Abstract
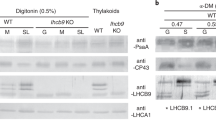
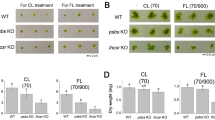
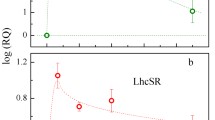
The chlorophyllide a oxygenase (CAO) plays a crucial role in the biosynthesis of chlorophyll b (Chl b). In the moss Physcomitrium patens (P. patens), two distinct gene copies, PpCAO1 and PpCAO2, are present. In this study, we investigate the differential expression of these CAOs following light exposure after a period of darkness (24 h) and demonstrate that the accumulation of Chl b is only abolished when both genes are knocked out. In the ppcao1cao2 mutant, most of the antenna proteins associated with both photosystems (PS) I and II are absent. Despite of the existence of LHCSR proteins and zeaxanthin, the mutant exhibits minimal non-photochemical quenching (NPQ) capacity. Nevertheless, the ppcao1cao2 mutant retains a certain level of pseudo-cyclic electron transport to provide photoprotection for PSI. These findings shed light on the dual dependency of Chl b synthesis on two CAOs and highlight the distinct effects of Chl b deprival on PSI and PSII core complexes in P. patens, a model species for bryophytes.






Similar content being viewed by others
Data availability
It is not applicable.
References
Ashton NW, Grimsley NH, Cove DJ (1979) Analysis of gametophytic development in the moss, Physcomitrella patens, using auxin and cytokinin resistant mutants. Planta 144:427–435. https://doi.org/10.1007/bf00380118
Bonente G, Ballottari M, Truong TB et al (2011) Analysis of LhcSR3, a protein essential for feedback de-excitation in the green alga Chlamydomonas reinhardtii. PLoS Biol 9:e1000577. https://doi.org/10.1371/journal.pbio.1000577
Bujaldon S, Kodama N, Rappaport F et al (2017) Functional accumulation of antenna proteins in chlorophyll b-Less Mutants of Chlamydomonas reinhardtii. Mol Plant 10:115–130. https://doi.org/10.1016/j.molp.2016.10.001
Casazza AP, Tarantino D, Soave C (2001) Preparation and functional characterization of thylakoids from Arabidopsis thaliana. Photosynth Res 68:175–180. https://doi.org/10.1023/a:1011818021875
Cove D (2005) The Moss Physcomitrella patens. Annu Rev Genet 39:339–358. https://doi.org/10.1146/annurev.genet.39.073003.110214
Dall’Osto L, Cazzaniga S, Havaux M et al (2010) Enhanced photoprotection by protein-bound vs free xanthophyll pools: a comparative analysis of chlorophyll b and xanthophyll biosynthesis mutants. Mol Plant 3:576–593. https://doi.org/10.1093/mp/ssp117
Derks A, Schaven K, Bruce D (2015) Diverse mechanisms for photoprotection in photosynthesis: dynamic regulation of photosystem II excitation in response to rapid environmental change. Biochem Biophys Acta 1847:468–485. https://doi.org/10.1016/j.bbabio.2015.02.008
Eggink LL, LoBrutto R, Brune DC et al (2004) Synthesis of chlorophyll b: localization of chlorophyllide a oxygenase and discovery of a stable radical in the catalytic subunit. BMC Plant Biol 4:5. https://doi.org/10.1186/1471-2229-4-5
Espineda CE, Linford AS, Devine D et al (1999) The AtCAO gene, encoding chlorophyll a oxygenase, is required for chlorophyll b synthesis in Arabidopsis thaliana. PNAS 96:10507–10511. https://doi.org/10.1073/pnas.96.18.10507
Friedland N, Negi S, Vinogradova-Shah T et al (2019) Fine-tuning the photosynthetic light harvesting apparatus for improved photosynthetic efficiency and biomass yield. Sci Rep 9:13028. https://doi.org/10.1038/s41598-019-49545-8
Gerotto C, Alboresi A, Giacometti GM et al (2012) Coexistence of plant and algal energy dissipation mechanisms in the moss Physcomitrella patens. New Phytol 196:763–773. https://doi.org/10.1111/j.1469-8137.2012.04345.x
Gerotto C, Alboresi A, Meneghesso A et al (2016) Flavodiiron proteins act as safety valve for electrons in Physcomitrella patens. PNAS 113:12322–12327. https://doi.org/10.1073/pnas.1606685113
Gerotto C, Trotta A, Bajwa AA et al (2019) Thylakoid protein phosphorylation dynamics in amoss mutant lacking serine/threonine protein kinase STN8. Plant Physiol 180:1582–1597. https://doi.org/10.1104/pp.19.00117
Giovagnetti V, Ruban AV (2018) The evolution of the photoprotective antenna proteins in oxygenic photosynthetic eukaryotes. Biochem Soc Trans 46:1263–1277. https://doi.org/10.1042/bst20170304
Green BR, Durnford DG (1996) The chlorophyll-carotenoid proteins of oxygenic photosynthesis. Annu Rev Plant Physiol Plant Mol Biol 47:685–714. https://doi.org/10.1146/annurev.arplant.47.1.685
Greene BA, Staehelin LA, Melis A (1988) Compensatory alterations in the photochemical apparatus of a photoregulatory, chlorophyll b-deficient mutant of Maize. Plant Physiol 87:365–370. https://doi.org/10.1104/pp.87.2.365
Havaux M, Dall’osto L, Bassi R (2007) Zeaxanthin has enhanced antioxidant capacity with respect to all other xanthophylls in Arabidopsis leaves and functions independent of binding to PSII antennae. Plant Physiol 145:1506–1520. https://doi.org/10.1104/pp.107.108480
Hoober JK, Eggink LL, Chen M (2007) Chlorophylls, ligands and assembly of light-harvesting complexes in chloroplasts. Photosynth Res 94:387–400. https://doi.org/10.1007/s11120-007-9181-1
Horn R, Grundmann G, Paulsen H (2007) Consecutive binding of chlorophylls a and b during the assembly in vitro of light-harvesting chlorophyll-a/b protein (LHCIIb). J Mol Biol 366:1045–1054. https://doi.org/10.1016/j.jmb.2006.11.069
Ilik P, Pavlovic A, Kouril R et al (2017) Alternative electron transport mediated by flavodiiron proteins is operational in organisms from cyanobacteria up to gymnosperms. New Phytol 214:967–972. https://doi.org/10.1111/nph.14536
Iwai M, Yokono M (2017) Light-harvesting antenna complexes in the moss Physcomitrella patens: implications for the evolutionary transition from green algae to land plants. Curr Opin Plant Biol 37:94–101. https://doi.org/10.1016/j.pbi.2017.04.002
Iwai M, Yokono M, Kono M et al (2015) Light-harvesting complex Lhcb9 confers a green alga-type photosystem I supercomplex to the moss Physcomitrella patens. Nature Plants 1:14008. https://doi.org/10.1038/nplants.2014.8
Järvi S, Suorsa M, Paakkarinen V et al (2011) Optimized native gel systems for separation of thylakoid protein complexes: novel super- and mega-complexes. Biochemical Journal 439:207–214. https://doi.org/10.1042/BJ20102155
Jung YJ, Lee HJ, Yu J et al (2021) Transcriptomic and physiological analysis of OsCAO1 knockout lines using the CRISPR/Cas9 system in rice. Plant Cell Rep 40:1013–1024. https://doi.org/10.1007/s00299-020-02607-y
Kim EH, Li XP, Razeghifard R et al (2009) The multiple roles of light-harvesting chlorophyll a/b-protein complexes define structure and optimize function of Arabidopsis chloroplasts: a study using two chlorophyll b-less mutants. Biochem Biophys Acta 1787:973–984. https://doi.org/10.1016/j.bbabio.2009.04.009
Koziol AG, Borza T, Ishida K et al (2007) Tracing the evolution of the light-harvesting antennae in chlorophyll a/b-containing organisms. Plant Physiol 143:1802–1816. https://doi.org/10.1104/pp.106.092536
Kunugi M, Takabayashi A, Tanaka A (2013) Evolutionary changes in chlorophyllide a oxygenase (CAO) structure contribute to the acquisition of a new light-harvesting complex in micromonas. J Biol Chem 288:19330–19341. https://doi.org/10.1074/jbc.M113.462663
Lee S, Kim JH, Yoo ES et al (2005) Differential regulation of chlorophyll a oxygenase genes in rice. Plant Mol Biol 57:805–818. https://doi.org/10.1007/s11103-005-2066-9
Lopez-Obando M, Hoffmann B, Géry C et al (2016) Simple and efficient targeting of multiple genes through CRISPR-Cas9 in Physcomitrella patens. G3-Genes Genom Genet 6:3647–3653. https://doi.org/10.1534/g3.116.033266
Malkin S, Armond PA, Mooney HA et al (1981) Photosystem II photosynthetic unit sizes from fluorescenceinduction in leaves. Plant Physiol 67:570–579. https://doi.org/10.1104/pp.67.3.570
Müller P, Li XP, Niyogi KK (2001) Non-photochemical quenching: a response to excess light energy. Plant Physiol 125:1558–1566. https://doi.org/10.1104/pp.125.4.1558
Mussgnug JH, Thomas-Hall S, Rupprecht J et al (2007) Engineering photosynthetic light capture: impacts on improved solar energy to biomass conversion. Plant Biotechnol J 5:802–814. https://doi.org/10.1111/j.1467-7652.2007.00285.x
Nagata N, Satoh S, Tanaka R et al (2004) Domain structures of chlorophyllide a oxygenase of green plants and Prochlorothrix hollandica in relation to catalytic functions. Planta 218:1019–1025. https://doi.org/10.1007/s00425-003-1181-6
Nick S, Meurer J, Soll J et al (2013) Nucleus-encoded light-harvesting chlorophyll a/b proteins are imported normally into chlorophyll b-free chloroplasts of Arabidopsis. Mol Plant 6:860–871. https://doi.org/10.1093/mp/sss113
Oster U, Tanaka R, Tanaka A et al (2000) Cloning and functional expression of the gene encoding the key enzyme for chlorophyll b biosynthesis (CAO) from Arabidopsis thaliana. Plant J 21:305–310. https://doi.org/10.1046/j.1365-313x.2000.00672.x
Perrine Z, Negi S, Sayre RT (2012) Optimization of photosynthetic light energy utilization by microalgae. Algal Res 1:134–142. https://doi.org/10.1016/j.algal.2012.07.002
Pinnola A, Dall’Osto L, Gerotto C et al (2013) Zeaxanthin binds to light-harvesting complex stress-related protein to enhance nonphotochemical quenching in Physcomitrella patens. Plant Cell 25:3519–3534. https://doi.org/10.1105/tpc.113.114538
Pinnola A, Cazzaniga S, Alboresi A et al (2015) Light-harvesting complex stress-related proteins catalyze excess energy dissipation in both photosystems of Physcomitrella patens. Plant Cell 27:3213–3227. https://doi.org/10.1105/tpc.15.00443
Pinnola A, Alboresi A, Nosek L et al (2018) A LHCB9-dependent photosystem I megacomplex induced under low light in Physcomitrella patens. Nat Plants 4:910–919. https://doi.org/10.1038/s41477-018-0270-2
Qin XC, Suga M, Kuang T et al (2015) Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex. Science 348:989–995. https://doi.org/10.1126/science.aab0214
Rensing SA, Lang D, Zimmer AD et al (2008) The Physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 319:64–69. https://doi.org/10.1126/science.1150646
Schaefer DG (2002) A new moss genetics: targeted mutagenesis in Physcomitrella patens. Annu Rev Plant Biol 53:477–501. https://doi.org/10.1146/annurev.arplant.53.100301.135202
Schaefer DG, Zrÿd JP (1997) Efficient gene targeting in the moss Physcomitrella patens. Plant J 11:1195–1206. https://doi.org/10.1046/j.1365-313x.1997.11061195.x
Schansker G, Srivastava A, Strasser RJ (2003) Characterization of the 820-nm transmission signal paralleling the chlorophyll a fluorescence rise (OJIP) in pea leaves. Funct Plant Biol 30:785–796. https://doi.org/10.1071/fp03032
Schansker G, Tóth SZ, Strasser RJ (2005) Methylviologen and dibromothymoquinone treatments of pea leaves reveal the role of photosystem I in the Chl a fluorescence rise OJIP. Biochim Biophys Acta 1706:250–261. https://doi.org/10.1016/j.bbabio.2004.11.006
Takabayashi A, Kurihara K, Kuwano M et al (2011) The oligomeric states of the photosystems and the light-harvesting complexes in the Chl b-less mutant. Plant Cell Physiol 52:2103–2114. https://doi.org/10.1093/pcp/pcr138
Tanaka R, Tanaka A (2005) Effects of chlorophyllide a oxygenase overexpression on light acclimation in Arabidopsis thaliana. Photosynth Res 85:327–340. https://doi.org/10.1007/s11120-005-6807-z
Tanaka R, Tanaka A (2011) Chlorophyll cycle regulates the construction and destruction of the light-harvesting complexes. Biochem Biophys Acta 1807:968–976. https://doi.org/10.1016/j.bbabio.2011.01.002
Tanaka A, Tanaka R (2019) The biochemistry, physiology, and evolution of the chlorophyll cycle—ScienceDirect. Adv Bot Res 90:183–212. https://doi.org/10.1016/bs.abr.2019.03.005
Tanaka A, Ito H, Tanaka R et al (1998) Chlorophyll a oxygenase (CAO) is involved in chlorophyll b formation from chlorophyll a. PNAS 95:12719–12723. https://doi.org/10.1073/pnas.95.21.12719
Thayer SS, Björkman O (1990) Leaf xanthophyll content and composition in sun and shade determined by HPLC. Photosynth Res 23:331–343. https://doi.org/10.1007/BF00034864
Way DA, Pearcy RW (2012) Sunflecks in trees and forests: from photosynthetic physiology to global change biology. Tree Physiol 32:1066–1081. https://doi.org/10.1093/treephys/tps064
Wu G, Ma L, Sayre RT et al (2020) Identification of the optimal light harvesting antenna size for high-light stress mitigation in plants. Front Plant Sci 11:505. https://doi.org/10.3389/fpls.2020.00505
Yamamoto H, Takahashi S, Badger MR et al (2016) Artificial remodelling of alternative electron flow by flavodiiron proteins in Arabidopsis. Nat Plants 2:16012. https://doi.org/10.1038/nplants.2016.12
Yan Q, Zhao L, Wang W et al (2021) Antenna arrangement and energy-transfer pathways of PSI-LHCI from the moss Physcomitrella patens. Cell Discov 7:10. https://doi.org/10.1038/s41421-021-00242-9
Yang C, Kosemund K, Cornet C et al (1999) Exchange of pigment-binding amino acids in light-harvesting chlorophyll a/b protein. Biochemistry 38:16205–16213. https://doi.org/10.1021/bi990738x
Funding
This work was supported by the Academician Workstation of Agricultural High-tech Industrial Area of the Yellow River Delta, National Center of Technology Innovation for Comprehensive Utilization of Saline-Alkali Land, Dongying, Shandong, China and Science & Technology Specific Projects in Agricultural High-tech Industrial Demonstration Area of the Yellow River Delta (Grant No: 2022SZX12) and the National Key R&D Program of China (Grant No: 2017YFA0503701).
Author information
Authors and Affiliations
Contributions
CL and CY planned and designed the research. LZ and CL carried out the experiments and took part in the data analysis. LZ and CL wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, L., Yang, C. & Liu, C. Revealing the significance of chlorophyll b in the moss Physcomitrium patens by knocking out two functional chlorophyllide a oxygenase. Photosynth Res 158, 171–180 (2023). https://doi.org/10.1007/s11120-023-01044-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-023-01044-8