Abstract
The heliobacteria, a family of anoxygenic phototrophs, possess the simplest known photosynthetic apparatus. Although they are photoheterotrophs in the light, the heliobacteria can also grow chemotrophically via pyruvate metabolism in the dark. In the heliobacteria, the cytochrome bc complex is responsible for oxidizing menaquinol and reducing cytochrome c553 in the electron flow cycle used for phototrophy. However, there is no known electron acceptor for the mobile cytochrome c553 other than the photochemical reaction center. We have, therefore, hypothesized that the cytochrome bc complex is necessary for phototrophy, but unnecessary for chemotrophic growth in the dark. We used a two-step method for CRISPR-based genome editing in Heliobacterium modesticaldum to delete the genes encoding the four major subunits of the cytochrome bc complex. Genotypic analysis verified the deletion of the petCBDA gene cluster encoding the catalytic components of the complex. Spectroscopic studies revealed that re-reduction of cytochrome c553 after flash-induced photo-oxidation was over 100 times slower in the ∆petCBDA mutant compared to the wild-type. Steady-state levels of oxidized P800 (the primary donor of the photochemical reaction center) were much higher in the ∆petCBDA mutant at every light level, consistent with a limitation in electron flow to the reaction center. The ∆petCBDA mutant was unable to grow phototrophically on acetate plus CO2 but could grow chemotrophically on pyruvate as a carbon source similar to the wild-type strain in the dark. The mutants could be complemented by reintroduction of the petCBDA gene cluster on a plasmid expressed from the clostridial eno promoter.







Similar content being viewed by others
References
Baker PL, Orf GS, Kevershan K, Pyne ME, Bicer T, Redding KE (2019a) Using the endogenous CRISPR-Cas system of Heliobacterium modesticaldum to delete the photochemical reaction center core subunit gene. Appl Environ Microbiol 85(23):e01644-e1719. https://doi.org/10.1128/AEM.01644-19
Baker PL, Orf GS, Khan Z, Espinoza L, Leung S, Kevershan K, Redding KE (2019b) A molecular biology tool kit for the phototrophic firmicute Heliobacterium modesticaldum. Appl Environ Microbiol 85(19):1–12. https://doi.org/10.1128/AEM.01287-19
Baymann F, Nitschke W (2010) Heliobacterial Rieske/cytb complex. Photosynth Res 104(2):177–187. https://doi.org/10.1007/s11120-009-9524-1
Crofts AR, Holland JT, Victoria D, Kolling DRJ, Dikanov SA, Gilbreth R, Lhee S, Kuras R, Kuras MG (2008) The Q-cycle reviewed: How well does a monomeric mechanism of the bc1 complex account for the function of a dimeric complex? Biochim Biophys Acta—Bioenerg 1777(7–8):1001–1019. https://doi.org/10.1016/j.bbabio.2008.04.037
Ducluzeau AL, Chenu E, Capowiez L, Baymann F (2008) The Rieske/cytochrome b complex of heliobacteria. Biochim Et Biophys Acta—Bioenerg 1777(9):1140–1146. https://doi.org/10.1016/j.bbabio.2008.04.026
Ferlez B, Cowgill J, Dong W, Gisriel C, Lin S, Flores M, Walters K, Cetnar D, Redding KE, Golbeck JH (2016) Thermodynamics of the electron acceptors in Heliobacterium modesticaldum: an exemplar of an early homodimeric type I photosynthetic reaction center. Biochemistry 55(16):2358–2370. https://doi.org/10.1021/acs.biochem.5b01320
Gisriel C, Sarrou I, Ferlez B, Golbeck JH, Redding KE, Fromme R (2017) Structure of a symmetric photosynthetic reaction center-photosystem. Science 357(6355):1021–1025. https://doi.org/10.1126/science.aan5611
Kashey TS, Luu DD, Cowgill JC, Baker PL, Redding KE (2018) Light-driven quinone reduction in heliobacterial membranes. Photosynth Res 138(1):1–9. https://doi.org/10.1007/s11120-018-0496-x
Kimble LK, Madigan MT (1992) Nitrogen fixation and nitrogen metabolism in heliobacteria. Arch Microbiol 158:155–161. https://doi.org/10.1007/BF00290810
Kimble LK, Mandelco L, Woese CR, Madigan MT (1995) Heliobacterium modesticaldum, sp. nov., a thermophilic heliobacterium of hot springs and volcanic soils. Arch Microbiol 163:259–267. https://doi.org/10.1007/BF00393378
Madigan MT (2006) The family Heliobacteriaceae. In: Dworkin M, Falkow S, Rosenberg E, Schleifer KH, Stackebrandt E (eds) The prokaryotes. Springer, New York, pp 951–964
Nonaka H, Keresztes G, Shinoda Y, Ikenaga Y, Abe M, Naito K, Inatomi K, Furukawa K, Inui M, Yukawa H (2006) Complete genome sequence of the dehalorespiring bacterium Desulfitobacterium hafniense Y51 and comparison with Dehalococcoides ethenogenes 195. J Bacteriol 188(6):2262–2274. https://doi.org/10.1128/JB.188.6.2262-2274.2006
Olson DG, Maloney M, Lanahan AA, Hon S, Hauser LJ, Lynd LR (2015) Identifying promoters for gene expression in Clostridium thermocellum. Metab Eng Commun 2:23–29. https://doi.org/10.1016/j.meteno.2015.03.002
Orf GS, Redding KE (2019) Expression and purification of affinity-tagged variants of the photochemical reaction center from Heliobacterium modesticaldum. Photosynth Res 142(3):335–348. https://doi.org/10.1007/s11120-019-00672-3
Orf GS, Gisriel C, Baker PL, Redding KE (2021) The pshX subunit of the photochemical reaction center from Heliobacterium modesticaldum acts as a low-energy antenna subunit. Photosynth Res (under review)
Redding KE, Sarrou I, Rappaport F, Santabarbara S, Lin S, Reifschneider KT (2014) Modulation of the fluorescence yield in heliobacterial cells by induction of charge recombination in the photosynthetic reaction center. Photosynth Res 120(1–2):221–235. https://doi.org/10.1007/s11120-013-9957-4
Rodrigues ML, Oliveira TF, Pereira IAC, Archer M (2006) X-ray structure of the membrane-bound cytochrome c quinol dehydrogenase NrfH reveals novel haem coordination. EMBO J 25(24):5951–5960. https://doi.org/10.1038/sj.emboj.7601439
Sarrou I, Khan Z, Cowgill J, Lin S, Brune D, Romberger S, Golbeck JH, Redding KE (2012) Purification of the photosynthetic reaction center from Heliobacterium modesticaldum. Photosynth Res 111(3):291–302. https://doi.org/10.1007/s11120-012-9726-9
Sattley WM, Madigan MT, Swingley WD, Cheung PC, Clocksin KM, Conrad AL, Dejesa LC et al (2008) The genome of Heliobacterium modesticaldum, a phototrophic representative of the Firmicutes containing the simplest photosynthetic apparatus. J Bacteriol 190(13):4687–4696. https://doi.org/10.1128/JB.00299-08
Sattley WM, Asao M, Tang JKH, Collins AM (2014) Energy conservation in heliobacteria: photosynthesis and central carbon metabolism. In: Hohmann-Marriott M (ed) The structual basis of biological energy generation, 1st edn. Springer, Dordrecht, pp 231–247
Sheehy D, Lu YK, Osman F, Alattar Z, Flores C, Sussman H, Zaare S, Dooling M, Meraban A, Baker P, Touchman JW, Redding KE (2018) Genome-wide transcriptional response during the shift to N2-fixing conditions in Heliobacterium modesticaldum. J Proteomics Bioinform 11(8):143–160. https://doi.org/10.4172/jpb.1000481
Simon J (2002) Enzymology and bioenergetics of respiratory nitrite ammonification. FEMS Microbiol Rev 26(3):285–309. https://doi.org/10.1016/S0168-6445(02)00111-0
Trost JT, Blankenship RE (1989) Isolation of a photoactive photosynthetic reaction center-core antenna complex from Heliobacillus mobilis. Biochemistry 28(26):9898–9904. https://doi.org/10.1021/bi00452a003
Xiong J, Inoue K, Bauer CE (1998) Tracking molecular evolution of photosynthesis by characterization of a major photosynthesis gene cluster from Heliobacillus mobilis. Proc Natl Acad Sci USA 9(25):14851–21456. https://doi.org/10.1073/pnas.95.25.14851
Yeliseev AA, Kaplan S (1999) A novel mechanism for the regulation of photosynthesis gene expression by the tspO outer membrane protein of Rhodobacter sphaeroides 2.4.1. J Biol Chem 274(30):21234–21243. https://doi.org/10.1074/jbc.274.30.21234
Yue H, Kang Y, Zhang H, Gao X, Blankenship RE (2012) Expression and characterization of the diheme cytochrome c subunit of the cytochrome bc complex in Heliobacterium modesticaldum. Arch Biochem Biophys 517(2):131–137. https://doi.org/10.1016/j.abb.2011.11.012
Acknowledgements
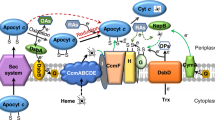
This work was funded by the Division of Chemical Sciences, Geosciences, and Biosciences, Office of Basic Energy Sciences of the US and Department of Energy through Grant DE-SC0010575 to KR. The authors thank William Johnson and Dr. Gregory Orf for creation of Figure 1.
Author information
Authors and Affiliations
Contributions
SL, PB, and KR performed all of the work described in this report. All the authors contributed to writing the paper.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any financial interests related to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Leung, S.W., Baker, P.L. & Redding, K.E. Deletion of the cytochrome bc complex from Heliobacterium modesticaldum results in viable but non-phototrophic cells. Photosynth Res 148, 137–152 (2021). https://doi.org/10.1007/s11120-021-00845-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-021-00845-z