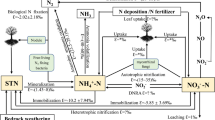
Abstract
Background and aims
To find out how N-saturated forests can return to an N-limited state, we examined the recovery of biotic N sinks under decreasing N supply.
Methods
. We studied a 40-year-old experiment in Pinus sylvestris forest, with control plots, N0, three N treatments, N1-N3, of which N3 was stopped after 20 years, allowing observation of recovery.
Results
In N3, the N concentration in foliage was still slightly elevated, but the N uptake capacity of ectomycorrhizal (ECM) roots in N3 was no longer lower than in N0. Per area the amount of a biomarker for fungi, here mainly attributed ECM, was higher in N3 and N0 than in N1 and N2. Retention of labeled 15NH4 + by the soil was greater in the control (99 %) and N3 (86 %), than in N1 (45 %) and N2 (29 %); we ascribe these differences to biotic retention because cation exchange capacity did not vary. Gross N mineralisation and retention of N correlated, negatively and positively, respectively, with abundance of ECM fungal biomarker.
Conclusions
. The results suggest a key role for ECM fungi in regulating the N cycle. We propose, in accordance with plant C allocation theory, that recovery is driven by increased tree below-ground C allocation to ECM roots and fungi.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Human activities have more than doubled the additions of reactive nitrogen to natural ecosystems (Vitousek et al. 1997; Bobbink et al. 2010). These additions continue to increase in the most rapidly expanding economies, in particular in Asia and South America, whereas in North America and Europe there have been slight reductions in the deposition of NO3 − and in Europe also of NH4 + since the late 1970s (Lajtha and Jones 2013). Most high-latitude forests in their natural state are characterized by low N supply (Tamm 1991; Jarvis and Linder 2000), but this changes where the deposition of N is high, i.e., near densely populated areas. Forests subject to high N load have been projected to go from a state of N limitation towards N saturation, a state at which the N supply is equal to, or exceeds, the N demand by plants and microbes (Aber et al. 1998). When this occurs, nitrogen is no longer effectively retained, the N cycle opens, notably as shown by increases in nitrification and denitrification and subsequent N losses through leaching of nitrate and gaseous losses of dinitrogen and nitrous oxide, a potent greenhouse gas (Aber et al. 1998). Increased nitrification and leaching of nitrate are associated with soil acidification (Aber et al. 1998). Moreover, nitrogen saturated forests are also associated with changes in species composition and tree health (Bobbink et al. 2010).
The rate at which previously N-saturated forests can return to their natural N-limited state is of considerable scientific and societal interest. However, the current biogeochemical model predictions on N limitation show high variability (Thomas et al. 2013). So called ‘roof-experiments’, in which plastic roofs have been installed under tree canopies in areas of high N deposition to prevent N deposition from reaching the soil, but soil is sprinkled with N-free ‘pre-industrial’ deposition, have shown that N leaching from forests can reduce from 70–90 kg N ha−1 year−1 to 5–15 kg N ha−1 year−1 in a few years (Boxman et al. 1998; Bredemeier et al. 1998). Large and rapid reductions in the leaching of NO3 −were also observed upon termination of an experimental N load of 90 kg N ha−1 year−1 for 20 years in a boreal pine forest (Johannisson et al. 1999). Other changes take more time (Bobbink et al. 2010). For example, almost 50 year after N additions, two species of mosses were found on N-fertilized plots, but not on control plots or plots fertilized with other elements (Strengbom et al. 2001).
Nitrogen saturation is defined as when the N supply exceeds the combined plant and microbial demand for N. Removal of this condition such that the forest becomes ‘un-saturated’ with N thus requires that the rate of external and internal ecosystem N supply to plants and microbes decreases relative to the supply of carbon through photosynthesis. This C flux is coupled to N primarily in foliage and in fine roots, mycorrhizal fungi and associated microorganisms. Importantly, the direct supply of plant-fixed C belowground and to the C-limited microorganisms varies temporally and spatially and is strongly dependent on the plant nutrient demand (Högberg et al. 2010; Nazir et al. 2010) in accordance with plant allocation theory (Cannell and Dewar 1994; Hermans et al. 2006). For better understanding of the mechanisms behind recovery i.e., resilience, it is thus of considerable interest to follow the temporal development of indicators of plant and microbial sink strength for N in forests, where the N supply has been reduced.
Many studies have recognized the soil C/N ratio as an indicator of the capacity of forest soils to retain N (Dise et al. 2009, and references therein). However, this ratio does not change quickly, and an increase would be too slow to capture the rapid decline in leaching of NO3 − in roof experiments, for example (Corre and Lamersdorf 2004). Since the abundance and physiologies of microorganisms are much more likely to respond dynamically, recent studies have examined the relations between leaching of NO3 −, and microbial community structure (Templer et al. 2008; Zechmeister-Boltenstern et al. 2011; Högberg et al. 2013) and ectomycorrhizal (ECM) fungi (Nilsson et al. 2007; Bahr et al. 2013; Högberg et al. 2013). There is also the suggestion that microbial synthesis and turnover of microbial products feeds the more slowly cycling soil organic C and N pools (Gleixner et al. 1999; Sollins et al. 2009; Knicker 2011; Schmidt et al. 2011; Cotrufo et al. 2013). Hence, studies of the microbial community structure, ECM fungi, and the physiologies of the organisms appear highly warranted in the context of forests recovering after N saturation.
Responses of soil microbiology and biochemistry, and N retention to the reduced N deposition have been reported mainly from roof-experiments (Enowashu et al. 2009; Kandeler et al. 2009; Dörr et al. 2012). Apart from these, few opportunities are available to analyse at the ecosystem scale the return to N limitation in forests subject to long-term high N loads. In particular, little attention has been paid to the N sink strength of different ecosystem components. In studies of the effect of termination of a high N load (90 kg N ha−1 year−1) in a long-term N-loading experiment we previously reported that N leaching decreased rapidly (Johannisson et al. 1999), that the N concentration of the foliage started to decline after 6 years, but that the rate of gross N mineralisation still remained high 14 years after termination of the high N load (Chen and Högberg 2006) when microbial community structure was little affected according to phospholipid fatty acid (PLFA) analysis (Högberg et al. 2007a, b). Also tree root uptake capacity for NH4 + showed little recovery 2 and 7 years after termination (Quist et al. 1999). Further detailed analysis of changes in the natural abundance of 15N in tree needles and in the soil profile actually revealed a decrease in the δ15N of tree foliage and a decrease in the δ15N down the soil profile indicating that the function of ECM symbiosis in tree N uptake was restored, i.e. began to be similar to that in the control plots, starting around 6 years after the high N treatment was terminated (Högberg et al. 2011). This proposition was further supported by a correlation between the δ15N of tree foliage and the PLFA 18:2ω6,9, a fungal biomarker (Högberg et al. 2011). PLFA 18:2ω6,9 in turn correlated with the percentage ECM sequences out of total fungal sequences, thus supporting earlier findings that this biomarker were indicative of ECM rather than saprobic fungi (Högberg et al. 2011). We have used this and other findings to provide an analysis of the impacts of declining N supply upon the N and C dynamics of forest soils, trees and their associated microbial symbionts and other soil microorganisms.
Here, we report on further changes of the N status in plants, soil, and the N cycle in this unique field experiment. We focus on potential changes in the microbial community composition, especially potential increases in ECM fungal abundances as the anticipated consequence of increased tree below-ground C allocation in response to decreasing N availability, and on indicators of the microbial and plant N sink strength. We also study the rates of gross N mineralisation as a measure of soil N turnover. Our null hypothesis is that the parameters and processes studied will remain altered in the terminated N treatment i.e. be en par with observations in the on-going N treatments. The null hypotheses will be rejected if data indicate no significant difference with the control plots, i.e., indicating that the studied system has returned to states characteristic of the N-limited boreal forests.
Material and methods
The experiment, the site and the forest
We studied the experiment E55, which is located in a boreal Pinus sylvestris. L. forest c. 100 km NW of Umeå in northern Sweden (64˚21′N, 19˚46′E, 260–275 m a.s.l.). The experiment was established by the late Carl Olof Tamm and his co-workers. Annual mean temperature is 1.2 °C, and the mean annual precipitation 595 mm. Background N deposition in the area is about 2 kg N ha−1 year−1. The soil is a glacial till with fine sand as the dominant fraction and the soil profile is classified as Haplic Podzol (FAO classification) or Typic Haplocryod (US Soil Taxonomy). The forest was established by planting 2-year-old seedlings in 1953, after a clear-felling of the old forest in 1951 and a prescribed burning in 1952. The experiment, which has been studied extensively for more than four decades, and the site, is described in greater detail by Tamm et al. (1999) and Högberg et al. (2006b; 2014a). We use data from the part of E55 involving additions of NH4NO3. Three 30 × 30 m plots per treatment were established and randomly distributed in three blocks. From 1971, N was added at four rates (Table 1). Treatments N1, N2, and N3 received on average 34, 68 and 108 kg N ha−1 year−1. The heaviest N load was terminated in 1990. Hence, we were able to study the recovery of the ecosystem from N saturation in comparison to a control (N0) and two on-going N treatments, N1, and N2. The latter was terminated in 2008, 1 or 2 years before the major measurements reported here, but this gave little time for recovery to occur after the very high N load of >60 kg N ha−1 year−1 over a period of 38 years. Selected characteristics of soil and microbial community composition in the different treatments are shown in Table 2.
Tree growth, foliar N concentration and δ 15N
Estimates of total stem wood production since the first fertilization are based on measurements in the inner 20 m × 20 m area of the plots, including breast-height diameters (1.3 m) for all trees and heights of sample trees. Secondary height functions per plot were used to estimate heights for unmeasured trees and stem wood volume was calculated by means of general volume functions for Pinus sylvestris in northern Sweden using diameter and height as independent variables (Näslund 1947; Brandel 1990). Data on total stem wood production included volumes of thinned and dead trees and represent the total stem wood production from 1971 to 2010 (Unit for Field-based Forest Research database, Swedish University of Agricultural Sciences). Samples of current-year needles were taken from the top whorls of 10 trees per plot every year between 1970 and 2011. Needles were oven-dried (70 °C, 48 h), and ground in a ball mill before analysed for N% and the natural abundance of 15N on an elemental analyser (Carlo Erba CHN 1110, Italy) coupled online to an isotope ratio mass spectrometer (Delta V, Thermo Fischer, USA). Small natural variations in the ratio 15N/14N are conventionally described in per mil (‰) deviations, δ, from the standard atmospheric N2, i.e. δ15Nsample = ((Rsample /Rstandard)—1) * 1000, where R is the ratio 15N/14N. We have previously reported data for foliar N% and δ15N up until 2006 (Högberg et al. 2011) and will here report data for that year and the following years up until 2011.
Root N uptake bioassays
Roots for an N uptake bioassay were sampled from four trees from each of the three plots per treatment on 15 June 2010. Roots were kept moist and stored in darkness at 4 °C overnight. The root N uptake bio-assay was used as an indicator of tree root N demand for NH4 + (Jones et al. 1991, 1994). The day after sampling, roots were washed under tap water and soaked in 0.5 mM CaCl2 solution for 30 min. The high affinity transport system of NH4 + into roots was tested in a solution with 20 atom % 15NH4Cl at a concentration of 70 μM (Kronzucker et al. 1995, 1996). Roots from the N0-N3 treatments were immersed in uptake solutions for 2 hours, one treatment at a time. Uptake of N was stopped by immersing roots in separate desorption solutions of 0.5 mM CaCl2. Roots were then dried for 36 h at 70 °C and ground in a ball mill and subsequently analysed for 15N on an elemental analyser (FlashEA 2000, Thermo Fisher Scientific) coupled online to an isotope ratio mass spectrometer (Delta V, Thermo Fisher Scientific, USA). Results of root bio-assays are highly variable, because the roots studied comprise a mix of roots of different branch orders and as a result samples vary considerably in the fraction of very fine and highly active roots (Rosengren et al. 2003). The roots also vary in terms of the associated ECM fungal species and the amount of ECM mycelium associated with the roots. For these reasons, and with the objective to compare our results from 2010 with identical bio-assays conducted in 1992 (Jones et al. 1994) and 1997 (Quist et al. 1999), we expressed our data on root N uptake in relation to those of tree roots from N0 plots (=100 %).
Soil sampling and pre-treatment
On 8 September 2009, the combined organic F+H horizon was sampled within the inner 20 × 20 m area of the plots by using a corer (10 cm diameter). Five bulk samples were collected by random at each plot (n = 5). Three plots were sampled per treatment and 12 plots were sampled in total, giving a total number of 60 samples. We kept samples in cooling boxes in the field and during transportation and at 4 ° C after arrival to the laboratory. We sieved the soil (4 mm mesh) and sorted the roots (>1 mm) by hand. Subsamples of soil were immediately frozen after preparation, cryo-milled, and stored at −20 °C for subsequent characterization of the microbial community composition. The soil pH, inorganic N, net N mineralisation, and gross N mineralisation were estimated on root free and homogenized soil the day after sampling in the field.
Soil pH, total C and N analysis
Soil pH was measured at a soil-to-water ratio of 1:3 (v/v). Organic matter (o.m.) content was estimated as % loss of dry matter after ignition (600 °C, 4 h) of dried soil (105 °C, 48 h). Soil was oven dried (105 °C for 48 h), ground in a ball mill before analysis of C, N, and 15N on an elemental analyser (Carlo Erba CHN 1110, Italy) coupled online to an isotope ratio mass spectrometer (Delta V, Thermo Fisher Scientific, USA).
Inorganic N pools, gross and net N mineralisation
We used the 15N pool dilution method for estimating the gross N mineralisation rate (Kirkham and Bartholomew 1954; Hart et al. 1994a) and followed the procedures described in Blaško et al. (2013). To avoid contamination by N with unknown 15N signature, we used acid washed equipment during analyses. Briefly, two subsamples of homogenized fresh soil equivalent to 25 g of dry weight were placed into separate plastic bags. Soils were labeled with in total 3.5 ml of 0.250 mM 15N-NH4Cl (99 atom %) distributed by six injections using a syringe. The 15N addition corresponded to 1 μg N per gram dry weight soil (Knowles and Blackburn 1993; Chen and Högberg 2006). Immediately after labeling (t0 sample), we extracted one of the two subsamples using 125 ml of 1 M KCl (Fractopur, Merck Germany) during 1 h of shaking (120 rotations per minute) followed by filtering the extracts by gravity (Munktell filter 00H). The same procedure was repeated with the second subsample after 24 h of incubation (t24 sample) at 17 °C. The filtrates were analyzed for NH4 + and NO3 − concentrations by flow injection analysis (FIAstar 5000, FOSS, Höganäs, Sweden). The micro diffusion technique was applied to transfer 14+15NH4 + from extracts to acidified paper disks (Stark and Hart 1996; Blaško et al. 2013). Paper disks were dried and placed into tin capsules and analyzed in an elemental analyzer (Carlo Erba CHN 1110) coupled online to an isotope ratio mass spectrometer (EA-IRMS, Delta V, Thermo Fisher Scientific). We followed the automatic blank correction method (Stark and Hart 1996), to correct for contributions by filter N to the sample 15N signature. Gross N mineralisation rates were calculated using the equations in Hart et al. (1994a,b).
Nitrogen retention
Nitrogen retention was calculated as the immobilized 15N at time zero, t0, in the 15N pool dilution studies (see methods for gross N mineralisation). The salt extractable or recovered 15N in the salt extractable NH4 + pool within 30 s from injection of tracer is: Recovery (%) = 15N (atom % in excess of natural abundance) * [NH4 +]t0 / 15N injected. We calculated the fraction of 15N retained, by subtracting the fraction of salt Extractable 15N from that of added: Retention (%) = 100 (%) – Recovery (%). Previous measures of CEC of the mor-layers (Aronsson et al. 1999), which was determined at pH 7.0 with ammonium acetate as extractant, and thus directly relevant for discussions about the retention of this N species in the soil, showed no significant differences between treatments (Two-way Anova, P = 0.306). The CECs were 492 ± 55 (mean ± 1.0 SE), 587 ± 69, 476 ± 40, and 507 ± 50 mmolc kg−1 dry soil, for the N0, N1, N2, and N3 treatments, respectively (Aronsson et al. 1999). Thus, we assumed abiotic retention of NH4 + to be similar among plots, and that the large variations in retention of NH4 + encountered reflected differences in biotic retention.
Soil microbial community structure
We analyzed PLFA biomarkers indicative of taxonomic groups of soil microorganisms (Tunlid and White 1992; Frostegård and Bååth 1996). Lipids were extracted from cryo-milled root-free soil equivalent to 0.15 g organic matter using the Bligh and Dyer (1959) extraction technique as modified by Frostegård et al. (1993). The lipid phase was fractionated on silica gel columns (Varian, HF Bond Elut LRC-Si, 100 mg, particle size 120 μm, Varian, Palo Alto, CA, USA) into neutral, glyco- and phospholipid-containing polar lipids. The phospholipid fraction was dried and stored at −20 °C under N2, and subjected to mild alkaline methanolysis. The derivatization procedures followed the mild alkaline methanolysis method (White et al. 1979). Methylnonadecanoate Me19:0 was added as an internal standard to each extract prior to derivatization of the PLFAs to their respective fatty acid methyl esters (FAMEs). Analysis by GC-FID. The FAME samples were analysed using a PDZ Europa Ltd., Northwich, UK. Each sample (volume of 1 μl) was injected onto a HP-5MS column (60 m length, 0.25 mm i.d., film thickness 0.32 mm; Agilent Technologies 19091S-416H, JandW Scientific, Santa Clara, CA, USA) via an injector held at 250 °C. The GC oven was initially held at 80 °C for 3 min, and then ramped at 20 °C min−1 to 170 °C following injection of the sample, then ramped at 2 °C min−1 to 210 °C and lastly ramped at 30 °C min−1 to a final temperature of 295 °C, where it was held for 10 min. The identity of individual PLFA peaks were determined by comparing retention times of authentic FAME standards (FAME 37-47885-U and P-BAME 24 47080-U, Supelco, Bellefonte, USA), and by comparing the retention times for individual PLFA peaks with peaks that had been identified by GC–MS (Jeol JMS-MStation GC/MS magnetic-sector mass spectrometer). Standard nomenclature was used to describe PLFA fatty acids: that is, by the total number of C atoms: number of double bonds, and ω followed by the position of the double bond from the methyl end of the molecule. The prefixes a, i, and cy, refer to anteiso, iso, cyclopropyl branching. 10Me indicates a methyl group on the 10th carbon from the carboxyl end of the molecule. The PLFA 18:2ω6,9 is the crown biomarker for fungi (Ratledge and Wilkinson 1988; Frostegård et al. 2011). Here, we use this PLFA as biomarker for ECM fungi, in particular. This was supported by reports from this and similar P sylvestris forests (i) of a strong relationship (R 2 adj = 0.9) between this biomarker and DNA-sequences of ECM fungi at this specific experiment (Högberg et al. 2011), (ii) of a significant relationship (R 2 adj = 0.9) between the 13C labelling of the biomarker in soil and ECM fungal sporocarps in a large-scale 13C tracer pulse-chase experiment in a nearby forest (Näsholm et al. 2013), (iii) of high sensitivity of this biomarker and ECM sequences to tree-girdling in another nearby forest (Yarwood et al. 2009), and of (iv) a dominance of DNA sequences of ECM fungi in F- and H-horizons of the mor layer (Lindahl et al. 2007; Clemmensen et al. 2013), i.e. the soil horizon sampled and studied here. The other putative fungal biomarker 18:1ω9 which does not correlate with the fungal biomarker ergosterol in this experiment (Högberg 2006) and does not respond to tree-girdling (Yarwood et al. 2009), is also indicative of bacteria (Frostegård et al. 2011) and is therefore not used as a fungal biomarker. Ten PLFAs were used as bacterial biomarkers: i15:0, a15:0, 15:0, i16:0, 16:1ω7, a17:0, cy17:0, 17:0, 18:1ω7, and cy19. The three most abundant terminally branched PLFAs (i.e., i15:0, i16:0, a17:0) were used as signature lipid biomarkers for gram-positive bacteria, while the three most abundant PLFAs characteristic of gram-negative bacteria were: 16:1ω7, 18:1ω7, and cy19:0. PLFAs 10Me17:0 and 10Me18:0 were used as biomarkers of actinobacteria. Indices used were ECM fungi/bacteria, saturated/monounsaturated (sat/mono), and cyclopropyl/precursors (cy/pre). Sat/mono ratio was represented by the sum of 14:0, 15:0, 16:0, 17:0, 18:0 divided by the sum of 16:1ω9, 16:1ω7, 16:1ω5, 18:1ω9, 18:1ω7. Cy/pre ratio was represented by the sum of cy17:0 and cy19:0 divided by the sum of the precursors 16:1ω7 and 18:1ω7.
Statistical analysis
In all statistical analysis, in tables and figures, we used plot means (N = 12 plots) of five bulk samples or treatment means (N = 3 plots for each treatment, one plot in each of three blocks) as specified in each case. Nitrogen treatments were assigned randomly to plots within the block, so each block contained control (N0), N1, N2 and N3 treatments. Effects of treatments and blocks on variables were tested by two-way analysis of variance (ANOVA). When a treatment effect was significant (P < 0.05), Holm-Sidak test for pairwise comparison followed the ANOVA to identify which treatments differed. We used plot mean values and the Pearson product moment correlation method for testing the strength of relationships between variables (N = 12 plots). Analyses of environmental and microbial community composition data were performed in SigmaStat statistical software (v4.0, Systat Software, Inc. CA, USA) and PC-ORD Multivariate Analysis of Ecological Data (version 6.0, MjM software, Gleneden Beach, OR, USA), respectively. Twenty-five PLFA biomarkers reflecting the microbial community composition were examined. We used (i) proportion data (mol % out of total abundance of PLFAs) for describing microbial community composition, (ii) moles PLFA normalized to amount organic matter (per gram organic matter) as a relative quantification of biomarkers, and (iii) moles PLFA per unit area (m2) as a quantitative estimate at an ecosystem level to describe the microbial communities. In all subsequent analyses we used fatty acids that were identifiable and present at 0.5 mol% or higher. Mole percent values sum to a constant value (100 %), and thus are not normally distributed. The proportion data were arcsine square root transformed before analysis as recommended (Zar 1984) to improve multivariate normality (McCune and Grace 2002; Balser and Firestone 2005). Non-metric Multidimensional Scaling (NMS) ordination ended up in linear relationships between the ordination scores (individual variables) and axes, and Pearson product moment correlations were used to examine these (McCune and Grace 2002). The structural similarity of PLFA profiles was examined and major groups of microbial communities were identified by hierarchal cluster analysis using non-transformed data (McCune and Grace 2002). The Euclidean distance and Ward’s linkage method was used, because it is space conserving (McCune and Grace 2002). The analysis was followed by multi-response permutation procedures (MRPP) using non-transformed data (McCune and Grace 2002). The same distance was used (Bray-Curtis) in NMS and MRPP ensuring agreement between these two analyses. The resulting test statistics, T and A, describes the separation between the groups and within-group homogeneity, respectively. The more negative T is, the stronger the separation. An A-statistic equal to 1, indicates that all items in the group are identical; when heterogeneity within groups equals expectation by chance A = 0. An A-value of 0.3 is considered high for environmental data (McCune and Grace 2002). A Mantel test was used to compare the microbial community composition in 2009 with that observed in August 2004 (Högberg et al. 2007b) by creating distance matrices and comparing sample distances for microbial communities (McCune and Grace 2002). Significance level was set at α = 0.05. Data are presented as mean ±1 SE.
Results
Tree growth and foliar analysis
Tree stem volume of plots was assessed in 1971, 1974, 1978, 1984, 1989, 1998, and 2010 (Fig. 1). Clearly, the treatments N1 and N2 have increased growth in the longer term, while N3 resulted in slightly lower growth (Fig. 1) than in the control after an initial positive response, of 40 % greater growth than the control (Högberg et al. 2006b). Needle concentration of N in the terminated N3 treatment was higher than that of the control, N0, but did not differ from that of N1, but was lower than that of the N2 treatment through the period 2006–2011 (Fig. 2a). The same was true for the δ15N of needles, but in this case the values for N3 were also lower than those for N1 plots at the end of the period of observation (Fig. 2b).
Tree stem volume growth in the long-term N addition experiment E55 at Norrliden, Sweden (c.f. Table 1 ). N3 and N2 treatments were terminated in 1990 and 2008, respectively. Total N loads were 1350, 2520, and 2160 kg ha−1 for N1, N2, and N3, respectively. N = 3 plots per treatment
Root N uptake capacity
Mean rates of N uptake were highest in roots from the control plots (Fig. 3), while we found the lowest N uptake in roots from the N2 plots. Nitrogen uptake in roots from the N3 plots tended to be higher, but not significantly so, than in N1 and N2 (Fig. 3), and did not differ significantly from in N0. In comparison to previous studies from 1992 and 1997 the root N uptake rates in 2010 (expressed as percentage of the control, N0), were similar for N1 and N2, but increased for N3, in which case as mentioned, the uptake rates for N3 in 2010 did not differ from those in the control (P = 0.349) as they did in 1992 and 1997 (Fig. 3).
Ectomycorrhizal root N uptake in years 1992, 1997, and 2010 in the long-term N addition experiment E55, Norrliden, Sweden. Root N uptake in N1-N3 is expressed as a percentage of the rates in N0 (control) measured in 1992 (Jones et al. 1994), 1997 (Quist et al. 1999) and 2010. Absolute root N uptake rates in N0 were 45, 27, and 112 mg kg−1 2 h−1 in 1992, 1997, and 2010, respectively. Treatments sharing the same letter are not significantly different. Means ±1 SE, N = 3 plots per treatment
Gross N mineralisation and retention of NH4 +-N in soil
Low gross N mineralisation as expressed per gram organic matter and day were found in the control plots and in the N3 plots, which differed significantly from the higher rates in the N1 plots (Table 3). When the gross N mineralisation was expressed per m2 and day, thus taking into account the larger amount of organic matter per m2 in N1-N3 plots, only N1 plots differed from the control plots (Table 3). The percentage NH4 +-N retained by the soil matrix including its organisms was very high in control and N3 plots, which differed significantly from both N1 and N2 plots (Table 3).
Soil microbial community structure
Long-term N loading caused a reduction by up to 44 % (P = 0.006) in total PLFAs when expressed per gram organic matter, but not when calculated per m2 (P = 0.619) (Fig. 4a, b). A recovery of the microbial biomarkers in terminated N3 plots was evident in the sense that N0 and N3 were not significantly (P = 0.504 and 0.172) different (Fig. 4a, b). In contrast to estimates per g organic matter (Fig. 4c), the quantity of ECM fungal biomarkers per unit area was the same in N3 and N0 but bacterial markers were more common in N3 than N0 (Fig. 4d). The proportion of gram-positive and actinobacterial biomarkers out of total PLFAs was higher in N1-N3 and they were almost twice as abundant per m2 in N3 plots (Table 2). The amounts of ECM fungal, but not bacterial, biomarkers per unit area correlated with δ15N in foliage (R = −0.704, P < 0.01) and N retention (R = 0.938, P < 0.001). The microbial indices ECM fungi/bacteria ratio and gram-positive/gram-negative ratio showed a declining pattern with on-going N-fertilization (P = 0.002 in both cases) and were lowest in N1 and N2 plots as compared to the control plots. The ECM fungi/bacteria ratio was 0.42 in the control, declined to 0.20, 0.15, and 0.19 in N1-N3, respectively, and was at most 63 % lower (in N2) compared with the control. The ECM fungi/bacteria ratio in N3 was lower than expected from the increase in ECM fungi because of the higher abundance of bacteria (Table 2). Abundance of gram-positive biomarkers increased more than the gram-negative ones across the N gradient, thus the gram-negative/gram-positive ratios declined (P = 0.002) from 2.42 ± 0.03 in control plots to 2.06 ± 0.05, 1.85 ± 0.06, and 2.01 ± 0.05 in N1, N2, and N3, respectively. We did not observe any linkages between microbial community composition and soil pH within the narrow range of 0.4 units among plots. The fraction of retained 15N tracer was highly linked to the microbial community composition as indicated by the correlation to sat/mono (R = −0.92, P < 0.001), cy/pre (R = −0.92, P < 0.001), ECM fungal abundance (R = 0.837, P < 0.001), bacterial abundance (R = 0.817, P = 0.001), and ECM fungi/bacteria ratios (R = 0.68, P < 0.05) (Fig. 5a-e). The NMS ordination of PLFA profiles (not shown) resulted in a two-dimensional solution describing 99 % of the variation, where the first axis described 95 % and the second axis described 4 %. Final stress of the ordination was low (0.9 after 34 iterations). The microbial community composition was linked to N retention (R = −0.82), soil C/N ratio (R = −0.92), needle and soil N percentages (R = 0.87 and 0.85) all four variables P < 0.001, soil inorganic N concentration (R = 0.58, P < 0.05), and gross N mineralisation rate per unit area (R = 0.65, P < 0.05). We identified distinct microbial communities in the N0, N1, N2, and N3, respectively. This was evident from the observation of no overlap between treatments in NMS, MRPP (general model, T = −5.4, A = 0.6, P < 0.001), and cluster analysis (Fig. S1a). After MRPP and pairwise comparison among treatments T varied between −2.6 and −3.0, A between 0.28 and 0.66, and P between 0.021 and 0.025. Mantel analysis showed a clear relationship between microbial communities from 2004 and 2009 (R = 0.828, P < 0.0001). The cluster analysis including both years PLFA profiles data showed that microbial community composition in N addition and terminated plots were unstable as indicated by the high similarity between years among control plots (microbial community data from 2004 and 2009 merged into the same group), whereas microbial communities in N1-N3 from the different years mainly clustered into separate groups (Fig. S1b).
Soil microbial community characteristics based on phospholipid fatty acid (PLFA) biomarkers in the long-term N addition experiment E55 at Norrliden, Sweden. a Total abundance of PLFA per gram organic matter. b Abundance of major taxonomic groups per gram organic matter. c Total abundance of PLFAs per unit area. d Abundance of major taxonomic groups per unit area. Means ±1 SE, N = 3 plots per treatment
Relationships between soil N retention and microbial biomarkers (phospholipid fatty acids) in Experiment E55, Norrliden, Sweden. a Saturated/monounsaturated ratio. b Cyclopropyl/precursors ratio. c ECM fungal biomarker. d Bacterial biomarkers. e ECM-fungi/ bacteria ratio. Points indicate the average of five replicates from each plot
Discussion
The progression from N-limitation to N-saturation of forests is relatively well described and understood (Aber et al. 1998; Magill et al. 2004; Bobbink et al. 2010). Much less is known concerning the rate(s) at which forests can return to N-limitation, and which the key processes that cause N-limitation are (Thomas et al. 2013). Nitrogen saturation occurs when the N supply exceeds the C supply from photosynthesis, which is also constrained by the supply of other nutrients (e.g., Tamm 1991). Recovery from this condition and transition to the natural state of N-limitation requires that the N supply diminishes in relation to the C supply from photosynthesis, such that it does no longer meet the combined N demand of plants and microbes.
Roofing of plots in areas of high N deposition has shown that the leaching of NO3 − declines very rapidly (Boxman et al. 1998; Bredemeier et al. 1998). Likewise, termination of N loading in the experiment studied here resulted in a very low rate of leaching of NO3 − a few years after the treatment was stopped (Johannisson et al. 1999). These examples show that removal of the external N supply can rapidly lead to the loss of a key symptom of N saturation. However, the N availability in such systems may apparently remain high for many years, although the concentration of the amino acid arginine, which is accumulating in N-saturated conifer trees (van Dijk and Roelofs 1988; Näsholm and Ericsson 1990), decreases within the first 5 years. The data presented here show that the N concentration of the pine needles is still significantly higher in the N3 plots than in control plots (Fig. 2a), which shows that the soil N supply is still greater in the N3 plots despite two decades of recovery.
A progressive decline in the C/N ratio of the litter-fall would allow more N to be bound to soil organic matter. However, this ratio changes very slowly; in 1988, 2 years before the termination of the N3 treatment, the C/N ratio of the mor-layer was 30.0 ± 0.2 (Aronsson et al. 1999), which compares with 25.9 ± 0.6 in 2000 (Högberg et al. 2006b) and 27.9 ± 0.6 in 2009 (Table 2), i.e. after 10 and 19 years of recovery (Quist et al. 1999). The data indicate that it might be difficult to determine this ratio exactly, e.g., because of difficulties to precisely define the horizon sampled, but also that the ratio changes slowly.
For this latter reason we postulated that it is more interesting to look at changes in soil organism communities and their physiology, which are likely to respond more quickly because of their short turn-over times (Högberg et al. 2008). As stated above there are no significant differences in CEC across the treatments (Aronsson et al. 1999), which means that the large differences in N retention (cf. N immobilization) seen here (Table 3) are most likely related to differences in the composition of the microbial community and changes in microbial physiology. Support of this proposition comes from the strong correlations between N retention, quantities of fungal and bacterial biomarkers, and microbial community composition indices e.g., the ECM fungi/bact, sat/mono, and cy/pre (Fig. 5). Our data showed as expected greater N immobilization capacity when ECM fungi were more abundant (Alberton and Kuyper 2009; Näsholm et al. 2013). Interestingly, not only the ECM fungi but also bacteria were stimulated in N3 plots (Fig. 4). The likely explanation is two-fold. Firstly, the indications of stronger C limitation among bacteria in N loaded than in control plots (Demoling et al. 2008; Kamble et al. 2013), suggests that enhanced belowground C supply after termination of N load could stimulate growth and biomass. Secondly, enhanced N availability should favor bacteria (over ECM fungi) under C-limitation and under reduced tree below-ground C allocation because of the relatively higher bacterial N demand per biomass C. Bacteria may thus support their biomass with less C per N assimilated under C-limitation than ECM fungi because of fundamental differences in their physiology and biomass C/N ratio (Keiblinger et al. 2010; Franklin et al. 2011). Moreover, our observation of higher sat/mono and cy/pre ratios in N1-N2 than in N0 and N3 plots, have been associated with low C and/or high N availabilities (Bossio and Scow 1998; Allison et al. 2005; van Diepen et al. 2010; Zechmeister-Boltenstern et al. 2011; Högberg et al. 2013) thus supporting the explanation above. However, it is not possible to differentiate between shifts in microbial community composition and resource availability. As regards the fungi/bacteria ratio, Zechmeister-Boltenstern et al. (2011) found that it correlated negatively to N deposition rate and NO3 − leaching. Using the same fungal biomarker as in this study the fungi/bacteria ratio also correlates with variations in N mineralisation in the landscape (Binkley and Menyailo 2005; Högberg et al. 2006a). We have shown that this reflects a greater N immobilization capacity at higher fungi/bacterial ratios among spruce forest stands, which we proposed reflected the higher relative abundance of ECM fungi and hence a higher direct plant C flux to the C-limited biota in soil (Högberg et al. 2013).
Tree below-ground C flux to roots and their ECM fungi is highly responsive to changes in N supply (Högberg et al. 2010), and this causes shifts in the fraction of N retained by the ECM soil mycelium as compared to the fraction transferred to the trees (Näsholm et al. 2013). In fact, Näsholm et al. (2013) showed that the ratio of the amount N transferred to the host trees over the amount C invested in the fungal symbiont is not constant, but changes non-linearly such that at low soil N availability very little N can be transferred to the host because the fungus must first satisfy its own demand for N. This would further enhance the N retaining capacity of already N-limited systems with ECM trees leading to an aggravation rather than alleviation of plant N limitation. Högberg et al. (2011) proposed that changes in the below-ground C flux to ECM fungi play a key role in this context: at low N supply this flux is large and promotes vigorous growth of ECM mycelium with a high capacity for N uptake and retention, while at a high N supply this C flux is small, which removes a large N sink from the soil system and opens the N cycle. Indeed, when expressed in mmol m−2, our data on the fungal PLFA 18:2ω6,9 biomarker showed ca. 50 % reduction in N1 and N2 plots as compared to control plots, but that there was no statistically significant difference between control and N3 plots (Fig. 4). This is most probably caused by increased mycorrhizal mycelial growth in response to greater tree below-ground C allocation in the more N-limited system. When fungal abundance was analyzed in 2004, i.e. 5 years before this study, the N3 plots were approaching the levels found in control plots (per gram organic matter) but did not yet differ significantly from the N1 and N2 plots although the levels of extractable inorganic N were clearly lower in N3 (Högberg et al. 2007b). However, after recalculation of fungal abundance to per unit area using 2009 data on organic matter content and assuming no major shifts in organic matter content between years, N3 differed from N1 and N2 (P < 0.001) .
The relation between retention of N in the ECM mycelium and the N transported to the tree foliage can also be inferred based on variations in δ15N in tree foliage, because the N in ECM fungal mycelium becomes enriched in 15N relative to the N transferred to the tree hosts (Hobbie and Högberg 2012). Previously we have demonstrated that a decrease in the δ15N in the tree foliage was indicative of a recovery of the role of ECM symbiosis in N uptake in N3 that started about 6 years after the termination of the high N-loading treatment (Högberg et al. 2011). In the same study, we demonstrated that the δ15N of tree foliage correlated with the PLFA 18:2ω6,9, which in turn correlated with ECM fungal sequences (Högberg et al. 2011) and we observed a clear decrease in ECM species in N2 and a recovery of species in the N3 treatment (Högberg et al. 2014b). We note that the trend of the δ15N of tree foliage in N3 to become similar to that of the control continues (Fig. 2). Similarly, there is a tendency of larger difference in δ15N between foliage and soil (Table 2) (Garten and Miegroet 1994), which further supports our suggestion that the importance of the ECM mycelium is increasing.
The data from the root N uptake bioassays showed that the difference between the recovering N3 plots and the control plots is no longer significant, which it was in 1992 and 1997 (Fig. 3). We would like to stress that these fine roots are mycorrhizal, and that the increased uptake capacity is another piece of evidence pointing at the importance of the tree below-ground C flux in regulating the soil N cycle.
We used measurements of gross N mineralisation as an indicator of the rate of soil N cycling and N availability. We found that the rate of this process was now similar in the N3 and the control plots when the rate is expressed per g soil organic matter and day, but still greater in N3 when the activity is expressed per m2 and day (Table 3), because of the greater amount of organic matter in N3 plots. Across all treatments there was a negative correlation between the ratio ECM fungi/bacteria and areal gross N mineralisation. Fungal and bacterial biomarkers were positively correlated to N retention but negatively so to the gross N mineralisation rates and amounts of extractable inorganic N, which further supports the notion of the microbial component as an important regulator of the soil N cycle.
Conclusions
Although the N availability is still elevated (as indicated by the concentration of N in tree foliage) in previously N loaded plots 20 years after N-loading ceased, there is now clear evidence that the N sink strength of the soil microbial community and the trees is increasing. Our observations heightened the importance of soil microorganisms in this context, and we propose that future studies of the interactions between N supply and N loss should focus more explicitly on the role of tree below-ground C allocation to roots and their associated microorganisms, notably their mycorrhizal symbionts, but attention should also be paid to the associated bacteria. Our proposition of the importance of biotic N retention is supported by strong evidence that the majority of soil N is organically bound in peptide-like compounds likely of microbial origin (Morier et al. 2008; Knicker 2011).
References
Aber J, McDowell W, Nadelhoffer K, Magill A, Berntson G, Kamakea M, McNulty S, Currie W, Rustad L, Fernandez I (1998) Nitrogen saturation in temperate forest ecosystems: hypotheses revisited. Bioscience 48:921–934. doi:10.2307/1313296
Alberton O, Kuyper TW (2009) Ectomycorrhizal fungi associated with Pinus sylvestris seedlings respond differently to increased carbon and nitrogen availability: implications for ecosystem responses to global change. Glob Change Biol 15:166–175. doi:10.1111/j.1365-2486.2008.01714.x
Allison VJ, Miller RM, Jastrow JD, Matamala R, Zak DR (2005) Changes in soil microbial community structure in a tallgrass prairie chronosequence. Soil Sci Soc Am J 69:1412–1421. doi:10.2136/sssaj2004.0252
Aronsson A, Flower-Ellis JGK, Popovic B, Tamm CO (1999) Details of soil, stand and responses to fertiliser treatments on the pine (Pinus sylvestris L.) sites Norrliden (1971–1989) and Lisselbo (1969–1988). p 51 Swedish University of Agricultural Sciences, Uppsala,
Bahr A, Ellström M, Akselsson C, Ekblad A, Mikusinska A, Wallander H (2013) Growth of ectomycorrhizal fungal mycelium along a Norway spruce forest nitrogen deposition gradient and its effect on nitrogen leakage. Soil Biol Biochem 59: 38–48.doi: org/10.1016/j.soilbio.2013.01.004
Balser TC, Firestone MK (2005) Linking microbial community composition and soil processes in a California annual grassland and mixed-conifer forest. Biogeochemistry 73:395–415. doi:10.1007/s10533-004-0372-y
Binkley D, Menyailo O (2005) Gaining insights on the effects of tree species on soils. In: Binkley D, Menyailo O (eds) Tree species effects on soils: implications for global change. Springer, Dordrecht, pp 1–16
Blaško R, Högberg P, Bach LH, Högberg MN (2013) Relations among soil microbial community composition, nitrogen turnover, and tree growth in N-loaded and previously N-loaded boreal spruce forest. For Ecol Manag 302:319–328. doi:10.1016/j.foreco.2013.02.035
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Bobbink R, Hicks K, Galloway J, Spranger T, Alkemade R, Ashmore M, Bustamante M, Cinderby S, Davidson E, Dentener F, Emmett B, Erisman JW, Fenn M, Gilliam F, Nordin A, Pardo L, De Vries W (2010) Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecol Appl 20:30–59. doi:10.1890/08-1140.1
Bossio DA, Scow KM (1998) Impacts of carbon and flooding on soil microbial communities: phospholipid fatty acid profiles and substrate utilization patterns. Microb Ecol 35:265–278. doi:10.1007/s002489900082
Boxman AW, van der Ven PJM, Roelofs JGM (1998) Ecosystem recovery after a decrease in nitrogen input to a Scots pine stand at Ysselsteyn, the Netherlands. For Ecol Manag 101:65–79. doi:10.1016/S0378-1127(97)00126-6
Brandel G (1990) Volume functions for individual trees; Scot pine (Pinus sylvestris), Norway spruce (Picea abies), and birch (Betula pendula and Betula pubescens). Summary including functions in English. Swedish University of Agricultural Sciences, Uppsala, Department of Forest production
Bredemeier M, Blanck K, Xu YJ, Tietema A, Boxman AW, Emmett B, Moldan F, Gundersen P, Schleppi P, Wright RF (1998) Input–output budgets at the NITREX sites. For Ecol Manag 101:57–64. doi:10.1016/S0378-1127(97)00125-4
Cannell MGR, Dewar RC (1994) Carbon allocation in trees - a review of concepts for modelling. Adv Ecosyst Res 25:59–104. doi:10.1016/S0065-2504(08)60213-5
Clemmensen K, Bahr A, Ovaskainen O, Dahlberg A, Ekblad A, Wallander H, Stenlid J, Finlay R, Wardle D, Lindahl B (2013) Roots and associated fungi drive long-term carbon sequestration in boreal forest. Science 339:1615–1618. doi:10.1126/science.1231923
Chen Y, Högberg P (2006) Gross nitrogen mineralisation rates still high 14 years after suspension of N input to a N-saturated forest. Soil Biol Biochem 38:2001–2003. doi:10.1016/j.soilbio.2005.11.026
Corre MD, Lamersdorf NP (2004) Reversal of nitrogen saturation after long-term deposition reduction: impact on sol nitrogen cycling. Ecology 85:3090–3104. doi:10.1890/03-0423
Cotrufo M, Wallenstein M, Boot C, Denef K, Paul E (2013) The Microbial Efficiency-Matrix Stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: do labile plant inputs form stable soil organic matter? Glob Chang Biol 19:988–995. doi:10.1111/gcb.12113
Demoling F, Ola Nilsson L, Bååth E (2008) Bacterial and fungal response to nitrogen fertilization in three coniferous forest soils. Soil Biol Biochem 40:370–379. doi:10.1016/j.soilbio.2007.08.019
Dise NB, Rothwell JJ, Gauci V, van der Salm C, de Vries W (2009) Predicting dissolved inorganic nitrogen leaching in European forests using two independent databases. Sci Total Environ 407:1798–1808. doi:10.1016/j.scitotenv.2008.11.003
Dörr N, Kaiser K, Sauheitl L, Lamersdorf N, Stange CF, Guggenberger G (2012) Fate of ammonium 15N in a Norway spruce forest under long-term reduction in atmospheric N deposition. Biogeochemistry 107:409–422. doi:10.1007/s10533-010-9561-z
Enowashu E, Poll C, Lamersdorf N, Kandeler E (2009) Microbial biomass and enzyme activities under reduced nitrogen deposition in a spruce forest soil. Appl Soil Ecol 43:11–21. doi:10.1016/j.apsoil.2009.05.003
Franklin O, Hall EK, Kaiser C, Battin TJ, Richter A (2011) Optimization of biomass composition explains microbial growth-stoichiometry relationships. Am Nat 177:E29–E42. doi:10.1086/657684
Frostegård A, Bååth E (1996) The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol Fertil Soils 22:59–65. doi:10.1007/BF00384433
Frostegård A, Bååth E, Tunlid A (1993) Shifts in the structure of soil microbial communities in limed forests as revealed by phospholipid fatty acid analysis. Soil Biol Biochem 25:723–730. doi:10.1016/0038-0717(93)90113-P
Frostegård Å, Tunlid A, Bååth E (2011) Use and misuse of PLFA measurements in soils. Soil Biol Biochem 43:1621–1625. doi:10.1016/j.soilbio.2010.11.021
Garten CT, Miegroet HV (1994) Relationships between soil nitrogen dynamics and natural 15N abundance in plant foliage from Great Smoky Mountains National Park. Can J For Res 24:1636–1645. doi:10.1139/x94-212
Gleixner G, Bol R, Balesdent J (1999) Molecular insight into soil carbon turnover. Rapid Commun Mass Spectrom 13:1278–1283. doi:10.1002/(SICI)1097-0231(19990715)13:13<1278::AID-RCM649>3.0.CO;2-N
Hart SC, Nason GE, Myrold DD, Perry DA (1994a) Dynamics of gross nitrogen transformations in an old-growth forest: the carbon connection. Ecology 75:880–891. doi:10.2307/1939413
Hart SC, Stark JM, Davidson EA, Firestone MK (eds) (1994b) Nitrogen mineralization, immobilization and nitrification. Soil Sci Soc Am, Madison
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass allocation? Trends Plant Sci 11:610. doi:10.1016/j.tplants.2006.10.007
Hobbie E, Högberg P (2012) Nitrogen isotopes link mycorrhizal fungi and plants to nitrogen dynamics. New Phytol 196:367–382. doi:10.1111/j.1469-8137.2012.04300.x
Högberg MN (2006) Discrepancies between ergosterol and the phospholipid fatty acid 18:2ω6,9 as biomarkers for fungi in boreal forest soils. Soil Biol Biochem 38:3431–3435. doi:10.1016/j.soilbio.2006.06.002
Högberg MN, Briones MJI, Keel SG, Metcalfe DB, Campbell C, Midwood AJ, Thornton B, Hurry V, Linder S, Näsholm T, Högberg P (2010) Quantification of effects of season and nitrogen supply on tree below-ground carbon transfer to ectomycorrhizal fungi and other soil organisms in a boreal pine forest. New Phytol 187:485–493. doi:10.1111/j.1469-8137.2010.03274.x
Högberg MN, Chen Y, Högberg P (2007a) Gross nitrogen mineralisation and fungi-to-bacteria ratios are negatively correlated in boreal forests. Biol Fertil Soils 44:363–366. doi:10.1007/s00374-007-0215-9
Högberg MN, Högberg P, Myrold DD (2007b) Is microbial community composition in boreal forest soils determined by pH, C-to-N ratio, the trees, or all three? Oecologia 150:590–601. doi:10.1007/s00442-006-0562-5
Högberg MN, Högbom L, Kleja D (2013) Soil microbial community indices as predictors of soil solution chemistry and N leaching in Picea abies (L.) Karst. forests in S. Sweden. Plant Soil 372:507–522. doi:10.1007/s11104-013-1742-9
Högberg MN, Myrold DD, Giesler R, Högberg P (2006a) Contrasting patterns of soil N-cycling in model ecosystems of Fennoscandian boreal forests. Oecologia 147:96–107. doi:10.1046/j.1469-8137.2003.00867.x
Högberg MN, Yarwood SA, Myrold DD (2014a) Fungal but not bacterial communities recover after termination of decadal nitrogen additions to boreal forest. Soil Biol Biochem 72:35–43. doi:10.1016/j.soilbio.2014.01.014
Högberg P, Fan H, Quist ME, Binkley D, Tamm C-O (2006b) Tree growth and soil acidification in response to 30 years of experimental nitrogen loading on boreal forest. Glob Chang Biol 12:489–499. doi:10.1111/j.1365-2486.2006.01102.x
Högberg P, Högberg MN, Göttlicher SG, Betson NR, Keel SG, Metcalfe DB, Campbell C, Schindlbacher A, Hurry V, Lundmark T, Linder S, Näsholm T (2008) High temporal resolution tracing of photosynthate carbon from the tree canopy to forest soil microorganisms. New Phytol 177:220–228. doi:10.1111/j.1469-8137.2007.02238.x
Högberg P, Johannisson C, Högberg MN (2014b) Is the high 15N natural abundance of trees in N-loaded forests caused by an internal ecosystem N isotope redistribution or a change in the ecosystem N isotope mass balance? Biogeochemistry. doi:10.1007/s10533-013-9873-x
Högberg P, Johannisson C, Yarwood S, Callesen I, Näsholm T, Myrold DD, Högberg MN (2011) Recovery of ectomycorrhiza after ‘nitrogen saturation’ of a conifer forest. New Phytol 189:515–525. doi:10.1111/j.1469-8137.2010.03485.x
Jarvis P, Linder S (2000) Constraints to growth of boreal forests. Nature 405:904–905. doi:10.1038/35016154
Johannisson C, Myrold DD, Högberg P (1999) Retention of nitrogen by a nitrogen-loaded Scotch pine forest. Soil Sci Soci Am J 63:383–389. doi:10.2136/sssaj1999.03615995006300020017x
Jones HE, Högberg P, Ohlsson H (1994) Nutritional assessment of a forest fertilisation experiment in northern Sweden by root bioassays. For Ecol Manag 64:59–69
Jones HE, Quarmby C, Harrison AF (1991) A root bioassay test for nitrogen deficiency in forest trees. For Ecol Manag 42:267–282. doi:10.1016/0378-1127(91)90029-U
Kamble PN, Rousk J, Frey SD, Bååth E (2013) Bacterial growth and growth-limiting nutrients following chronic nitrogen additions to a hardwood forest soil. Soil Biol Biochem 59:32–37. doi:10.1016/j.soilbio.2012.12.017
Kandeler E, Brune T, Enowashu E, Dörr N, Guggenberger G, Lamersdorf N, Philippot L (2009) Response of total and nitrate-dissimilating bacteria to reduced N deposition in a spruce forest soil profile. FEMS Microbiol Ecol 67:444–454. doi:10.1111/j.1574-6941.2008.00632.x
Keiblinger KM, Hall EK, Wanek W, Szukics U, Hämmerle I, Ellersdorfer G, Böck S, Strauss J, Sterflinger K, Richter A, Zechmeister-Boltenstern S (2010) The effect of resource quantity and resource stoichiometry on microbial carbon-use-efficiency. FEMS Microbiol Ecol 73:430–440. doi:10.1111/j.1574-6941.2010.00912.x
Kirkham D, Bartholomew WV (1954) Equations for following nutrient transformations in soil, utilizing tracer data. Proc Soil Sci Soc Am 18:33–34. doi:10.2136/sssaj1954.03615995001800010009x
Knicker H (2011) Soil organic N - an under-rated player for C sequestration in soils? Soil Biol Biochem 43:1118–1129. doi:10.1016/j.soilbio.2011.02.020
Knowles R, Blackburn TH (eds) (1993) Nitrogen isotope techniques. Academic, San Diego
Kronzucker H, Siddiqi M, Glass ADM (1995) Analysis of 15NH4 + efflux in spruce roots (a test case for phase identification in compartmental analysis). Plant Physiol 109:481–490. doi:10.1104/pp. 109.2.481
Kronzucker H, Siddiqi M, Glass ADM (1996) Kinetics of NH4 + Influx in Spruce. Plant Physiol 110:773–779. doi:10.1104/pp. 110.3.773
Lajtha K, Jones J (2013) Trends in cation, nitrogen, sulphate and hydrogen ion concentrations in precipitation in the United States and Europe from 1978 to 2010: a new look at an old problem. Biogeochemistry 116:303–334. doi:10.1007/s10533-013-9860-2
Lindahl BD, Ihrmark K, Boberg J, Trumbore SE, Högberg P, Stenlid J, Finlay RD (2007) Spatial separation of litter decomposition and mycorrhizal nitrogen uptake in a boreal forest. New Phytol 173:611–620
Magill AH, Aber JD, Currie WS, Nadelhoffer KJ, Martin ME, McDowell WH, Melillo JM, Steudler P (2004) Ecosystem response to 15 years of chronic nitrogen additions at the Harvard Forest LTER, Massachusetts, USA. For Ecol Manag 196:7–28. doi:10.1016/j.foreco.2004.03.033
McCune B, Grace JB (2002) Analysis of ecological communities MjM Software Design, Gleneden Beach
Morier I, Schleppi P, Siegwolf R, Knicker H, Guenat C (2008) 15N immobilization in forest soil: a sterilization experiment coupled with 15CPMAS NMR spectroscopy. Euro J Soil Sci 59:467–475. doi:10.1111/j.1365-2389.2007.00998.x
Nazir R, Warmink JA, Boersma H, Van Elsas JD (2010) Mechanisms that promote bacterial fitness in fungal-affected soil microhabitats. FEMS Microbiol Ecol 71:169–185. doi:10.1111/j.1574-6941.2009.00807.x
Nilsson L, Bååth E, Falkengren-Grerup U, Wallander H (2007) Growth of ectomycorrhizal mycelia and composition of soil microbial communities in oak forest soils along a nitrogen deposition gradient. Oecologia 153:375. doi:10.1007/s00442-007-0735-x
Näsholm T, Ericsson A (1990) Seasonal changes in amino acids, protein and total nitrogen in needles of fertilized Scots pine trees. Tree Physiol 6:267–281. doi:10.1093/treephys/6.3.267
Näsholm T, Högberg P, Franklin O, Metcalfe D, Keel SG, Campbell C, Hurry V, Linder S, Högberg MN (2013) Are ectomycorrhizal fungi alleviating or aggravating nitrogen limitation of tree growth in boreal forests? New Phytol 198:214–221. doi:10.1111/nph.12139
Näslund M (1947) Functions and tables for computing the cubic volume of standing trees. Pine, spruce, and birch in southern Sweden, and in the whole of Sweden. Summary including functions in English. Meddelande från Statens Skogsforskningsinstitut, Swedish University of Agricultural Sciences, Stockholm,
Quist ME, Näsholm T, Lindeberg J, Johannisson C, Högbom L, Högberg P (1999) Responses of a nitrogen-saturated forest to a sharp decrease in nitrogen input. J Environ Qual 28:1970–1977. doi:10.2134/jeq1999.00472425002800060037x
Ratledge C, Wilkinson SG (1988) Microbial lipids. Academic, London
Rosengren U, Sleep D, Jones HE, Thelin G (2003) Increasing the sensitivity of the 15N root bioassay technique: suggested procedures. Commun Soil Sci Plant Anal 34:2363–2373. doi:10.1081/css-120024070
Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kögel-Knabner I, Lehmann J, Manning DAC, Nannipieri P, Rasse DP, Weiner S, Trumbore SE (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:49–56. doi:10.1038/nature10386
Sollins P, Kramer M, Swanston C, Lajtha K, Filley T, Aufdenkampe A, Wagai R, Bowden R (2009) Sequential density fractionation across soils of contrasting mineralogy: evidence for both microbial- and mineral-controlled soil organic matter stabilization. Biogeochemistry 96:209. doi:10.1007/s10533-009-9359-z
Stark JM, Hart SC (1996) Diffusion technique for preparing salt solutions, Kjeldahl digests, and persulfate digests for nitrogen-15 analysis. Soil Sci Soc Am J 60:1846–1855. doi:10.2136/sssaj1996.03615995006000060033x
Strengbom J, Nordin A, Näsholm T, Ericson L (2001) Slow recovery of boreal forest ecosystem following decreased nitrogen input. Funct Ecol 15:451–457. doi:10.1046/j.0269-8463.2001.00538.x
Tamm C-O (1991) Nitrogen in Terrestrial Ecosystems Springer-Verlag, Berlin
Tamm C-O, Aronsson A, Popovic B, Flower-Ellis J (1999) Optimum nutrition and nitrogen saturation in Scots pine stands. Studia Forestalia Suecia 206:1–126, http://pub.epsilon.slu.se/3013/
Templer PH, Silver WL, Pett-Ridge J, DeAngelis KM, Firestone MK (2008) Plant and microbial controls on nitrogen retention and loss in a humid tropical forest. Ecology 89:3030–3040. doi:10.1890/07-1631.1
Thomas RQ, Zaehle S, Templer PH, Goodale CL (2013) Global patterns of nitrogen limitation: confronting two global biogeochemical models with observations. Glob Chang Biol 19:2986–2998. doi:10.1111/gcb.12281
Tunlid A, White DC (1992) Biochemical analysis of biomass, community structure, nutritional status, and metabolic activity of microbial communities in soil. In: Stotzky G, Bollag J-M (eds) Soil Biochemistry. Dekker, New York, pp 229–261
van Diepen LTA, Lilleskov EA, Pregitzer KS, Miller RM (2010) Simulated nitrogen deposition causes a decline of intra- and extraradical abundance of arbuscular mycorrhizal fungi and changes in microbial community structure in northern hardwood forests. Ecosystems 13:683–695. doi:10.1007/s10021-010-9347-0
van Dijk HFG, Roelofs JGM (1988) Effects of excessive ammonium deposition on the nutritional status and condition of pine needles. Physiol Plant 73:494–501. doi:10.1111/j.1399-3054.1988.tb05431.x
White DC, Davis WM, Nickles JS, King JD, Bobbie RJ (1979) Determination of the sedimentary microbial biomass by extractable lipid phosphate. Oecologia 40:51–62. doi:10.1007/BF00388810
Vitousek PM, Aber JD, Howarth RW, Likens GE, Matson PA, Schindler DW, Schlesinger WH, Tilman DG (1997) Human alteration of the global nitrogen cycle: sources and consequences. Ecol Appl 7:737–750. doi:10.1890/1051-0761(1997)007[0737:HAOTGN]2.0.CO;2
Yarwood SA, Myrold DD, Högberg MN (2009) Termination of belowground C allocation by trees alters soil fungal and bacterial communities in a boreal forest soil. FEMS Microbiol Ecol 70:151–162. doi: 10.1111/j.1574-6941.2009.00733.x
Zar JH (1984) Biostatistical Analysis Prentice Hall, New Jersey.
Zechmeister-Boltenstern S, Michel K, Pfeffer M (2011) Soil microbial community structure in European forests in relation to forest type and atmospheric nitrogen deposition. Plant Soil 343:37–50. doi:10.1007/s11104-010-0528-6
Acknowledgments
We thank Prof. Hjalmar Laudon, Swedish University of Agricultural Sciences, for internal review of the manuscript. Funding was provided by the Swedish Research Council for Environment, Agricultural Sciences, and Spatial Planning (FORMAS) and the Swedish University of Agricultural Sciences (a so called KoN grant) to M.N.H., and by the Swedish Science Council (VR) to P.H.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Duncan D. Cameron.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Högberg, M.N., Blaško, R., Bach, L.H. et al. The return of an experimentally N-saturated boreal forest to an N-limited state: observations on the soil microbial community structure, biotic N retention capacity and gross N mineralisation. Plant Soil 381, 45–60 (2014). https://doi.org/10.1007/s11104-014-2091-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-014-2091-z