow-up studies are limited in numbers and methodology, more studies are required exploring long-term QoL. This information may aid the development of support and interventions aiming at increasing resilience for those at risk of a poor QoL post-GAT.
References
Bouman WP, Schwend AS, Motmans J, Smiley A, Safer JD, Deutsch MB, et al. Language and trans health. Int J Transgenderism. 2017;18(1):1–6.
Coleman E, Bockting W, Botzer M, Cohen-Kettenis P, DeCuypere G, Feldman J, et al. Standards of care for the health of transsexual, transgender, and gender-nonconforming people, version 7. Int J Transgenderism. 2012;13(4):165–232.
Budge SL, Adelson JL, Howard KA. Anxiety and depression in transgender individuals: the roles of transition status, loss, social support, and coping. J Consult Clin Psychol. 2013;81(3):545–57.
Clements-Nolle K, Marx R, Katz M. Attempted suicide among transgender persons: the influence of gender-based discrimination and victimization. J Homosex. 2006;51(3):53–69.
Davey A, Arcelus J, Meyer C, Bouman WP. Self-injury among trans individuals and matched controls: prevalence and associated factors. Health Soc Care Community. 2016;24(4):485–94.
Kattari SK, Whitfield DL, Walls NE, Langenderfer-Magruder L, Ramos D. Policing gender through housing and employment discrimination: comparison of discrimination experiences of transgender and cisgender LGBQ individuals. J Soc Social Work Res. 2016;7(3):427–47.
Nemoto T, Bödeker B, Iwamoto M. Social support, exposure to violence and transphobia, and correlates of depression among male-to-female transgender women with a history of sex work. Am J Public Health. 2011;101(10):1980–8.
Rotondi NK, Bauer GR, Scanlon K, Kaay M, Travers R, Travers A. Prevalence of and risk and protective factors for depression in female-to-male transgender Ontarians: trans PULSE project. Can J Commun Ment Health. 2012;30(2):135–55.
White Hughto JM, Reisner SL, Pachankis JE. Transgender stigma and health: a critical review of stigma determinants, mechanisms, and interventions. Soc Sci Med. 2015;147:222–31.
Jones BA, Haycraft E, Bouman WP, Brewin N, Claes L, Arcelus J. Risk factors for eating disorder psychopathology within the treatment seeking transgender population: the role of cross-sex hormone treatment. Eur Eat Disord Rev. 2018;26:120–8. https://doi.org/10.1002/erv.2576.
Jones BA, Haycraft E, Bouman WP, Arcelus J. The levels and predictors of physical activity engagement within the treatment-seeking transgender population: a matched control study. J Phys Act Health. 2017;20:1–9. https://doi.org/10.1123/jpah.2017/0298.
Jones BA, Arcelus J, Bouman WP, Haycraft E. Barriers and facilitators of physical activity and sport participation among young transgender adults who are medically transitioning. Int J Transgenderism. 2017;18(2):227–38.
Dhejne C, Van Vlerken R, Heylens G, Arcelus J. Mental health and gender dysphoria: a review of the literature. Int Rev Psychiatry. 2016;28(1):44–57.
Leibowitz S, de Vries AL. Gender dysphoria in adolescence. Int Rev Psychiatry. 2016;28(1):21–35.
Marshall E, Claes L, Bouman WP, Witcomb GL, Arcelus J. Non-suicidal self-injury and suicidality in trans people: a systematic review of the literature. Int Rev Psychiatry. 2016;28(1):58–69.
Millet N, Longworth J, Arcelus J. Prevalence of anxiety symptoms and disorders in the transgender population: a systematic review of the literature. Int J Transgenderism. 2017;18(1):27–38.
Newfield E, Hart S, Dibble S, Kohler L. Female-to-male transgender quality of life. Qual Life Res. 2006;15(9):1447–57.
Fredriksen-Goldsen KI, Kim HJ, Shiu C, Goldsen J, Emlet CA. Successful aging among LGBT older adults: physical and mental health-related quality of life by age group. Gerontologist. 2014;55(1):154–68.
Borthwick-Duffy SA. Quality of life and quality of care in mental retardation. In: Mental retardation in the year 2000. New York: Springer; 1992. p. 52–66.
De Vries AL, McGuire JK, Steensma TD, Wagenaar EC, Doreleijers TA, Cohen-Kettenis PT. Young adult psychological outcome after puberty suppression and gender reassignment. Pediatrics. 2014;134(4):696–704.
Gómez-Gil E, Zubiaurre-Elorza L, De Antonio IE, Guillamon A, Salamero M. Determinants of quality of life in Spanish transsexuals attending a gender unit before genital sex reassignment surgery. Qual Life Res. 2014;23(2):669–76.
Gorin-Lazard A, Baumstarck K, Boyer L, Maquigneau A, Gebleux S, Penochet JC, et al. Is hormonal therapy associated with better quality of life in transsexuals? A cross-sectional study. J Sex Med. 2012;9(2):531–41.
Gorin-Lazard A, Baumstarck K, Boyer L, Maquigneau A, Penochet JC, Pringuey D, et al. Hormonal therapy is associated with better self-esteem, mood, and quality of life in transsexuals. J Nerv Ment Dis. 2013;201(11):996–1000.
Hancock AB. An ICF perspective on voice-related quality of life of American transgender women. J Voice. 2016;31(1):115–e1.
Jokić-Begić N, Lauri Korajlija A, Jurin T. Psychosocial adjustment to sex reassignment surgery: a qualitative examination and personal experiences of six transsexual persons in Croatia. Sci World J. 2014;
Lawrence AA. Factors associated with satisfaction or regret following male-to-female sex reassignment surgery. Arch Sex Behav. 2003;32(4):299–315.
Murad MH, Elamin MB, Garcia MZ, Mullan RJ, Murad A, Erwin PJ, et al. Hormonal therapy and sex reassignment: a systematic review and meta-analysis of quality of life and psychosocial outcomes. Clin Endocrinol. 2010;72(2):214–31.
Smith YL, Van Goozen SH, Kuiper AJ, Cohen-Kettenis PT. Sex reassignment: outcomes and predictors of treatment for adolescent and adult transsexuals. Psychol Med. 2005;35(1):89–99.
White Hughto JM, Reisner SL. A systematic review of the effects of hormone therapy on psychological functioning and quality of life in transgender individuals. Transgender Health. 2016;1(1):21–31.
Hancock AB, Krissinger J, Owen K. Voice perceptions and quality of life of transgender people. J Voice. 2011;25(5):553–8.
Meister J, Hagen R, Shehata-Dieler W, Kühn H, Kraus F, Kleinsasser N. Pitch elevation in male-to-female transgender persons—the Würzburg approach. J Voice. 2017;31(2):244–e7.
Mora E, Carrillo A, Giribet A, Becerra A, Lucio MJ, Cobeta I. Translation, cultural adaptation, and preliminary evaluation of the Spanish version of the transgender voice questionnaire for male-to-female transsexuals (TVQ MtF). J Voice 2017 Jun 7. Retrieved from https://doi.org/10.1016/j.jvoice.2017.05.012.
World Health Organization. Defining sexual health: Report of a technical consultation on sexual health, 28–31 January 2002 [Internet].
Bartolucci C, Gómez-Gil E, Salamero M, Esteva I, Guillamón A, Zubiaurre L, et al. Sexual quality of life in gender-dysphoric adults before genital sex reassignment surgery. J Sex Med. 2015;12(1):180–8.
van de Grift TC, Kreukels BP, Elfering L, Özer M, Bouman MB, Buncamper ME, et al. Body image in transmen: multidimensional measurement and the effects of mastectomy. J Sex Med. 2016;13(11):1778–86.
Witcomb GL, Bouman WP, Brewin N, Richards C, Fernandez-Aranda F, Arcelus J. Body image dissatisfaction and eating-related psychopathology in trans individuals: a matched control study. Eur Eat Disord Rev. 2015;23(4):287–93.
Jones BA, Arcelus J, Bouman WP, Haycraft E. Sport and transgender people: a systematic review of the literature relating to sport participation and competitive sport policies. Sports Med. 2017;47(4):701–16.
Jones BA, Haycraft E, Murjan S, Arcelus J. Body dissatisfaction and disordered eating in trans people: a systematic review of the literature. Int Rev Psychiatry. 2016;28(1):81–94.
WHO Group. The World Health Organization quality of life assessment (WHOQOL): development and general psychometric properties. Soc Sci Med. 1998;46(12):1569–85.
de Cuypere GR, Elaut E, Heylens GU, Van Maele GE, Selvaggi G, T’Sjoen G, et al. Long-term follow-up: psychosocial outcome of Belgian transsexuals after sex reassignment surgery. Theol Sex. 2006;15(2):126–33.
Rehman J, Lazer S, Benet AE, Schaefer LC, Melman A. The reported sex and surgery satisfactions of 28 postoperative male-to-female transsexual patients. Arch Sex Behav. 1999;28(1):71–89.
Colton Meier SC, Pardo ST, Labuski C, Babcock J. Measures of clinical health among female-to-male transgender persons as a function of sexual orientation. Arch Sex Behav. 2013;42(3):463–74.
Wierckx K, Van Caenegem E, Elaut E, Dedecker D, Van de Peer F, Toye K, et al. Quality of life and sexual health after sex reassignment surgery in transsexual men. J Sex Med. 2011;8(12):3379–88.
Bockting W, Coleman E. Developmental stages of the transgender coming-out process. Principles of transgender medicine and surgery. 2016 May 27:137.
Skevington SM, Lotfy M, O'Connell KA. The World Health Organization's WHOQOL-BREF quality of life assessment: psychometric properties and results of the international field trial. A report from the WHOQOL group. Qual Life Res. 2004;13(2):299–310.
Button KS, Ioannidis JP, Mokrysz C, Nosek BA, Flint J, Robinson ES, et al. Confidence and precision increase with high statistical power. Nat Rev Neurosci. 2013;14(8):585.
Dickersin K. The existence of publication bias and risk factors for its occurrence. JAMA. 1990;263(10):1385–9.
Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 2015;4(1):1.
Ibrahim AK, Kelly SJ, Adams CE, Glazebrook C. A systematic review of studies of depression prevalence in university students. J Psychiatr Res. 2013 Mar 31;47(3):391–400.
Kulig M, Perleth M, Langer G, Meerpohl JJ, Gartlehner G, Kaminski-Hartenthaler A, et al. GRADE guidelines: 6. Rating the quality of evidence–imprecision. Z Evidenz Fortbild Qual Gesundheitswes. 2012;106(9):677–88.
Loney PL, Chambers LW, Bennett KJ, Roberts JG, Stratford PW. Critical appraisal of the health research literature prevalence or incidence of a health problem. Chronic Dis Inj Can. 1998;19(4):170.
NICE. The Guidelines Manual. London: National Institute for Health and Care Excellence. Retrieved from http://www.nice.org.uk/article/pmg6/chapter/6-reviewing-the-evidence/
Hawthorne G, Herrman H, Murphy B. Interpreting the WHOQOL-BREF: preliminary population norms and effect sizes. Soc Indic Res. 2006 May 1;77(1):37–59.
Yang X, Zhao L, Wang L, Hao C, Gu Y, Song W, et al. Quality of life of transgender women from China and associated factors: a cross-sectional study. J Sex Med. 2016;13(6):977–87.
Auer MK, Liedl A, Fuss J, Nieder T, Briken P, Stalla GK, et al. High impact of sleeping problems on quality of life in transgender individuals: a cross-sectional multicenter study. PLoS One. 2017;12(2):e0171640.
Cardoso da Silva DC, Schwarz K, Fontanari AM, Costa AB, Massuda R, Henriques AA, et al. WHOQOL-100 before and after sex reassignment surgery in Brazilian male-to-female transsexual individuals. J Sex Med. 2016;13(6):988–93.
Hoy-Ellis CP, Shiu C, Sullivan KM, Kim HJ, Sturges AM, Fredriksen-Goldsen KI. Prior military service, identity stigma, and mental health among transgender older adults. The Gerontologist. 2017;57(suppl_1):S63–71.
Review Manager (RevMan) [Computer program]. Version 5. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Deeks JJ, Higgins J, Altman DG. Analysing data and undertaking meta-analyses. Cochrane handbook for systematic reviews of interventions: Cochrane book series. 2008:243–96.
Higgins JP, Green S. Cochrane handbook for systematic reviews of interventions: Wiley; 2011.
Huedo-Medina TB, Sánchez-Meca J, Marín-Martínez F, Botella J. Assessing heterogeneity in meta-analysis: Q statistic or I2 index? Psychol Methods. 2006;11(2):193–206.
Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med Care. 1992:473–83.
Ware JE Jr. SF-36 health survey update. Spine. 2000;25(24):3130–9.
Lindqvist EK, Sigurjonsson H, Möllermark C, Rinder J, Farnebo F, Lundgren TK. Quality of life improves early after gender reassignment surgery in transgender women. Eur J Plast Surg. 2017;40(3):223–6.
Motmans J, Meier P, Ponnet K, T'sjoen G. Female and male transgender quality of life: socioeconomic and medical differences. J Sex Med. 2012;9(3):743–50.
Parola N, Bonierbale M, Lemaire A, Aghababian V, Michel A, Lançon C. Study of quality of life for transsexuals after hormonal and surgical reassignment. Theol Sex. 2010;19(1):24–8.
Ware JE, Kosinski M, Bjorner JB, Turner-Bowker DM, Gandek B, Maruish ME. sf-36v2 health survey: Administration guide for clinical trial Investigators. Lincoln: QualityMetric Incorporated. Lincoln: Quality Metric. 2008. p. 34.
Ainsworth TA, Spiegel JH. Quality of life of individuals with and without facial feminization surgery or gender reassignment surgery. Qual Life Res. 2010;19(7):1019–24.
Bouman WP, Davey A, Meyer C, Witcomb GL, Arcelus J. Predictors of psychological well-being among treatment seeking transgender individuals. Sex Relation Ther. 2016;31(3):359–75.
Davey A, Bouman WP, Arcelus J, Meyer C. Social support and psychological well-being in gender dysphoria: a comparison of patients with matched controls. J Sex Med. 2014;11(12):2976–85.
Colton Meier SL, Fitzgerald KM, Pardo ST, Babcock J. The effects of hormonal gender affirmation treatment on mental health in female-to-male transsexuals. Journal of gay & lesbian mental health. 2011;15(3):281–99.
Ware JE, Kosinski Jr M, Turner Bowker DM, Gandek B. How to score version 2 of the SF-12v2® health survey (with a supplement documenting SF-12® health survey). Lincoln, RI: Quality Metric. 2002.
Castellano E, Crespi C, Dell’Aquila C, Rosato R, Catalano C, Mineccia V, et al. Quality of life and hormones after sex reassignment surgery. J Endocrinol Invest. 2015;38(12):1373–81.
Manieri C, Castellano E, Crespi C, Di Bisceglie C, Dell’Aquila C, Gualerzi A, et al. Medical treatment of subjects with gender identity disorder: the experience in an Italian public health center. International journal of transgenderism. 2014;15(2):53–65.
Eser E, Fidaner H, Fidaner C, Eser SY, Elbi H, Göker E. Psychometric properties of the WHOQOL-100 and WHOQOL-BREF. J Psychiatry Psychol Psychopharmacol. 1999;7(2):23–40. [Turkish].
Başar K, Öz G, Karakaya J. Perceived discrimination, social support, and quality of life in gender dysphoria. J Sex Med. 2016;13(7):1133–41.
Zannotti M, Pringuey D. A method for quality of life assessment in psychiatry: the S-QUA-LA (subjective QUAlity of life analysis). Qual Life Newsletter. 1992;4(6):1992.
Chrastina J, Ivanová K, Krejčířová O, Ježorská Š. Results of subjective quality of life research using SQUALA standardized questionnaire. Profese Online. 2014;7(1):1–8.
Reese PR, Pleil AM, Okano GJ, Kelleher CJ. Multinational study of reliability and validity of the King's health questionnaire in patients with overactive bladder. Qual Life Res. 2003;12(4):427–42.
Kelleher CJ, Cardozo LD, Khullar V, Salvatore S. A new questionnaire to assess the quality of life of urinary incontinent women. BJOG Int J Obstet Gynaecol. 1997;104(12):1374–9.
Kuhn A, Bodmer C, Stadlmayr W, Kuhn P, Mueller MD, Birkhäuser M. Quality of life 15 years after sex reassignment surgery for transsexualism. Fertil Steril. 2009;92(5):1685–9.
Jacobson BH, Johnson A, Grywalski C, Silbergleit A, Jacobson G, Benninger MS, et al. The voice handicap index (VHI): development and validation. Am J Speech Lang Pathol. 1997;6(3):66–70.
T'Sjoen G, Moerman M, Van Borsel J, Feyen E, Rubens R, Monstrey S, et al. Impact of voice in transsexuals. Int J Transgenderism. 2006;9(1):1–7.
Davies S, Adler RK, Hirsch S, Mordaunt M. Transgender Self-Evaluation Questionnaire. Voice and communication therapy for the transgender/transsexual client: A comprehensive clinical guide. 2006:485.
Hancock AB, Owen K, Seigfriedt L, Brundage S. Reliability and validity of the TSEQ. In World Professional Association of Transgender Health conference, Oslo, Norway 2009.
Cash TF, Fleming EC. The impact of body image experiences: development of the body image quality of life inventory. Int J Eat Disord. 2002;31(4):455–60.
Perrone AM, Cerpolini S, Maria Salfi NC, Ceccarelli C, De Giorgi LB, Formelli G, et al. Effect of long-term testosterone administration on the endometrium of female-to-male (FtM) transsexuals. J Sex Med. 2009;6(11):3193–200.
Fisher AD, Castellini G, Bandini E, Casale H, Fanni E, Benni L, et al. Cross-sex hormonal treatment and body uneasiness in individuals with gender dysphoria. J Sex Med. 2014;11(3):709–19.
Bouman WP, Claes L, Brewin N, Crawford JR, Millet N, Fernandez-Aranda F, et al. Transgender and anxiety: a comparative study between transgender people and the general population. Int J Transgenderism. 2017;18(1):16–26.
Witcomb GL, Bouman WP, Claes L, Brewin N, Crawford JR, Arcelus J. Levels of depression in transgender people and its predictors: results of a large matched control study with transgender people accessing clinical services. J Affect Disord. 2018;235:308–15.
Levitt HM, Ippolito MR. Being transgender: navigating minority stressors and developing authentic self-presentation. Psychol Women Q. 2014;38(1):46–64.
Lombardi EL, Wilchins RA, Priesing D, Malouf D. Gender violence: transgender experiences with violence and discrimination. J Homosex. 2002;42(1):89–101.
Schalock RL. The concept of quality of life: what we know and do not know. J Intellect Disabil Res. 2004;48(3):203–16.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Nobili, A., Glazebrook, C. & Arcelus, J. Quality of life of treatment-seeking transgender adults: A systematic review and meta-analysis. Rev Endocr Metab Disord 19, 199–220 (2018). https://doi.org/10.1007/s11154-018-9459-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-018-9459-y