Abstract
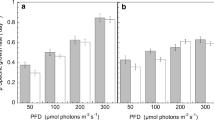
Microalgae cultivation utilizes the energy of sunlight to reduce carbon dioxide (CO2) for producing renewable energy feedstock. The commercial success of the biological fixation of carbon in a consistent manner depends upon the availability of a robust microalgae strain. In the present work, we report the identification of a novel marine Nannochloris sp. through multiparametric photosynthetic evaluation. Detailed photobiological analysis of this strain has revealed a smaller functional antenna, faster relaxation kinetics of non-photochemical quenching, and a high photosynthetic rate with increasing light and temperatures. Furthermore, laboratory scale growth assessment demonstrated a broad range halotolerance of 10–70 parts per thousand (PPT) and high-temperature tolerance up to 45 °C. Such traits led to the translation of biomass productivity potential from the laboratory scale (0.2–3.0 L) to the outdoor 50,000 L raceway pond scale (500-m2) without any pond crashes. The current investigation revealed outdoor single-day peak areal biomass productivity of 43 g m−2 d−1 in summer with an annual (March 2019–February 2020) average productivity of 20 g m−2 d−1 in seawater. From a sustainability perspective, this is the first report of successful round-the-year (> 347 days) multi-season (summer, monsoon, and winter) outdoor cultivation of Nannochloris sp. in broad seawater salinity (1–57 PPT), wide temperature ranges (15–40 °C), and in fluctuating light conditions. Concurrently, outdoor cultivation of this strain demonstrated conducive fatty acid distribution, including increased unsaturated fatty acids in winter. This inherent characteristic might play a role in protecting photosynthesis machinery at low temperatures and in high light stress. Altogether, our marine Nannochloris sp. showed tremendous potential for commercial scale cultivation to produce biofuels, food ingredients, and a sustainable source for vegetarian protein.












Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article in the form of figures and tables (and its supplementary information files). The data that support the findings of this study are available from Reliance Industries Ltd. India, but restrictions apply to the availability of these data, and so are not publicly available. Data are however available from Reliance Industries Ltd. India. upon reasonable request and with nondisclosure agreement.
Abbreviations
- ABS/RC:
-
Mo (1/VJ) (1/ ΦPo), absorption flux per reaction centre
- AFDW:
-
Ash-free dry weight
- α:
-
The maximum light utilization coefficient or quantum efficiency of photosynthesis
- Chl:
-
Chlorophyll
- CHNS:
-
Carbon Hydrogen Nitrogen Sulphur content
- DIo/ RC:
-
(ABS/RC)-(TRo/RC), the ratio of the total dissipation of untrapped excitation energy from all RCs with respect to the number of active RCs.
- ePBR:
-
Environmental photobioreactor
- Ek :
-
Light saturation index
- FAME:
-
Fatty acid methyl esters
- F o :
-
F50µS, fluorescence intensity at 50 µs, when all reaction centres are open
- F m :
-
Maximal fluorescence intensity (100–200 ms) measured at dark-adapted state when photosystem II reaction centres are closed
- F v :
-
(FM-Fo), Photosystem II variable fluorescence
- F t :
-
Fluorescence intensity at given time, t
- F v/F m :
-
[1- (FO/ Fm))], maximal quantum yield of photosystem II photochemistry
- Ic :
-
Light compensation point
- Mo :
-
4 (F300µS –Fo)/(Fm-Fo) represents initial slope of fluorescence kinetics
- ML:
-
Maximum likelihood
- NPQ:
-
Non photochemical quenching
- OD750 :
-
Optical density measured at 750 nm wavelength
- PCR:
-
Polymerase chain reaction
- PIabs :
-
1- (Fo/ Fm) (Mo /Vj)−1 x (Fm- Fo) Fo−1 X (1- Vj) Vj−1, Performance index (potential) for energy conservation from photons absorbed by PSII to the reduction of intersystem electron acceptors
- PI:
-
Photosynthesis Irradiance
- Pmax :
-
Maximum photosynthetic rate
- PSII:
-
Photosystem II
- PSE:
-
Photosynthetic efficiency measured in terms of ‘%’
- PPT:
-
Parts per thousand
- τ:
-
Tau, NPQ relaxation time scales
- TRo/RC:
-
The maximal energy trapping flux per RC
- θ:
-
Convexity factor (degree of photoinhibition)
- qE:
-
Coefficient for energy related quenching
- qZ:
-
Coefficient for zeaxanthin related quenching
- UPA:
-
Urea phosphoric acid
- VJ :
-
(F2Ms-Fo) / (Fm- Fo)
- Vt :
-
(FT- Fo) / (Fm- Fo)
References
Allakhverdiev SI, Los DA, Murata N (2009) Regulatory roles in photosynthesis of unsaturated fatty acids in membrane lipids. In: Wada H, Murata N (eds) Lipids in photosynthesis. advances in photosynthesis and respiration. Springer, Dordrecht
Amini Khoeyi Z, Seyfabadi J, Ramezanpour Z (2012) Effect of light intensity and photoperiod on biomass and fatty acid composition of the microalgae, Chlorella vulgaris. Aquacult Int 20:41–49. https://doi.org/10.1007/s10499-011-9440-1
Armbruster U, Carrillo LR, Venema K, Pavlovic L, Schmidtmann E, Kornfeld A, Jahns P, Berry JA, Kramer DM, Jonikas MC (2014) Ion antiport accelerates photosynthetic acclimation in fluctuating light environments. Nat Commun. https://doi.org/10.1038/NCOMMS6439
Arun J, Gopinath KP, Sivaramakrishnan R, SundarRajan PS, Malolan R, Pugazhendhi A (2021) Technical insights into the production of green fuel from CO2 sequestered algal biomass: a conceptual review on green energy. Sci Total Environ 755(2):142636. https://doi.org/10.1016/j.scitotenv.2020.142636
Banerjee A, Paul K, Varshney A, Nandru R, Badhwar R, Sapre A and Dasgupta S (2021) Chapter 8 Soilless indoor smart agriculture as an emerging enabler technology for food and nutrition security amidst climate change In Plant Nutrition and Food Security in the Era of Climate Change. Elsevier, Academic Press, eBook ISBN 9780128230930. https://doi.org/10.1016/B978-0-12-822916-3.00004-4
Banks JM (2017) Continuous excitation chlorophyll fluorescence parameters: a review for practitioners. Tree Physiol 37(8):1128–1136. https://doi.org/10.1093/treephys/tpx059
Barten R, Djohan Y, Evers W, Wijffels R, Barbosa M (2021) Towards industrial production of microalgae without temperature control: The effect of diel temperature fluctuations on microalgal physiology. J Biotechnol 336:56–63. https://doi.org/10.1016/j.jbiotec.2021.06.017
Bennett DIG, Amarnath K, Park S, Steen CJ, Morris JM, Fleming GR (2019) Models and mechanisms of the rapidly reversible regulation of photosynthetic light harvesting. Open Biol 9(4):190043. https://doi.org/10.1098/RSOB.190043
Betterle N, Ballottari M, Zorzan S, de Bianchi S, Cazzaniga S, Dall’Osto L, Morosinotto T, Bassi R (2009) Light-induced dissociation of an antenna hetero-oligomer is needed for non-photochemical quenching induction. J Biol Chem 284(22):15255–15266. https://doi.org/10.1074/jbc.M808625200
Borowitzka MA, Vonshak A (2017) Scaling up microalgal cultures to commercial scale. Eur J Phycol 52(4):407–418. https://doi.org/10.1080/09670262.2017.1365177
Bounnit T, Saadaoui I, Rasheed R, Schipper K, Al Muraikhi M, Al Jabri H (2020) Sustainable production of nannochloris atomus biomass towards biodiesel production. Sustainability. https://doi.org/10.3390/su12052008
Bujaldon S, Kodama N, Rappaport F, Subramanyam R, de Vitry C, Takahashi Y, Wollman FA (2017) Functional accumulation of antenna proteins in chlorophyll b-less mutants of Chlamydomonas reinhardtii. Mol Plant 10(1):115–130. https://doi.org/10.1016/j.molp.2016.10.001
Butcher RW (1952) Contributions to our knowledge of the smaller Marine algae. J Mar Biol Assoc UK 31(1):175–191. https://doi.org/10.1017/S0025315400003751
Cazzaniga S, Dall’Osto L, Szaub J, Scibilia L, Ballottari M, Purton S, Bassi R (2014) Domestication of the green alga Chlorella sorokiniana: reduction of antenna size improves light-use efficiency in a photobioreactor. Biotechnol Biofuels 7(1):1–13. https://doi.org/10.1186/s13068-014-0157-z
Chaisutyakorn P, Praiboon KC (2018) The effect of temperature on growth and lipid and fatty acid composition on marine microalgae used for biodiesel production. J Appl Phycol 30(1):37–45. https://doi.org/10.1007/s10811-017-1186-3
Chandrashekharaiah PS, Paul V, Kodgire S, Chawada H, Thorat R, Kushwaha S, Sanyal D, Dasgupta S (2021) Photoautotrophic cultivation of Chlamydomonas reinhardtii in open ponds of greenhouse. Arch Microbiol 203:1439–1450. https://doi.org/10.1007/s00203-020-02124-2
Choudhary P, Assemany PP, Naaz F, Bhattacharya A, JdeS C, EdeAdoC C, Calijuri ML, Pant KK, Malik A (2020) A review of biochemical and thermochemical energy conversion routes of wastewater grown algal biomass. Sci Total Environ 726:137961. https://doi.org/10.1016/j.scitotenv.2020.137961
Choudhary P, Goripati VS, Khade M, Savant S, Musale A, Kumar GRK, Chelliah MS, Dasgupta S (2021) Empowering blue economy: From underrated ecosystem to sustainable industry. J Environ Manage 291(312):112697
Concas A, Lutzu GA, Locci AM, Cao G (2013) Nannochloris eucaryotum growth: kinetic analysis and use of 100% CO2. Adv Environ Res 2(1):19–33. https://doi.org/10.12989/aer.2013.2.1.019
Dahmen-Ben Moussa I, Chtourou H, Hassairi I, Sayadi S, Dhouib A (2019) The effect of switching environmental conditions on content and structure of lipid produced by a wild strain Picochlorum sp. Renew Energy 134:406–415. https://doi.org/10.1016/J.RENENE.2018.11.050
Dall’Osto L, Cazzaniga S, Bressan M, Paleeèk D, Židek K, Niyogi KK, Fleming GR, Zigmantas D, Bassi R (2017) Two mechanisms for dissipation of excess light in monomeric and trimeric light-harvesting complexes. Nature Plants 3(5):1–9. https://doi.org/10.1038/nplants.2017.33
De Vree JH, Bosma R, Janssen M, Barbosa MJ, Wijffels RH (2015) Comparison of four outdoor pilot-scale photobioreactors. Biotechnol Biofuels 8(1):1–12. https://doi.org/10.1186/s13068-015-0400-2
Dogaris I, Brown TR, Loya B, Philippidis G (2016) Cultivation study of the marine microalga Picochlorum oculatum and outdoor deployment in a novel bioreactor for high-density production of algal cell mass. Biomass Bioenerg 89:11–23. https://doi.org/10.1016/j.biombioe.2016.02.018
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochimica Et Biophysica Acta (BBA). 990(1):87–92. https://doi.org/10.1016/S0304-4165(89)80016-9
Gouveia L, Oliveira AC, Congestri R, Bruno L, Soares AT, Menezes RS, Filho NRA, Tzovenis I (2017) 10- Biodiesel from microalgae. In: Gonzalez-Fernandez C, Muñoz R (eds) Woodhead publishing series in energy microalgae-based biofuels and bioproducts. Woodhead Publishing, Cambridge, pp 235–258
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chanley MH (eds) Culture of marine invertebrate animals. Springer, Boston, MA
Haddad R, Alemzadeh E, Ahmadi AR, Hosseini R, Moezzi M (2014) Identification of Chlorophyceae based on 18S rDNA sequences from Persian Gulf. Iranian Journal of Microbiology 6(6):437–442
Ippoliti D, González A, Martín I, Sevilla JMF, Pistocchi R, Acién FG (2016) Outdoor production of Tisochrysis lutea in pilot-scale tubular photobioreactors. J Appl Phycol 28(6):3159–3166. https://doi.org/10.1007/s10811-016-0856-x
Jazzar S, Quesada-Medina J, Olivares-Carrillo P, Marzouki MN, Acién-Fernández FG, Fernández-Sevilla JM, Molina-Grima E, Smaali I (2015) A whole biodiesel conversion process combining isolation, cultivation and in situ supercritical methanol transesterification of native microalgae. Biores Technol 190:281–288. https://doi.org/10.1016/j.biortech.2015.04.097
Jukes HT, Cantor RC (1969) Evolution of protein molecules. In: Munro HN (ed) Mammalian protein metabolism. Academic Press, New York, pp 21–132
Karuppasamy S, Musale AS, Soni B, Bhadra B, Gujarathi N, Sundaram M, Sapre A, Dasgupta S, Kumar C (2018) Integrated grazer management mediated by chemicals for sustainable cultivation of algae in open ponds. Algal Res 35:439–448. https://doi.org/10.1016/j.algal.2018.09.017
Khaw YS, Khong NMH, Shaharuddin NA, Yusoff FM (2020) A simple 18S rDNA approach for the identification of cultured eukaryotic microalgae with an emphasis on primers. J Microbiol Methods 172:105890. https://doi.org/10.1016/j.mimet.2020.105890
Krause G, Weis E (1991) Chlorophyll fluorescence and photosynthesis: The Basics. Annu Rev Plant Physiol Plant Mol Biol 42:313–349. https://doi.org/10.1002/9780470122563.ch3
Krishnan A, Likhogrud M, Cano M et al (2021) Picochlorum celeri as a model system for robust outdoor algal growth in seawater. Sci Rep 11:11649. https://doi.org/10.1038/s41598-021-91106-5
Kromdijk J, Głowacka K, Leonelli L, Gabilly ST, Iwai M, Niyogi KK, Long SP (2016) Improving photosynthesis and crop productivity by accelerating recovery from photoprotection. Science 354(6314):857–861. https://doi.org/10.1126/science.aai8878
Kula M, Kalaji HM, Skoczowski A (2017) Culture density influence on the photosynthetic efficiency of microalgae growing under different spectral compositions of light. J Photochem Photobiol, B 167:290–298. https://doi.org/10.1016/j.jphotobiol.2017.01.013
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Kumar V, Sharma N, Jaiswal KK, Vlaskin MS, Nanda M, Tripathi MK, Kumar S (2021) Microalgae with a truncated light-harvesting antenna to maximize photosynthetic efficiency and biomass productivity: Recent advances and current challenges. Process Biochem 104:83–91. https://doi.org/10.1016/j.procbio.2021.03.006
Laurens LML, Markham J, Templeton DW, Christensen ED, Van Wychen S, Vadelius EW, Chen-Glasser M, Dong T, Davis R, Pienkos PT (2017) Development of algae biorefinery concepts for biofuels and bioproducts; a perspective on process-compatible products and their impact on cost-reduction. Energy Environ Sci 10(8):1716–1738. https://doi.org/10.1039/c7ee01306j
Leong YK, Chew KW, Chen W-H, Chang J-S, Show PL (2021) Reuniting the biogeochemistry of algae for a low-carbon circular bioeconomy. Trends Plant Sci 26(7):729–740. https://doi.org/10.1016/j.tplants.2020.12.010
Lobo FDA, de Barros MP, Dalmagro HJ, Dalmolin ÂC, Pereira WE, de Souza ÉC, Vourlitis GL, Rodríguez Ortíz CE (2013) Fitting net photosynthetic light-response curves with Microsoft Excel - a critical look at the models. Photosynthetica 51(3):445–456. https://doi.org/10.1007/s11099-013-0045-y
López-Rosales AR, Ancona-Canché K, Chavarria-Hernandez JC, Barahona-Pérez F, Toledano-Thompson T, Garduño-Solórzano G, López-Adrian S, Canto-Canché B, Polanco-Lugo E, Valdez-Ojeda R (2019) Fatty acids, hydrocarbons and terpenes of nannochloropsis and nannochloris isolates with potential for biofuel production. Energies. https://doi.org/10.3390/en12010130
Maddikeri GL, Veluswamy GK, Khopkar AR (2019) A system for improving fluid circulation in a raceway pond. (Patent No.16/085, 191), WIPO (PCT)- WO2017175076A1. https://patents.google.com/patent/WO2017175076A1/en
Malkin S, Armond PA, Mooney HA, Fork DC (1981) Photosystem II photosynthetic unit sizes from fluorescence induction in leaves. Plant Physiol 67(3):570–579. https://doi.org/10.1104/pp.67.3.570
Mandan C, Madhesan B, Bhujade R, Sapre A (2020) A process for producing crude bio-oil (Patent No. US 10,800,974 B2), WIPO (PCT)- WO2018116036A1. https://patents.google.com/patent/WO2018116036A1/en
Manjre S, Paul K, Patil S, Pai P, Banerjee A, Sarkar P, Teredesai A, Shukla MR, Dasgupta S (2022) Evaluating the effect of seasonal conditions on metabolism and photosynthetic performance of Picochlorum sp and its influence on biomass productivity. Bioresour Technol Rep 18:101029. https://doi.org/10.1016/j.biteb.2022.101029
Mathur S, Allakhverdiev SI, Jajoo A (2011) Analysis of high temperature stress on the dynamics of antenna size and reducing side heterogeneity of Photosystem II in wheat leaves (Triticum aestivum). Biochem Biophys Acta 1:22–29. https://doi.org/10.1016/j.bbabio.2010.09.001
Merlo S, Gabarrell Durany X, Pedroso Tonon A, Rossi S (2021) Marine microalgae contribution to sustainable development. Water 13:1373. https://doi.org/10.3390/w13101373
Mohan N, Mahulkar AV, Veluswamy GK, Khopkar AR, Maddikeri GL, Kirdat NN, Banerjee A, Yelchuri R (2020) Algal cultivation system and a process for biomass production. Patent No. US 2020/0385662 A1, U.S. Patent Application No. 16/607. https://patents.google.com/patent/US20200385662A1/en
Müller P, Li XP, Niyogi KK (2001) Update on photosynthesis non-photochemical quenching. a response to excess light energy. Plant Physiol 125(4):1558–1566
Onyeaka H, Miri T, Obileke K-C, Hart A, Anumudu C, Zainab T, Al-Sharify ZT (2021) Minimizing carbon footprint via microalgae as a biological capture. Carbon Capture Sci Technol 1:100007. https://doi.org/10.1016/j.ccst.2021.100007
Patil S, Pandit R, Lali A (2017) Photosynthetic acclimation of Chlorella saccharophila to heat stress. Phycol Res 65(2):160–165. https://doi.org/10.1111/pre.12171
Patil S, Prakash G, Lali AM (2020) Reduced chlorophyll antenna mutants of Chlorella saccharophila for higher photosynthetic efficiency and biomass productivity under high light intensities. J Appl Phycol 32(3):1559–1567. https://doi.org/10.1007/s10811-020-02081-9
Rajvanshi M, Sayre R (2020) Recent advances in algal biomass production. In: Basso TP, Basso TO, Basso LC (eds) Biotechnological applications of biomass. IntechOpen, London
Rajvanshi M, Gautam K, Manjre S, Krishna Kumar GR, Kumar C, Govindachary S, Dasgupta S (2019) Stoichiometrically balanced nutrient management using a newly designed nutrient medium for large scale cultivation of Cyanobacterium aponinum. J Appl Phycol 31(5):2779–2789. https://doi.org/10.1007/s10811-019-01851-4
Ras M, Steyer JP, Bernard O (2013) Temperature effect on microalgae: a crucial factor for outdoor production. Rev Environ Sci Biotechnol 12(2):153–164. https://doi.org/10.1007/s11157-013-9310-6
Rasheed R, Thaher M, Younes N, Bounnit T, Schipper K, Nasrallah GK, Al Jabri H, Gifuni I, Goncalves O, Pruvost J (2022) Solar cultivation of microalgae in a desert environment for the development of techno-functional feed ingredients for aquaculture in Qatar. Sci Total Environ 835:155538. https://doi.org/10.1016/j.scitotenv.2022.155538
Ruangsomboon S, Dimak J, Jongput B, Wiwatanaratanabutr I, Kanyawongha P (2020) Outdoor open pond batch production of green microalga Botryococcus braunii for high hydrocarbon production: enhanced production with salinity. Sci Rep 10(1):1–12. https://doi.org/10.1038/s41598-020-59645-5
Saadaoui I, Al Ghazal G, Bounnit T, Al Khulaifi F, Al Jabri H, Potts M (2016) Evidence of thermo and halotolerant Nannochloris isolate suitable for biodiesel production in Qatar Culture Collection of Cyanobacteria and Microalgae. Algal Res 14:39–47. https://doi.org/10.1016/j.algal.2015.12.019
Schoepp NG, Stewart RL, Sun V, Quigley AJ, Mendola D, Mayfield SP, Burkart MD (2014) System and method for research-scale outdoor production of microalgae and cyanobacteria. Biores Technol 166:273–281. https://doi.org/10.1016/j.biortech.2014.05.046
Schuurmans MR, Schuurmans MJ, Bekker M, Kromkamp JC, Matthijs HCP, Hellingwerf KJ (2014) The redox potential of the plastoquinone pool of the cyanobacterium synechocystis species strain PCC 6803 is under strict homeostatic control. Plant Physiol 165(1):463–475. https://doi.org/10.1104/pp.114.237313
Stanier RY, Kunisawa R, Mandel M, Cohen-Bazire G (1971) Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol Rev 35(2):171–205. https://doi.org/10.1128/mmbr.35.2.171-205.1971
Subhash VG, Chugh N, Iyer S, Waghmare A, Musale AS, Nandru R, Dixit RB, Gaikwad MS, Menon D, Thorat R, Kumar GRK, Nagle V, Sagaram US, Dasgupta S (2020) Application of in vitro protein solubility for selection of microalgae biomass as protein ingredient in animal and aquafeed. J Appl Phycol 32(6):3955–3970. https://doi.org/10.1007/s10811-020-02235-9
Sukenik A, Bennett J, Falkowski P (1987) Light-saturated photosynthesis - limitation by electron transport or carbon fixation? Biochimica Et Biophysica Acta (BBA). 891(3):205–215. https://doi.org/10.1016/0005-2728(87)90216-7
Szalonek M, Sierpien B, Rymaszewski W, Gieczewska K, Garstka M, Lichocka M, Sass L, Paul K, Vass I, Vankova R, Dobrev P, Szczesny P, Marczewski W, Krusiewicz D, Strzelczyk-Zyta D, Hennig J, Konopka-Postupolska D (2015) Potato annexin STANN1 promotes drought tolerance and mitigates light stress in transgenic Solanum tuberosum L. plants. PLoS ONE 10(7):1–38. https://doi.org/10.1371/journal.pone.0132683
Vieira de Mendonça H, Assemany P, Abreu M, Couto E, Maciel AM, Duarte RL, Barbosa dos Santos MG, Reis A (2021) Microalgae in a global world: New solutions for old problems? Renew Energy 165:842–862. https://doi.org/10.1016/j.renene.2020.11.014
Wei L, Huang X, Huang Z (2015) Temperature effects on lipid properties of microalgae Tetraselmis subcordiformis and Nannochloropsis oculata as biofuel resources. Chin J Oceanol Limnol 33(1):99–106. https://doi.org/10.1007/s00343-015-3346-0
Weissman JC, Likhogrud M, Thomas DC, Fang W, Karns DAJ, Chung JW, Nielsen R, Posewitz MC (2018) High-light selection produces a fast-growing Picochlorum celeri. Algal Res 36:17–28. https://doi.org/10.1016/J.ALGAL.2018.09.024
Wijffels RH, Barbosa MJ (2010) An outlook on microalgal biofuels. Science 329(5993):796–799. https://doi.org/10.1126/science.1189003
Wu H, Abasova L, Cheregi O, Deák Z, Gao K, Vass I (2011) D1 protein turnover is involved in protection of Photosystem II against UV-B induced damage in the cyanobacterium Arthrospira (Spirulina) platensis. J Photochem Photobiol B 104(1–2):320–325. https://doi.org/10.1016/j.jphotobiol.2011.01.004
Yadav N, Singh DP (2021) Photosynthetic efficiency and compositional alterations in microalgae Chlorella vulgaris in response to changes in the pH condition. Vegetos 34:119–126. https://doi.org/10.1007/s42535-021-00186-1
Yusuf MA, Kumar D, Rajwanshi R, Strasser RJ, Tsimilli-Michael M, Govindjee SNB (2010) Overexpression of γ-tocopherol methyl transferase gene in transgenic Brassica juncea plants alleviates abiotic stress: physiological and chlorophyll a fluorescence measurements. Biochem Biophys Acta 1797(8):1428–1438. https://doi.org/10.1016/j.bbabio.2010.02.002
Zaks J, Amarnath K, Kramer DM, Niyogi KK, Fleming GR (2012) A kinetic model of rapidly reversible nonphotochemical quenching. Proc Natl Acad Sci USA 109(39):15757–15762. https://doi.org/10.1073/pnas.1211017109
Zarmi Y, Gordon JM, Mahulkar A, Khopkar AR, Patil SD, Banerjee A, Reddy BG, Griffin TP, Sapre A (2020) Enhanced algal photosynthetic photon efficiency by pulsed light. iScience 23(5):101115. https://doi.org/10.1016/j.isci.2020.101115
Zavřel T, Szabó M, Tamburic B, Evenhuis C, Kuzhiumparambil U, Literáková P, Larkum AWD, Raven JA, Červený J, Ralph PJ (2018) Effect of carbon limitation on photosynthetic electron transport in Nannochloropsis oculata. J Photochem Photobiol B 181:31–43. https://doi.org/10.1016/j.jphotobiol.2018.02.020
Živčák M, Brestič M, Olšovská K, Slámka P (2008) Performance index as a sensitive indicator of water stress in Triticum aestivum L. Plant Soil Environ 54:133–139. https://doi.org/10.17221/392-pse
Acknowledgements
The authors are thankful to Reliance Industries Limited for the financial support. Authors are extremely thankfully to Dr. Tomal Dattaroy (RIL) for editing of the manuscript and Dr. Bhaskar Bhadra (RIL) for helping with strain molecular identification studies. We would like to express thanks to analytical R&D team, Gagva, RIL for performing raceway pond microalgae sample AFDW estimations.
Author information
Authors and Affiliations
Contributions
KP: performed the photobiology experiments, prepared figures and/or tables, analyzed data; and wrote, compiled, edited and revised drafts of the paper. MG: performed strain growth characterization and morphological experiments in PBR, prepared figures and/or tables and wrote on experimental conditions as well as strain morphology part. PC: analyzed the biochemical data, prepared figures, wrote parts of introduction, edited and revised drafts of the paper. MN: performed outdoor cultivation experiments, analyzed and wrote on the environmental and PSE data, reviewed and revised drafts of the paper. PP and SDP: performed the photobiology experiments, analyzed the data, prepared figures and/or Tables and wrote the photobiology parts of paper. YP: performed and wrote on molecular identification experiments. AC: deigned the primers and conducted the molecular characterization experiments. VN and MC: conceived and coordinated the experiments, reviewed drafts of the paper. AB: conceptualized the study and the project, conceived and designed the experiments, analyzed the data, wrote, compiled the manuscript, reviewed drafts of the paper and corrected the manuscript. AS and SD: conceived the project, revised the article, resources arranged and supported internal funding.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. The authors also declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Paul, K., Gaikwad, M., Choudhary, P. et al. Year-round sustainable biomass production potential of Nannochloris sp. in outdoor raceway pond enabled through strategic photobiological screening. Photosynth Res 154, 303–328 (2022). https://doi.org/10.1007/s11120-022-00984-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-022-00984-x