Abstract
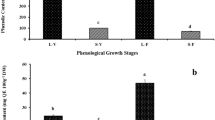
In order to comprehensively analyze the antioxidant substances in sour jujube, total phenolic content (TPC) and total flavonoids contents (TFC) in different organs, including stem, leaf, flower, fruit pulp, and seed were analyzed for their contents and antioxidant activities. The results showed that leaves possessed significantly higher TPC and TFC (20.4 and 20.5 mg/g, respectively) than the other organs and have the highest antioxidant activity, which were also higher than the wild blueberry (A well-known for its high TPC). Subsequently, the variations in the antioxidant content and antioxidant activity of leaves were analyzed during leaf development. TPC in leaves sampled in may and august were significantly higher than that in other months, while the highest one was found in may. The n-hexane, ethyl acetate, n-butanol, and water fractions obtained from the main methanol extract of sour jujube leaves were evaluated for TPC and TFC and their antioxidant activity and it was found that ethyl acetate fraction displayed the highest TPC and TFC (184.5 and 193.3 mg/g, respectively), as well as the best antioxidant activity. In addition, using LC-MS and HPLC, ethyl acetate fraction was analyzed from qualitative and quantitative aspects; 31-one phenolic compounds, including catechin (33.0 mg/g), epigallocatechin (15.3 mg/g), quercetin 3-O-glucoside (11.4 mg/g), naringenin (6.7 mg/g), esculetin (4.8 mg/g), and chlorogenic acid (4.6 mg/g) were identified. Catechin, esculetin, epigallocatechin, chlorogenic acid, quercetin 3-O-glucoside, and naringenin exhibited high antioxidant activity. These results provide a theoretical basis for further study and utilization of flavonoid and polyphenols in sour jujube.

Similar content being viewed by others
Data Availability
Not available.
Abbreviations
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- DW:
-
dry weight
- GAE:
-
gallic acid equivalents
- HPLC:
-
high performance liquid chromatography
- IC50 :
-
values correspond to the sample concentration achieving 50% of activity
- LC-MS:
-
liquid chromatography mass spectrometry
- RE:
-
rutin equivalent
- TFC:
-
total flavonoids content
- TPC:
-
total phenolic content
- SD:
-
standard deviation
References
Abbas M, Saeed F, Anjum FM, Afzaal M, Tufail T, Bashir MS, Ishtiaq A, Hussain S, Suleria HAR (2016) Natural polyphenols: an overview. Int J Food Prop 20(8):1689–1699. https://doi.org/10.1080/10942912.2016.1220393
Figueira JA, Porto-Figueira P, Pereira JAM, Camara JS (2021) Free low-molecular weight phenolics composition and bioactivity of Vaccinium padifolium sm fruits. Food Res Int 148:110580. https://doi.org/10.1016/j.foodres.2021.110580
Zou F, Li X, Yang R, Zhang R, Zhao X (2022) Effects and underlying mechanisms of food polyphenols in treating gouty arthritis: a review on nutritional intake and joint health. J Food Biochem 46(2):e14072. https://doi.org/10.1111/jfbc.14072
Ali S, Alam M, Khatoon F, Fatima U, Elasbali AM, Adnan M, Islam A, Hassan MI, Snoussi M, De Feo V (2022) Natural products can be used in therapeutic management of COVID-19: probable mechanistic insights. Biomed Pharmacother 147:112658. https://doi.org/10.1016/j.biopha.2022.112658
Tayab MA, Islam MN, Chowdhury KAA, Tasnim FM (2022) Targeting neuroinflammation by polyphenols: a promising therapeutic approach against inflammation-associated depression. Biomed Pharmacother 147:112668. https://doi.org/10.1016/j.biopha.2022.112668
Ungarala R, Munikumar M, Sinha SN, Kumar D, Sunder RS, Challa S (2022) Assessment of antioxidant, immunomodulatory activity of oxidised epigallocatechin-3-gallate (green tea polyphenol) and its action on the main protease of SARS-CoV-2-An in vitro and in silico spproach. Antioxid (Basel) 11(2). https://doi.org/10.3390/antiox11020294
Wang L, Song J, Liu A, Xiao B, Li S, Wen Z, Lu Y, Du G (2020) Research progress of the antiviral bioactivities of natural flavonoids. Nat Prod Bioprospect 10(5):271–283. https://doi.org/10.1007/s13659-020-00257-x
Zilli AMH, Zilli EM (2021) Review of evidence and perspectives of flavonoids on metabolic syndrome and neurodegenerative disease. Protein Pept Let 28(7):725–734. https://doi.org/10.2174/0929866528666210127152359
Wang L, Luo Z, Liu Z, Zhao J, Deng W, Wei H, Liu P, Liu M (2019) Genome size variation within species of chinese jujube (Ziziphus jujuba Mill.) and its wild ancestor sour jujube (Z. acidojujuba Cheng et Liu). Forests 10(5). https://doi.org/10.3390/f10050460
Li S, Deng B, Tian S, Guo M, Liu H, Zhao X (2021) Metabolic and transcriptomic analyses reveal different metabolite biosynthesis profiles between leaf buds and mature leaves in Ziziphus jujuba mill. Food Chem 347:129005. https://doi.org/10.1016/j.foodchem.2021.129005
Hua Y, Guo S, Xie H, Zhu Y, Yan H, Tao WW, Shang EX, Qian DW, Duan JA (2021) Ziziphus jujuba Mill. var. spinosa (Bunge) Hu ex H. F. Chou seed ameliorates insomnia in rats by regulating metabolomics and intestinal flora composition. Front Pharmacol 12:653767. https://doi.org/10.3389/fphar.2021.653767
Rajaei A, Salarbashi D, Asrari N, Fazly Bazzaz BS, Aboutorabzade SM, Shaddel R (2020) Antioxidant, antimicrobial, and cytotoxic activities of extracts from the seed and pulp of jujube (Ziziphus jujuba) grown in Iran. Food Sci Nutr 9(2):682–691. https://doi.org/10.1002/fsn3.2031
Zhang H, Jiang L, Ye S, Ye Y, Ren F (2010) Systematic Evaluation of antioxidant capacities of the ethanolic extract of different tissues of jujube (Ziziphus jujuba Mill.) from China. Food Chem Toxicol 48(6):1461–1465. https://doi.org/10.1016/j.fct.2010.03.011
Pastore S, Potapovich A, Kostyuk V, Mariani V, Lulli D, De Luca C, Korkina L (2009) Plant polyphenols effectively protect HaCaT cells from ultraviolet c-triggered necrosis and suppress inflammatory chemokine expression. Ann N Y Acad Sci 1171:305–313. https://doi.org/10.1111/j.1749-6632.2009.04684.x
Pan J, Tong X, Guo B (2016) Progress of effects of light on plant flavonoids. China J Chinese Mat Medica 41(21):3897–3903
Bernal-Gallardo JO, Molina-Torres J, Angoa-Pérez MV, Cárdenas-Valdovinos JG, García-Ruíz I, Ceja-Díaz JA, Mena-Violante HG (2021) Phenolic compound content and the antioxidant and antimicrobial activity of wild blueberries (Vaccinium stenophyllum Steud.) fruits extracts during ripening. Horticulturae 8(1). https://doi.org/10.3390/horticulturae8010015
Kaur SPM (2014) Study of Total phenolic and flavonoid content, antioxidant activity and antimicrobial properties of medicinal plants. J Microbiol 1. https://doi.org/10.15406/jmen.2014.01.00005
Khandaker L, Akond ASMGM, Ali MB, Oba S (2010) Biomass yield and accumulations of bioactive compounds in red amaranth (Amaranthus tricolor L.) grown under different colored shade polyethylene in spring season. Sci Hortic 123(3):289–294. https://doi.org/10.1016/j.scienta.2009.09.012
Bai Y, Xu Y, Wang B, Li S, Guo F, Hua H, Zhao Y, Yu Z (2017) Comparison of phenolic compounds, antioxidant and antidiabetic activities between selected edible beans and their different growth periods leaves. J Funct Foods 35:694–702. https://doi.org/10.1016/j.jff.2017.06.036
Hao JF, Li YY, Jia YS, Wang ZJ, Rong R, Bao J, Zhao MQ, Fu ZH, Ge GT (2021) Comparative analysis of major flavonoids among parts of lactuca indica during different growth periods. Molecules 26(24):13. https://doi.org/10.3390/molecules26247445
Schmidt S, Zietz M, Schreiner M, Rohn S, Kroh LW, Krumbein A (2010) Genotypic and climatic influences on the concentration and composition of flavonoids in kale (Brassica oleracea var. sabellica). Food Chem 119(4):1293–1299. https://doi.org/10.1016/j.foodchem.2009.09.004
Bergquist SAM, Gertsson UE, Knuthsen P, Olsson ME (2005) Flavonoids in baby spinach (Spinacia oleracea L.): changes during plant growth and storage. J Agric Food Chem 53(24):9459–9464. https://doi.org/10.1021/jf051430h
Bubols GB, Vianna DD, Medina-Remon A, von Poser G, Lamuela-Raventos RM, Eifler-Lima VL, Garcia SC (2013) The antioxidant activity of coumarins and flavonoids. Mini Rev Med Chem 13(3):318–334
Funding
This work was supported by Ministry of Science and Technology of the People’s Republic of China [Grant number 2021YFD1000404] and Department of Education of Hebei Province [Grant number 19227141D].
Author information
Authors and Affiliations
Contributions
Changwei Ao: Conceptualization, Supervision, Writing – review and editing; Zijuan Wang: Data curation, Writing - original draft; Xiaowen An, Bimal Chitrakar, Jiamin Li, Ye Yuan, Kexin Liu, Xinyu Nie, Yifan Zhang and Xin Zhao: Formal analysis; Zhihui Zhao, Mengjun Liu: Methodology, Software, Validation.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest.
Ethics Approval
This article does not contain any studies with human or animal subjects.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Z., An, X., Chitrakar, B. et al. Spatial and Temporal Distribution of Phenolic and Flavonoid Compounds in Sour Jujube (Ziziphus. Acidojujuba Cheng et Liu) and Their Antioxidant Activities. Plant Foods Hum Nutr 78, 46–51 (2023). https://doi.org/10.1007/s11130-022-01015-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11130-022-01015-9