Abstract
Background and aims
Enhanced understanding of plant and nutrient interactions is key to improving yields. We adapted the model for QUantitative Evaluation of the Fertility of Tropical Soils (QUEFTS) to assess cassava yield response to soil and fertilizer nutrients in West Africa.
Methods
Data from on-station and farmer’s field experiments across Togo and Ghana were used.
Results
Soil nutrient supply ranged from 86 to 177, 18–24 and 70–104 kg ha−1 of N, P and K, respectively, and induced variable cassava responses to fertilizer application. Considering harvest (HI) in deriving the physiological nutrient use efficiencies for maximum dilution (PhEmax) and for maximum accumulation (PhEmin), which are key QUEFTS parameters, improved model predictions with reduction in normalized root mean square error from 32 to 13% at Davié (Southern Togo) and from 18 to 13% at Kumasi (Southern Ghana). Model overestimated yields in Nyankpala (Northern Ghana) where drought stress reduced yields. Estimated PhEmin and PhEmax at HI of 0.50 were 41 and 96 kg kg−1 N, 232 and 589 kg kg−1 P, and 34 and 160 kg kg−1 K.
Conclusions
QUEFTS can be used for site-specific estimates of cassava yield responses to fertilizers under rain-fed conditions in West Africa, provided that yield is primarily constrained by N, P and K supplies, and not by drought or other nutrients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The differences between potential or attainable yields and actual yields, known as yield gaps, are large for cassava (Manihot esculenta, Crantz) in West Africa. Yields of fresh storage roots in smallholder farmers’ fields average only 11.2 Mg ha−1 (average 2000–2013) (FAOSTAT 2014), equal to about 4 Mg ha−1 storage root dry matter (DM). This yield is far less than yields of 60 Mg ha−1 (20–24 Mg ha−1 storage root DM) recorded in researcher-managed field experiments in the region (Odedina et al. 2009). A primary constraint is poor soil fertility resulting from the combination of inherently small soil nutrient stocks (Smaling et al. 1997) and continuous cropping with negligible nutrient inputs (Sanchez et al. 1997). Furthermore, soil fertility exhibits strong variability, both among and within farms (Adjei-Nsiah et al. 2007) and fields, making blanket fertilizer recommendations inappropriate. Accurate assessment of nutrient supplies on a site-specific basis is important for enhanced estimates of crop yields. A sound understanding of nutrient uptake and nutrient conversion into crop yield is also required.
Despite the importance of cassava as a staple food and cash crop, there has been little attention for cassava yield predictions in response to nutrient supplies, uptakes and physiological use efficiency. The process-oriented dynamic model CROPSIM cassava (Matthews and Hunt 1994; Singh et al. 1998) designed to simulate cassava growth and development was restricted to the assessment of potential, water-limited and nitrogen-limited yields. Thus, CROPSIM assumes that P and K are not limiting cassava growth, and is therefore not suited to assess nutrient limited yields in the nutrient depleted production systems of West Africa. Particularly, K deficiency is important in such low external nutrient use systems since cassava as a root crop has a high K demand (El-Sharkawy and Cadavid 2000; Howeler 2002; Pellet and El-Sharkawy 1997). The QUEFTS (QUantitative Evaluation of the Fertility of Tropical Soils) model could be a practical tool to assess nutrient requirements in cassava production systems. QUEFTS is a relatively simple and static model that predicts crop yields based on the interactions between the three macronutrients N, P and K, the physiological nutrient use efficiency (PhE) of the crop and the climate and location specific maximum yield as boundary condition (Janssen and Guiking 1990; Janssen et al. 1990). QUEFTS was originally developed for soil fertility evaluation, nutrient requirements assessment and yield prediction for maize under tropical conditions (Janssen et al. 1990). It has been successfully tested in East Africa (Smaling and Janssen 1993), and thereafter adapted for other crops including rice (Haefele et al. 2003; Sattari et al. 2014; Witt et al. 1999), wheat (Pathak et al. 2003) and grain legumes (Franke et al. 2014) in various parts of the world. It has also been used to assess cassava yield and nutrient requirements under Indian conditions in Asia (Byju et al. 2012).
The evaluation of soil supply of nutrient by the original version of QUEFTS was based on soil chemical properties including a minimum dataset of soil organic carbon, available phosphorus, exchangeable potassium and pH (Janssen et al. 1990). However, soil supply of nutrients can also be determined from nutrient omission trials as the total plant uptake from one missing nutrient plots as described in the site-specific nutrient management system for rice production in India (Dobermann and White 1998; Witt et al. 1999). In the QUEFTS model adapted for cassava in India, the assessment of soil supply of nutrients has been done on a zone-specific basis leading to different equations relating soil chemical properties to soil nutrient stock estimates for each zone (Byju et al. 2012). The applicability of these equations for West Africa conditions has not yet been investigated.
The PhE, a key crop specific parameter of QUEFTS also called the internal nutrient use efficiency, is defined as the mass ratio of the economic components of a crop (grain, storage roots) to the quantity of nutrient uptake in the whole crop. As such, PhE is sensitive to HI (kg dry matter yield kg−1 total biomass dry matter), and to differences in the nutrient content of edible and other crop components (Sattari et al. 2014). Whereas cereals maintain most of their leaves until maturation or harvest, cassava leaf biomass is shed during dry seasons. For this reason, it is important to consider senesced leaves in the calculation of the total biomass produced, hence of HI. For cassava, differences in HI between cultivars can be large. These were not considered in the QUEFTS model developed for cassava grown under Indian agro-ecological conditions (Byju et al. 2012), which prevents derivation of variety-specific fertilizer recommendations for cassava. Although HI is measured at harvest, its value is known a priori for every cultivar as provided by breeders. However, besides genetics, HI strongly depends on the environment and management practices.
The current paper aims to assess cassava yield estimates as affected by HI, physiological nutrient use efficiency (PhE), indigenous soil nutrient supplies and fertilizers in West Africa using the QUEFTS model. We hypothesized that accounting for the effects of HI improves the yield prediction of cassava in West Africa, where different cultivars are grown with varying HI. We investigated the relationships between N, P and K uptake and cassava yield and the relationship between soil parameters and quantities of soil N, P and K supplies. This allowed us to calibrate and test the QUEFTS model, and predict cassava yields for different soil conditions and fertilizer amendments. For this purpose data from five different field experiments conducted between 2007 and 2010 in West Africa were used.
Material and methods
Model calibration
Dataset for model calibration
The dataset used for model calibration was collected in three field experiments conducted over two years in three agro-ecological zones (AEZ) of West Africa. The sites were Davié in Togo in the Coastal Savannah, Kumasi in the Deciduous Forest AEZ and Nyankpala in the Southern Guinea Savannah AEZ of Ghana (Table 1). Prior to crop establishment, soil samples were collected at five positions in each site up to 20 cm soil depth. Per site, these samples were mixed and a composite sample of 500 g was taken for laboratory analysis. Soils were analysed for soil organic carbon (SOC), soil total nitrogen, exchangeable cations, soil texture, pH-water, and available phosphorus (P-Bray-I) (Table 2), using the procedures described by Houba et al. (1995). The Rhodic Ferralsols at Davié is a loamy sand with known K deficiency (Table 2), the Ferric Acrisol at Kumasi is a silt loam with high SOC content, and the Gleyi-ferric lixisol at Nyankpala is sandy loam with low SOC content (Table 2) according to the soil fertility classification of Howeler (2002).
The experiments ran for two consecutive seasons at each site between 2007 and 2010 (Table 1). A randomised complete block design (RCBD) with replicates in four blocks and 10 fertilizer N-P-K rates (kg N, P and K ha−1: 0–0-0, 0–40–130, 40–40–130, 80–0-130, 80–20–130, 80–40–0, 80–40-65, 80–40–130, 40–20-65 and 100–50-170) was used. Nitrogen was applied as urea (46%N, Davié and Kumasi) or sulphate of ammonia (21%N, Nyankpala), P as triple super phosphate (TSP: 20%P) and K as potassium chloride (KCl: 50%K). All the TSP and one third of the urea and KCl were applied 30 days after planting (DAP); the remaining urea and KCl at 45 days after the first application. Soil bunds were constructed around each experimental plot to prevent lateral fertilizer contamination between plots. At each site, a locally-popular improved cassava cultivar was planted at the recommended planting density: “Gbazekoute” cultivar (TME-419) in Togo with a planting scheme of 0.8 × 0.8 m (15,625 plants ha−1) in 6.0 × 5.6 m plots, and “Afisiafi” cultivar (TME-3281 or TME-771) in Ghana with a planting scheme of 1.0 × 1.0 m (10,000 plants ha−1) in 7.0 × 7.0 m plots. Hand weeding was carried out four times during the growing season.
Dry matter (DM) yields of storage roots and aboveground biomass (stem and leaves) were measured at final harvest on a harvest plot of 5.12 m2 (eight plant stands) per experimental plot excluding the two border rows. Sub-samples of each harvested plant part (leaf, stem, roots) of each treatment were oven-dried at 70 °C to constant weight and DM mass fractions were determined. Dried plant organs were ground and digested using a H2SO4 – salicylic acid – H2O2 – Selenium mixture. Total N concentration was measured in this extract using a colorimetric method based on Berthelot’s reaction (Sommer et al. 1992), total P concentration based on the method of the molybdo-phosphate complex with ascorbic acid as a reducing agent and K concentration by atomic absorption spectrophotometry using the Perkin Elmer model Analyst 400 (Houba et al. 1995).
Yield prediction procedure of the original QUEFTS model
The prediction of crop yield in response to nutrient supplies by QUEFTS follows four main steps (Janssen and Guiking 1990; Janssen et al. 1990). Step 1: the nutrient supply from soil and inputs of organic materials or fertilizer is estimated. Step 2: the uptake of a nutrient is calculated as a function of the total supply of that nutrient, and the interaction with the two other macronutrients; Step 3: for each nutrient uptake, two yields are calculated by the model, one corresponding to a situation where the nutrient is maximally diluted in the crop, and another one corresponding to a situation of maximum accumulation of that nutrient in the crop. Step 4: using the yield ranges defined in Step 3, the yield is calculated for each pair of nutrients, and the average yield of all pairs of nutrients is retained as the final yield estimate of the crop. These yields are calculated considering the climate and location-specific maximum yield of the cultivar as boundary condition.
Calibration of QUEFTS for cassava following the four steps for yield prediction of the model
Assessment of soil and input supplies of available nutrients (Step1)
The supply of the total available nutrient (denoted as TAβ for a given nutrient β) for the crop was estimated from the supply of soil available nutrients (SAβ) and the supply from fertilizer inputs (SIβ) as follows:
Where TAβ is the total amount of available β; β stands for a given nutrient (N, P or K); MRF for the apparent maximum recovery fraction of that nutrient; I the amount of input (fertilizer nutrient) applied.
The assessment of SAβ followed two procedures to choose the one that matches better the experimental data (observed SAβ values). We first tested existing equations for assessing SAβ values (based on initial soil chemical properties comprising pH, SOC, available P and exchangeable K measured before planting) used in previous QUEFTS versions. The latter included the original version by Janssen et al. (1990) as modified version by Sattari et al. (2014) and the version developed for cassava in India by Byju et al. (2012). For the latter, SAβ equations for Kerala and Andhra Pradesh cassava production zones (Byju et al. 2012) were used since P-Bray 1 was available. Subsequently, the values of SAβ and SIβ were graphically determined by plotting the observed maximum uptake (y axis) against fertilizer application rates (x axis) of a given nutrient. Given probable effects of fertilizer application on nutrient uptakes, all treatments with the same application rate of the relevant nutrient were used to calculate SAβ and SIβ to ensure that the maximum observed uptake of the nutrient of interest at a given rate is captured. For instance, the treatments assessed at 0 kg N ha−1 were 0–0-0 and 0–40–130; at 40 kg N, these were 40–20-65 and 40–40–130; at 80 kg N ha−1, these were 80–0-130, 80–20–130, 80–40–0, 80–40-65 and 80–40–130. Among these treatments, the nutrient uptake in the treatment with the highest yield was taken as a proxy for SAβ + SIβ, since the relevant nutrient was expected to be more limiting in this treatment than in the others. In addition, various percentiles (75th and 87.5th) of the distribution of N, P and K uptakes were tested, as well as nutrient uptake in the treatments theoretically most appropriate for the purpose (e.g. 0–40–130, 40–40–130 and 80–40–130 for N). This was done per replicate as well as for the average of the four replicates. After these tests, it was decided to take the 75th percentile uptake found in all treatments with an equal application of the relevant nutrient. This selection was based on the fit between observed nutrient uptake and calculated nutrient uptake determined in Step 2 of QUEFTS (next Section). Where only two treatments could be compared (e.g. 0–0-0 and 0–40–130), the 75th percentile equalled: L + 0.75 × (H-L), where L and H stand for the lower and the higher value found in the two treatments. Plotting the calculated nutrient uptake (y-axis) versus the fertilizer application rates of a nutrient (x-axis) provided a linear regression of which the value of the intercept with the y-axis was used as SAβ, and the slope was considered as MRFβ.
Calculated uptake in relation to supply of nutrients (step 2)
To calculate the uptake of each of the three nutrients, the original procedure of QUEFTS was followed (Janssen et al. 1990; Sattari et al. 2014). The uptake of nutrient 1 is calculated twice: i) as a function of the supplies of nutrients 1 and 2, and ii) as a function of the supplies of nutrients 1 and 3. The lesser of the two outcomes is considered more realistic following Liebig’s law of the minimum and referred to as ‘calculated nutrient uptake’. Calculated uptake of a given nutrient is less than the total supply. The latter refers to the maximum quantity of that nutrient that can be taken up. According to Janssen et al. (1990), the calculated uptake of a given nutrient equals the total supply only if all other growth conditions are optimum. Calculated nutrient uptakes were determined for each site based on specific SAβ and SIβ values, and compared to the observed uptakes.
Relationships between yield and nutrient uptake (steps 3 and 4 of QUEFTS)
In Step 3, calculated nutrient uptake is converted into estimates of yield ranges based on the minimum and maximum PhE of the relevant nutrient. Two approaches were tested to derive the minimum and maximum PhE values from the model calibration dataset. The first approach (Approach 1) consisted of plotting observed nutrient uptake (Uβ) in storage roots and tops (leaves plus stems) against observed storage root yield (Y), and to determine upper and lower boundary lines (Byju et al. 2012; Janssen et al. 1990; Pathak et al. 2003; Witt et al. 1999). Following Witt et al. (1999), boundary lines for yields at maximum dilution (Y d ) and maximum accumulation (Y a ) were drawn based on data within the upper and lower 2.5 percentiles, respectively. The ratio of Yβ d /Uβ represents the maximum PhE (PhEmax), and the ratio Yβ a /Uβ the minimum PhE (PhEmin) of a given nutrient β. As recommended by several studies (Byju et al. 2012; Witt et al. 1999), to ensure that crop growth was mainly limited by nutrients, observations with an HI less than 0.40 were removed (six observations were removed, corresponding to 2.5% of the dataset with in total 240 observations). It was assumed that the intercepts of the boundary lines with the x-axis, describing the minimum nutrient uptakes required to produce measurable yield (Janssen et al. 1990) were nil since even the smallest nutrient uptakes values in our dataset were enough to produce storage roots yields.
The alternative approach (Approach 2) to derive PhEmin and PhEmax was used recently in cereals yield predictions (Sattari et al. 2014) for situations with strongly varying values of HI. For cassava it holds:
where Mass roots, Mass tops and Mass total stand for roots mass, tops mass and total (bio)mass respectively. Mass total is the sum of roots and tops masses.
Total uptake of a nutrient (Uβ) is:
where Cβ roots and Cβ tops are the mass fractions (g nutrient kg−1 DM) in cassava roots and tops, respectively, and Uβ is expressed in kg β per ha. 1000 is a conversion factor from g to kg.
PhEβ, expressed in kg DM kg−1 nutrient, is:
Substitution of Eq. 3 in Eq. 4 yields:
PhEmax and PhEmin values can be calculated as:
where Cβ min and Cβ max denote the minimum and maximum values of mass fractions (g kg−1) of a given nutrient. These values were obtained either from literature (Nijhof 1987) or derived from our model calibration experiments. We used Cβ min and Cβ max values of the entire dataset (model calibration experiments) to calculate PhEmax and PhEmin.
The two approaches to derive PhEmin and PhEmax values were tested based on the assumption that the best approach will provide the most accurate estimate of the yield if the estimates of the uptake of N, P and K are accurate (good fit between calculated and observed uptake of N, P and K). On that basis, Steps 3 and 4 were run to calculate yields with observed uptake of N, P and K as input variables, and subsequently calculated yields were compared with observed yields. The approach providing the most accurate estimates of observed yields was applied in Step 2 of QUEFTS to calculate nutrient uptakes as a function of nutrient supplies. The medium PhE denoted as PhEmed was calculated as the average value between PhEmax and PhEmin. The maximum yields obtained per site for each of the two growing seasons in the model calibration experiments were used as climate, location and cultivar specific maximum yield required as a boundary condition to run QUEFTS.
Model testing
Data collected in two additional on-farm fertilizer trials in Togo and Ghana were used to test model performance. Different rates of NPK fertilizers (kg ha−1) were used: 0–0-0, 20–10-80, 40–20-65, 60–25-120 and 100–40-150 at Davié-Tekpo, Gbave and Sevekpota in Southern Togo, and NPK: 0–0-0, 48–0-95, 68–28-155, 82–28-155, 98–55-183 in Savelugu and Gbanlahi in Northern Region of Ghana. Fertilizer applications methods were the same as in the experiments described above. Except for Sevekpota where individual farmers hosted a single replicate of the 5 treatments (7 farmers in total), each farmer field at the other locations had four replicates laid out following a RCBD. Planting density followed recommended practices of each area. Healthy cuttings of ‘Gbazekoute’ cultivar were planted April 26, 2010 in southern Togo at a density of 15,625 plant ha−1 (0.8× 0.8 m on 6.0× 5.6 m sub-plots), and the storage roots were harvested on March 22, 2011. In Ghana, the planting of Afisiafi cultivar cuttings was performed on June 21, 2011 in Gbanlahi and on June 22, 2011 in Savelugu at a density of 10,000 plant ha−1 (1.0 × 1.0 m on 7.0 × 7.0 m sub-plots) and the harvest on December 18, 2012 and December 12, 2012 respectively. Data were collected on dry matter yields of storage roots, stems and leaves at all sites, and soil chemical data (obtained as described above in Section “Dataset for model calibration”) from the Togolese sites only (Table 2).
The performance of the model was tested using the PhEmin and PhEmax values found to be best in the comparison of the two approaches (See Section “Relationship between yield and nutrient uptake”). Since no plant chemical data (plant N, P and K uptakes) and no minus one fertilizer nutrient treatments (nutrient omission treatments) were available in the model testing experiment, we assessed how well the model estimates cassava yield response to mineral fertilizer rates when the yield of the control plot (no fertilizer plot) is assumed well estimated by the model. Based on that principle, observed control plot yields were used to assess SAβ values at each site. For this reason, yield from control plots were excluded when testing the model performance. From a given starting value for each nutrient each location, SAβ were subsequently adjusted until good agreements were found between calculated and observed yields on the control plots. After SAβ values were obtained, the model’s ability to estimate cassava yield in response to fertilizer applications was evaluated using only the treatments that did receive fertilizer in the model testing trials. The evaluation was done first with MRF values derived from the model calibration trials. In following runs, MRF values were adjusted per site to test model sensitivity to MRF values and their effects on yield predictions of the model.
Model calculations were compared to observations using: the Root Mean Squared Error (RMSE), the Normalised Root Mean Squared Error (NRMSE) (Loague and Green 1991), the Willmott index of agreement (Willmott et al. 1985), the comparison with the 1:1 line, and the coefficient of determination (R 2) and the regression line slope. Differences between sites in observed yields and uptake of N, P and K were quantified using linear mixed models, with site as fixed factor, and year and block as random factors. The analysis of the differences in yields and uptake of N, P and K between years was done per site using general linear models. A probability threshold P of 0.05 was used in all analyses to assess significance.
Results
Model calibration
Observed cassava storage roots yield and nutrient uptake
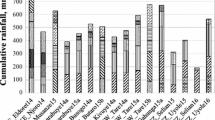
Storage root yields (± standard deviation) significantly (P < 0.001) differed between sites when averaged over all 10 treatments in the model calibration experiments, and amounted 13,248 ± 3144, 10,544 ± 3591 and 6538 ± 2228 kg ha−1 in Davié, Kumasi and Nyankpala, respectively. Yields obtained in 2008 were larger than those achieved in 2007 at Davié (14,043 vs 12,453 kg ha−1 respectively, P = 0.023) and at Nyankpala (7745 vs 5331 kg ha−1 respectively, P < 0.001) (Fig. 1); higher yields were also obtained in 2008 than in 2009 in Kumasi (13,269 vs 7749 kg ha−1 respectively, P < 0.001). The amount of rainfall during the growing season was highest in 2008 compared to 2007 in Davié and Nyankpala, and to 2009 in Kumasi (Table 1). Differences in nutrient uptakes between seasons reflected yields (Fig. 1). The largest total N and P uptake was found in Davié (P < 0.001) and the smallest in Nyankpala, whereas the smallest K uptake was obtained in Kumasi and the largest in Davié (P < 0.001). Averaged over all 10 treatments and growing seasons, total nutrient uptakes (± standard deviation) per growing season were 196.3 ± 62.4 kg N, 33.3 ± 10.8 kg P and 152.7 ± 69.9 kg K ha−1 in Davié, 100.8 ± 26.9 kg N, 22.3 ± 6.8 kg P and 68.0 ± 24.4 kg K ha−1 in Kumasi, 103.5 ± 35.5 kg N, 17.1 ± 6.4 kg P and 121.1 ± 47.4 kg K ha−1 in Nyankpala.
Relationships between N, P and K uptakes and roots DM yields, and the estimated yields at maximum dilution and maximum accumulation of a nutrient (Yβd and Yβa), and the medium value between Yβd and Yβa (Yβm). Each point represents the average value of four replicates. Data from all the treatments (10 fertilizer combinations) of the model parameterisation experiment are included. The dry matter of cassava was on average 38% and 36% of fresh matter for Gbazekoute and Afisiafi respectively
Supply of available soil and fertilizer nutrients (step 1)
The assessment of SAβ based on initial soil properties (Table 2) using equations by Janssen et al. (1990) (original QUEFTS) and by Byju et al. (2012) yielded large deviations relatively to the observed plant uptakes from one missing nutrient plots (Table 3). The SAP was strongly underestimated by both the Janssen et al. (1990) and Byju et al. (2012) equations for SAβ by 34–83%. Deviations in estimating SAN and SAK were also large with both methods, ranging from underestimation by 12–84% for N to overestimation by 9 to 339% for K. This indicates that SAβ equations by Janssen et al. (1990) defined for maize are not applicable for cassava. Likewise, SAβ equations of Byju et al. (2012) are not applicable to West African conditions, suggesting the need for their adaptation. Although, we had 240 observation points for plant uptake data, only three composite soil chemical and physical data were available for the three model calibration sites, and there was no plant N, P and K uptake data from the five validation trial locations. Those were insufficient to derive strong relationships between SAβ and measured soil parameters. Under such circumstances, the alternative approach of assessing SAβ and MRFβ using linear regression approach appeared more appropriate and generated values that differed between sites (Table 4), especially for N and K. The SAβ of N (SAN) decreased in the order of Davié > Kumasi > Nyankpala. Similarly, SAβ of K (SAK) decreased in the order of Nyankpala > Davié > Kumasi. The variation in SAP (SAβ for P) between sites was small, since all three sites had soils with a low available P status (Table 2) according to Howeler (2002). In Davié, MRF for K was very large (Table 4), likely due to the pronounced soil K deficiency there. However, a MRF for K above 100% was not expected, though this phenomenon has also been reported by Pellet and El-Sharkawy (1997) as the result of the ability of the crop to remove large amounts of K from the soil. Values of MRF at Kumasi were very small, suggesting limited external nutrient uptake and little nutrient limitations for cassava production. The SOC content on this site was larger than elsewhere (Table 2). The smaller MRF for K at Nyankpala with a larger exchangeable K content when compared to Davié was expected.
Calculated uptake in relation to total supply of nutrients (step 2)
Nutrient uptakes calculated with Step 2 of QUEFTS were in good agreement with the observed uptakes of N, P and K as indicated by the value of the slope of the regression line and R 2 close to 1 (Fig. 2). Regression analyses for each site separately gave slightly smaller R 2 values (not shown) than a single analysis for all sites together.
Uptake of N (a), P (b) and K (c) as calculated in Step 2 in relation to observed uptake, and the associated regression line. Input variables for Step 2 were the soil and input supplies of nutrients estimated in Step 1. Each point represents the average observed uptake of eight values (four replicates, two seasons)
Physiological nutrient use efficiency (steps 3 and 4)
The two approaches for deriving PhEmin and PhEmax are illustrated in Fig. 1 (Approach 1, not HI related) and Fig. 3 (Approach 2, HI related). In Approach 1, PhEmin and PhEmax values (Fig. 1) represent 2.5 and 97.5th percentiles of all points and correspond to the boundary line for maximum accumulation (Y a ) and for maximum dilution (Y d ) respectively. The six site/year combinations have different positions in the envelopes, with Kumasi 2008 being closer to the boundary line for maximum dilution (Y d ) for N (Fig. 1a) and K (Fig. 1c), and Nyankpala closer to the boundary line of maximum accumulation (Y a ), especially for K. The points in the scatter graph of P uptake and roots yield (Fig. 1b) are closer together than those for N and K (Fig 1a, c, respectively), especially at low P uptake.
Physiological nutrient use efficiency (PhE) of N, P and K in relation to harvest index (HI). PhEmax and PhEmin represent physiological nutrient use efficiency at maximum dilution and maximum accumulation, respectively, and PhEmed the medium value between PhEmax and PhEmin. Each point is calculated with Eqs. 6 and 7 and measured nutrient mass fractions of both cultivars combined (Table 5), and represents the average of four replicates. Nijhof curves were also based on these equations, but with nutrient mass fractions from Nijhof (1987) (Table 5)
In Approach 2, the HI and the minimum and maximum mass fractions in roots and tops (Table 5) were used in Eqs. 6 and 7 to derive PhEmax and PhEmin (Table 6). Measured root nutrient mass fractions were generally within the ranges given by Nijhof (1987). Fig. 3 shows that PhE varies with HI across sites and years. It also shows that PhE of N was small compared with literature since all points are situated between PhEmed and PhEmin of Nijhof (1987). Fig. 3 also shows that PhE of P is within a comparable range across the three sites, and that PhE of K is generally large at Davié and Kumasi but small at Nyankpala, pointing out large K supply at the latter site. Furthermore, the largest values of PhE of K were achieved at high HI values, and vice versa, indicating that PhE of K increases with HI.
The comparison of the two approaches to determine PhEmin and PhEmax suggested that Approach 2 worked better at Davié and Kumasi (Table 7). Although the performances of the two approaches were comparable in terms of R 2, Approach 2 provided more accuracy in the prediction with smaller RMSE and NRMSE, and a Willmott index closer to 1. These results stress the importance of accounting for the influence of HI on PhEmax and PhEmin in predicting cassava yields.
Model performance was best for Davié with calculated and observed yields scattered around the 1:1 line, and poorest for Nyankpala with an overestimation of observed yields by the model (Fig. 4). Since average values of HI were used by QUEFTS whereas HI varied over seasons, observed yields were overestimated in case the real HI was smaller than the average HI, and underestimated in case the real HI was larger than the average HI. At Nyankpala, calculated yields were much larger than observed yields (Figs 4 and 5). This is in agreement with the low PhE values observed at this site, which suggests an inefficient nutrient use due to luxury nutrient uptake. Planting was late in Nyankpala in the first year (June 29, 2007), whereas the rainy season ran from April to October, meaning that the crop benefited from four months of rain at most. The second half of the growing season the crop likely suffered from drought, causing a low PhE.
Relations between yields calculated with Step 3 and 4 of QUEFTS using HI related PhE boundary lines (Approach 2) and observed yields for Davié (a), Kumasi (b) and Nyankpala (c). Input variables for Step 3 were the observed nutrient uptakes. HI values were set at 0.50 for Davié and at 0.65 for Kumasi and Nyankpala. Each point represents the average yield of four replicates
The comparison of PhE values using an hypothetical cultivar with an HI value of 0.4 (Table 6) to those reported under Indian agro-ecological conditions by Byju et al. (2012) revealed that PhE values are higher in India, especially for P, pointing to stronger P dilution than in West Africa. Only PhEKmax was higher in West Africa, reflecting poor K availability, which was especially evident on the Ferralsols in Davié (Fig. 3).
Yields in relation to the total supply of available nutrients (steps 1–4)
Using the calibrated QUEFTS (PhEmin, PhEmax and HI; Table 6), SAβ and MRF values (Table 4), the best fit between observed and calculated yields were obtained at Davié (Fig. 5). At Kumasi, calculated and observed yields agreed better in 2008 than in 2009 (Fig. 5) when observed yields were smaller than calculated yields. The smaller observed yields in 2009 compared to 2008 were likely due to smaller amounts and inadequate distribution of rainfall in 2008. About 49% of total rainfall in the growing season (Table 1) occurred in the first month after planting (not shown). Most of this water was likely lost through evaporation as soil coverage by cassava was small in the first month after planting. At Nyankpala, calculated yields were strongly overestimated in both years (Figs 4 and 5). As suggested above, the growth conditions in Nyankpala during the first part of the growing seasons allowed the crop to take up available nutrients to produce top biomass, while drought likely limited growth later in the season, causing an inefficient translocation of photosynthetates to the storage roots, hence strongly hindering storage root yield.
Model testing
Calculated yields agreed well with observed yields (Fig. 6). This indicates that the model can effectively estimate cassava response to fertilizer N, P and K (Fig. 6a), provided that SAβ values are estimated in such a way to adequately assess yields on control plots. However, the use of site specific MRF values slightly improved yield predictions (Fig. 6b), indicating that the difference between calculated and observed yields were at least partly due to differences in MRF values between sites.
Calculated yields in relation to observed yields in the model verification trials with common MRF values (a) or adjusted per site (b). Input variables for Step 1 were estimated soil supplies of available nutrients of Table 8 and maximum recovery fractions of Tables 4 (Fig. 6a) and 8 (Fig. 6b). Each point represents the average yield of two to five replicates
Discussion
This paper showed that the model can provide sound estimates of cassava yields when SAβ and MRFβ are accurately assessed and that PhEmin and PhEmax are estimated based on HI in areas where HI is very variable. The use of previous QUEFTS equations by Janssen et al. (1990) and Byju et al. (2012) for assessing SAβ underestimated SAN by 12–84% and SAP by 34–83%, and overestimated SAK by 9–339% (Table 3). Most of SAβ values were underestimated by Janssen et al. (1990) equations for all three nutrients (Table 3) probably because such equations were developed for cereals, which have a much shorter growing period (3–4 months) than cassava (6 to more than 12 months). Moreover, cassava is more effective than cereals in P uptake under P-limiting conditions due to cassava’s strong mycorrhizal symbiosis (Kang and Okeke 1984; Sieverding and Leihner 1984). In the case of the Byju et al. (2012) equations for assessing SAβ, variable trends were observed for SAN, while SAP was underestimated by 34–74% and SAK overestimated by 81–339%. Further improvement of the model should consider relationships between nutrient stocks and soil chemical properties in West Africa as well as links with crop duration, soil depth, management and rainfall that can affect nutrient supplies and uptake by cassava.
The alternative method adopted in this study by graphically assessing SAβ provided results that reflected differences between sites, especially for N and K (Table 4). The largest value of SAβ for N (SAN) was obtained at Davié, rather than Kumasi which had larger SOC, because Kumasi had larger PhE N for the same amount of N uptake (Fig. 1a). Moreover, the C:N ratio of Kumasi site was lower compared to Davié (Table 2), indicating higher SOC mineralization leading to higher N availability and susceptibility to losses in Kumasi. The highest SAK was estimated at Nyankpala, because of the high availability of K in the soil (Table 2). Similar SAP values were obtained across all sites since all sites were poor in available P.
The graphically estimated MRF values (Table 4) also reflected soil nutrient availability across sites. The strong K deficiency explained the high MRF of K at Davié. The large SOC at Kumasi with large soil N supply resulted in a relatively small MRF of N at this site. The MRF of P varied across sites, with the smallest value obtained at Nyankpala and the largest at Davié. Since all sites had soils with low available P, the difference in MRF of P may be attributed to differences in P requirements to meet the yield potential across sites, and to mycorrhizal enhancing effects on P use efficiency of cassava (Kang and Okeke 1984; Sieverding and Leihner 1984).
The evaluation of the relationships between nutrient uptakes and yields of cassava showed that accurate estimates of nutrient uptakes resulted in accurate assessments of yields in Davié and Kumasi (Fig. 4). This suggests that relationships characterized by PhEmax, PhEmin and HI (Eqs 6 and 7) provided a satisfying description of reality. Considering harvest (HI) in deriving PhEmax and PhEmin improved model predictions with reduction in NRMSE from 32 to 13% at Davié (Southern Togo) and from 18 to 13% at Kumasi (Southern Ghana) (Table 7). The situation was different at Nyankpala where both approaches (with and without considering HI in assessing PhEmax and PhEmin) generated high NRMSE values (47–60%) indicating high deviations between observed and calculated yields. At Nyankpala, QUEFTS-calculated yields were one and a half times larger than observed yields, which can be ascribed to the occurrence of drought while the crop was still in the active vegetative stage (Alves 2002). This constitutes a major limitation of the current versions of the model as the model applicability is limited in more arid zones, as in the case of Nyankpala in Northern Ghana. Process-based models with smaller time-step of integration to assess the growth at different stage of development of the plant are more relevant for these zones. Unfortunately the few existing process-based models are also constrained for the assessment of the dynamics of all the macronutrients, especially K, which is important for cassava production in West Africa. An integrated approach combining QUEFTS with these process-based models could be useful under such growing conditions. However, at the moment, no validated process-based model for cassava is available under West African conditions. The overestimation of yields at Nyankpala can also be attributed to nutrient deficiencies (other than N, P and K): the small concentration of magnesium (4.9 mmol kg−1) below the critical value of 6.0 mmol kg−1 for cassava (Snapp 1998), could have contributed to the overall weak response of cassava at this site. Strong yield responses to magnesium were obtained in Colombia on depleted soils (CIAT 1985).
The comparison of the studied cultivars with the Indian cultivars used by Byju et al. (2012) on the basis of an HI value of 0.40 revealed that our cultivars had lower PhEmax for P and higher PhEmax for K (Table 6). In other words, they diluted less P and more K than the Indian cultivars. This suggests that the physiological use efficiency of P can be further improved in West Africa. Breeding to improve the dependency of cassava cultivars on mycorrhizal association for higher P use efficiency could contribute to improving this P physiological use efficiency, which will lift up the physiological use efficiency of N and K. Since higher physiological use efficiency of nutrient implies reduced nutrient requirement for the same target yield, this will generate larger returns on investments to farmers. The higher PhEmax for K of the cultivars grown in our study compared to the Indian cultivar is due to the fact that K was very deficient and the most limiting nutrient for cassava on the Ferralsols in Davié, as also observed on the Ferralsols in Southern Benin (Carsky and Toukourou 2005).
Calculated yields were close to observed values in the model testing experiments (Fig. 6). With SAβ estimates set at a value that QUEFTS compared best to observed control plot yields, the model was able to properly predict cassava responses to combined N, P and K applications. The absence of plant and soil chemical analyses data to derive SAβ is common in sub-Saharan Africa. The method used in this paper of deriving SAβ from control plots without fertilizer can be used when observed yield data from these plots are available. In case yields and plant N, P and K content data from nutrient omission trials (Dobermann et al. 2002; Witt et al. 1999) are available, plant uptakes from one missing nutrient plots can be used as proxy for SAβ. However, nutrient omission trials cannot be conducted everywhere, hence the need to have sufficient soil data to relate plant uptakes from these nutrient omission plots with soil chemical data that are more available in order to ease the extrapolation of the results. Otherwise, the method used in the model parameterization trial in this paper can be applied. This latter method has the advantage of providing a good proxy of the maximum recovery fractions (MRFβ) as the slope of the regression line between the maximum plant uptake and the fertilizer rate of a given nutrient, along with the SAβ assessed as the intercept of this regression line. However, the availability of plant and soil chemical data is ideal to relate SAβ to soil parameters like in Step 1 equations of the original version of QUEFTS. The calculations were further improved by use of site specific MRF values (Fig. 6b), highlighting the importance of location specific soil nutrient management for achieving higher cassava yields.
Conclusions
QUEFTS was adapted to estimate cassava yield and responses to mineral fertilizers under rain-fed conditions in West Africa. In years with normal rainfall, the model calculations produced yield estimates close to those observed, but the model overestimated yields under drought conditions. While the current model could be improved through further model testing experiments in other locations in West Africa and with the development of equations for estimating SAβ to cassava based on soil properties, it provides a useful tool for estimating cassava yield response to fertilizer applications. The strong crop responses to N, P and K highlight the importance of replenishing soil nutrients through external nutrient supplies in cassava production systems. Moreover, our study highlighted the importance of relating the estimates of PhE for maximum accumulation and PhE for maximum dilution to HI in cassava. Since PhE increased with HI, plant breeders should work towards developing cultivars with enhanced HI to improve nutrient use efficiency and yields in cassava production systems in West Africa.
References
Adjei-Nsiah S, Kuyper TW, Leeuwis C, Abekoe MK, Giller KE (2007) Evaluating sustainable and profitable cropping sequences with cassava and four legume crops: effects on soil fertility and maize yields in the forest/savannah transitional agro-ecological zone of Ghana. Field Crop Res 103:87–97
Alves AAC (2002) Cassava botany and physiology. In: Hillocks RJ, Thresh JM, Bellotti AC (eds) Cassava: biology, production and utilization. CABI, Wallingford
Byju G, Nedunchezhiyan M, Ravindran CS, Santhosh Mithra VS, Ravi V, Naskar SK (2012) Modeling the response of cassava to fertilizers: a site-specific nutrient management approach for greater tuberous root yield. Commun Soil Sci Plant Anal 43:1149–1162
Carsky RJ, Toukourou MA (2005) Identification of nutrients limiting cassava yield maintenance on a sedimentary soil in southern Benin, West Africa. Nutr Cycl Agroecosyst 71:151–162
CIAT (1985) Cassava program. In: CIAT (ed) annual report for 1982 and 1983. CIAT, Cali
Dobermann A, White PF (1998) Strategies for nutrient management in irrigated and rainfed lowland rice systems. Nutr Cycl Agroecosyst 53:1–18
Dobermann A, Witt C, Dawe D, Abdulrachman S, Gines HC, Nagarajan R, Satawathananont S, Son TT, Tan PS, Wang GH (2002) Site-specific nutrient management for intensive rice cropping systems in Asia. Field Crop Res 74:37–66
El-Sharkawy MA, Cadavid LF (2000) Genetic variation within cassava germplasm in response to potassium. Exp Agric 36:323–334
FAOSTAT (2014) FAO statistics. FAO Statistics Division, Rome
Franke AC, van den Brand GJ, Giller KE (2014) Which farmers benefit most from sustainable intensification? An ex-ante impact assessment of expanding grain legume production in Malawi. Eur J Agron 58:28–38
Haefele SM, Wopereis MCS, Ndiaye MK, Barro SE, Ould Isselmou M (2003) Internal nutrient efficiencies, fertilizer recovery rates and indigenous nutrient supply of irrigated lowland rice in Sahelian West Africa. Field Crop Res 80:19–32
Houba VJG, Van der Lee JJ, Novozamsky I (1995) Soil analysis procedures, other procedures. vol. 5B. Wageningen Agricultural University, Wageningen, pp 217
Howeler RH (2002) Cassava mineral nutrition and fertilization. In: Hillocks RJ, Thresh JM, Bellotti AC (eds) Cassava: biology, production and utilization. CABI, Wallingford
Janssen BH, Guiking FCT (1990) Modelling the response of crops to fertilizers. In: van Beusichem ML (ed) Plant nutrition - physiology and applications. Kluwer Academic Publishers, Dordrecht
Janssen BH, Guiking FCT, van der Eijk D, Smaling EMA, Wolf J, van Reuler H (1990) A system for quantitative evaluation of the fertility of tropical soils (QUEFTS). Geoderma 46:299–318
Kang BT, Okeke JE (1984) Nitrogen and potassium responses of two cassava varieties grown on an alfisol in southern Nigeria. Proceedings, 6th symposium International Society of Tropical Root Crops, lima, Peru, 21-26 February 1983, pp 231–37
Loague K, Green RE (1991) Statistical and graphical methods for evaluating solute transport models: overview and application. J Contam Hydrol 7 (1-2):51–73
Matthews RB, Hunt LA (1994) GUMCAS: a model describing the growth of cassava (Manihot Esculenta L. Crantz). Field Crop Res 36:69–84
Nijhof K (1987) Concentration of macro-elements in economic products and residues of (sub)tropical field crops. Staff working paper SWO-87-08 52
Odedina SA, Odedina JN, Ogunkoya MO, Ojeniyi SO (2009) Agronomic evaluation of new cassava varieties introduced to farmers in Nigeria. In: African crop science conference proceedings. African Crop Science Society, Uganda, pp 77–80
Pathak H, Aggarwal PK, Roetter R, Kalra N, Bandyopadhaya SK, Prasad S, Van Keulen H (2003) Modelling the quantitative evaluation of soil nutrient supply, nutrient use efficiency, and fertilizer requirements of wheat in India. Nutr Cycl Agroecosyst 65:105–113
Pellet DM, El-Sharkawy MA (1997) Cassava varietal response to fertilization: growth dynamics and implications for cropping sustainability. Exp Agric 33:353–365
Sanchez PA, Shepherd KD, Soule MJ, Place FM, Buresh RJ, Izac A-MN, Mokwunye AU, Kwesiga FR, Ndiritu CG, Woomer PL (1997) Soil fertility replenishment in Africa: an investment in natural resource capital. In: Buresh RJ, Sanchez PA, Calhoun FG (eds) Replenishing soil fertility in Africa. Soil Science Society of America and America Society of Agronomy, Madison
Sattari SZ, van Ittersum MK, Bouwman AF, Smit AL, Janssen BH (2014) Crop yield response to soil fertility and N, P, K inputs in different environments: testing and improving the QUEFTS model. Field Crop Res 157:35–46
Sieverding E, Leihner DE (1984) Influence of crop rotation and intercropping of cassava with legumes on VA mycorrhizal symbiosis of cassava. Plant Soil 80:143–146
Singh U, Matthews RB, Griffin TS, Ritchie JT, Hunt LA, Goenaga R (1998) Modeling growth and development of root and tuber crops. Systems Approaches for Sustainable Agricultural Development 7:129–156
Smaling EMA, Janssen BH (1993) Calibration of quefts, a model predicting nutrient uptake and yields from chemical soil fertility indices. Geoderma 59:21–44
Smaling EMA, Nandwa SM, Janssen BH (1997) Soil fertility in Africa is at stake! In: Buresh RJ, Sanchez PA, Calhoun FG (eds) Replenishing soil fertility in Africa. Soil Science Society of America and American Society of Agronomy, Wisconsin
Snapp SS (1998) Soil nutrient status of smallholder farms in Malawi. Commun Soil Sci Plant Anal 29:2571–2588
Sommer SG, Kjellerup V, Kristjansen O (1992) Determination of total ammonium nitrogen in pig and cattle slurry - sample preparation and analysis. Acta Agriculturae Scandinavica Section B-Soil and Plant Science 42:146–151
Willmott CJ, Ackleson SG, Davis RE, Feddema JJ, Klink KM, Legates DR, O'Donnell J, Rowe CM (1985) Statistics for the evaluation and comparison of models. J Geophys Res 90 (C5):8995
Witt C, Dobermann A, Abdulrachman S, Gines HC, Guanghuo W, Nagarajan R, Satawatananont S, Thuc Son T, Sy Tan P, Van Tiem L (1999) Internal nutrient efficiencies of irrigated lowland rice in tropical and subtropical Asia. Field Crop Res 63:113–138
Acknowledgements
We are grateful to many researchers and support staff that contributed in different ways for the successful completion of this study funded by DGIS through the SAADA project implemented by the International Fertilizer Development Centre (IFDC).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: John A. Kirkegaard.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Ezui, K.S., Franke, A.C., Ahiabor, B.D.K. et al. Understanding cassava yield response to soil and fertilizer nutrient supply in West Africa. Plant Soil 420, 331–347 (2017). https://doi.org/10.1007/s11104-017-3387-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3387-6