Abstract
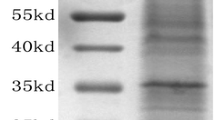
Propolis is a natural mixture of honeybee-released and plant-derived compounds produced by honeybees. Poplar propolis is rich in bioactive polyphenolic compounds, and due to its many health benefits, it is commonly used as a food supplement or functional food ingredient. However, it is the only honeybee product whose proteome hasn’t been analyzed. Here, we report a first proteome analysis of poplar-type propolis, a challenging glue-type resinous sample for protein characterization. Raw propolis mixture was precipitated with cold acetone to obtain the protein fraction. Proteins were digested with trypsin, and generated peptides were analyzed on nano-ESI-qTOF SYNAPT G2-Si mass spectrometer (MS) by data-independent acquisition (DIA) and data-dependent acquisition (DDA). Identified peptides and inferred proteins suggest the presence of new bioactive molecules as components of propolis. The poplar-type propolis proteome is composed of a mixture of proteins from the Apis and Populus genera. This is the first-ever report of the proteome of any type of propolis.


Similar content being viewed by others
Data Availability
All raw data are available upon request to the corresponding author.
References
Šuran J, Cepanec I, Mašek T, Radić B, Radić S, Tlak Gajger I, Vlainić J (2021) Propolis extract and its bioactive compounds-from traditional to modern extraction technologies. Molecules 26:2930. https://doi.org/10.3390/molecules26102930
Scarselli R, Donadio E, Giuffrida MG, Fortunato D, Conti A, Balestreri E, Felicioli R, Pinzauti M, Sabatini AG, Felicioli A (2005) Towards royal jelly proteome. Proteomics 5:769–776. https://doi.org/10.1002/pmic.200401149.Erratum.In:Proteomics5:1454
Schönleben S, Sickmann A, Mueller MJ, Reinders J (2007) Proteome analysis of Apis mellifera royal jelly. Anal Bioanal Chem 389:1087–1093. https://doi.org/10.1007/s00216-007-1498-2
Li J, Wang T, Zhang Z, Pan Y (2007) Proteomic analysis of royal jelly from three strains of western honeybees (Apis mellifera). J Agric Food Chem 55:8411–8422. https://doi.org/10.1021/jf0717440
Buttstedt A, Moritz RF, Erler S (2014) Origin and function of the major royal jelly proteins of the honeybee (Apis mellifera) as members of the yellow gene family. Biol Rev Camb Philos Soc 89:255–269. https://doi.org/10.1111/brv.12052
Furusawa T, Rakwal R, Nam HW, Shibato J, Agrawal GK, Kim YS, Ogawa Y, Yoshida Y, Kouzuma Y, Masuo Y, Yonekura M (2008) Comprehensive royal jelly (RJ) proteomics using one- and two-dimensional proteomics platforms reveals novel RJ proteins and potential phospho/glycoproteins. J Proteome Res 7:3194–3229. https://doi.org/10.1021/pr800061j
Kaplan N, Linial M (2006) ProtoBee: hierarchical classification and annotation of the honey bee proteome. Genome Res 16:1431–1438. http://www.genome.org/cgi/doi/10.1101/gr.4916306
Bilikova K, Krakova TK, Yamaguchi K, Yamaguchi Y (2015) Major royal jelly proteins as markers of authenticity and quality of honey. Arh Hig Rada Toksikol 66:259–267. https://doi.org/10.1515/aiht-2015-66-2653
Bong J, Middleditch M, Loomes KM, Stephens JM (2021) Proteomic analysis of honey. Identification of unique peptide markers for authentication of NZ mānuka (Leptospermum scoparium) honey. Food Chem 350:128442. https://doi.org/10.1016/j.foodchem.2020.128442
Paget BW, Kleffmann T, Whiteman KE, Thomas MF, McMahon CD (2023) Quantitative comparison of manuka and clover honey proteomes with royal jelly. PLoS ONE 18:e0272898. https://doi.org/10.1371/journal.pone.0272898
Schmitzová J, Klaudiny J, Albert S, Schröder W, Schreckengost W, Hanes J, Júdová J, Simúth J (1998) A family of major royal jelly proteins of the honeybee Apis mellifera L. Cell Mol Life Sci 54:1020–1030. https://doi.org/10.1007/s000180050229
Botezan S, Baci GM, Bagameri L, Paşca C, Dezmirean DS (2023) Current status of the bioactive properties of royal jelly: a comprehensive review with a focus on its anticancer, anti-inflammatory, and antioxidant effects. Molecules 28:1510. https://doi.org/10.3390/molecules28031510
Kohno K, Okamoto I, Sano O, Arai N, Iwaki K, Ikeda M, Kurimoto M (2004) Royal jelly inhibits the production of proinflammatory cytokines by activated macrophages. Biosci Biotechnol Biochem 68:138–145. https://doi.org/10.1271/bbb.68.138
Minegaki N, Koshizuka T, Hatasa K, Kondo H, Kato H, Tannaka M, Takahashi K, Tsuji M, Inoue N (2022) The C-terminal penta-peptide repeats of major royal jelly protein 3 ameliorate the progression of inflammation in vivo and in vitro. Biol Pharm Bull 45:583–589. https://doi.org/10.1248/bpb.b21-00922
Lee K, Seo PJ (2022) Wound-induced systemic responses and their coordination by electrical signals. Front Plant Sci 13:880680. https://doi.org/10.3389/fpls.2022.880680
Pečenková T, Pejchar P, Moravec T, Drs M, Haluška S, Šantrůček J, Potocká A, Žárský V, Potocký M (2022) Immunity functions of Arabidopsis pathogenesis-related 1 are coupled but not confined to its C-terminus processing and trafficking. Mol Plant Pathol 23:664–678. https://doi.org/10.1111/mpp.13187
Major IT, Constabel CP (2008) Functional analysis of the Kunitz trypsin inhibitor family in poplar reveals biochemical diversity and multiplicity in defense against herbivores. Plant Physiol 146:888–903. https://doi.org/10.1104/pp.107.106229
Zhang Q, Xu J, Zhou X, Liu Z (2022) CAP superfamily proteins from venomous animals: who we are and what to do? Int J Biol Macromol 221:691–702. https://doi.org/10.1016/j.ijbiomac.2022.09.079
Kaur S, Samota MK, Choudhary M, Choudhary M, Pandey AK, Sharma A, Thakur J (2022) How do plants defend themselves against pathogens- Biochemical mechanisms and genetic interventions. Physiol Mol Biol Plants 28:485–504. https://doi.org/10.1007/s12298-022-01146-y
Wangorsch A, Scheurer S, Blanca M, Blanca-Lopez N, Somoza ML, Martín-Pedraza L (2022) Allergenic properties and molecular characteristics of PR-1 proteins. Front Allergy 3. https://doi.org/10.3389/falgy.2022.824717
Arnaiz A, Talavera-Mateo L, Gonzalez-Melendi P, Martinez M, Diaz I, Santamaria ME (2018) Arabidopsis Kunitz trypsin inhibitors in defense against spider mites. Front Plant Sci 9. https://doi.org/10.3389/fpls.2018.00986
Cid-Gallegos MS, Corzo-Ríos LJ, Jiménez-Martínez C, Sánchez-Chino XM (2022) Protease inhibitors from plants as therapeutic agents- a review. Plant Foods Hum Nutr 77:20–29. https://doi.org/10.1007/s11130-022-00949-4
Machado RJ, Monteiro NK, Migliolo L, Silva ON, Pinto MF, Oliveira AS, Franco OL, Kiyota S, Bemquerer MP, Uchoa AF, Morais AH, Santos EA (2013) Characterization and pharmacological properties of a novel multifunctional Kunitz inhibitor from Erythrina velutina seeds. PLoS ONE 8:e63571. https://doi.org/10.1371/journal.pone.0063571
Lichtenstein F, Iqbal A, de Lima Will SEA, Bosch RV, DeOcesano-Pereira C, Goldfeder MB, Chammas R, Trufen CEM, Morais KLP, de Souza JG, Natalino RJM, de Azevedo IJ, Nishiyama Junior MY, Oliveira U, Alves FIA, Araujo JM, Lobba ARM, Chudzinski-Tavassi AM (2020) Modulation of stress and immune response by Amblyomin-X results in tumor cell death in a horse melanoma model. Sci Rep 10:6388. https://doi.org/10.1038/s41598-020-63275-2
Funding
This study was performed as a part of the project named «Beehive as a natural resource for antibiotic alternatives» (Grant number 2018–74), financed by the private Sweden foundation Ekhagastiftelsen, founded by Gösta Videgård.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Raw material collection and preparation were performed by Jelena Šuran and Božo Radić, and material extraction and proteomic analysis was performed by Amela Hozić and Mario Cindrić. Bioinformatic analysis was performed by Dilza Trevisan-Silva. The first draft of the manuscript was written by Dilza Trevisan-Silva and Jelena Šuran, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Conflict of Interest
None declared.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Šuran, J., Radić, B., Trevisan-Silva, D. et al. First Proteome Analysis of Poplar-Type Propolis. Plant Foods Hum Nutr 79, 83–89 (2024). https://doi.org/10.1007/s11130-023-01127-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-023-01127-w