Abstract
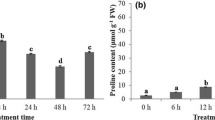
Water deficit is one of the main abiotic factors that affect plant productivity in subtropical regions. To identify genes induced during the water stress response in Bermudagrass (Cynodon dactylon), cDNA macroarrays were used. The macroarray analysis identified 189 drought-responsive candidate genes from C. dactylon, of which 120 were up-regulated and 69 were down-regulated. The candidate genes were classified into seven groups by cluster analysis of expression levels across two intensities and three durations of imposed stress. Annotation using BLASTX suggested that up-regulated genes may be involved in proline biosynthesis, signal transduction pathways, protein repair systems, and removal of toxins, while down-regulated genes were mostly related to basic plant metabolism such as photosynthesis and glycolysis. The functional classification of gene ontology (GO) was consistent with the BLASTX results, also suggesting some crosstalk between abiotic and biotic stress. Comparative analysis of cis-regulatory elements from the candidate genes implicated specific elements in drought response in Bermudagrass. Although only a subset of genes was studied, Bermudagrass shared many drought-responsive genes and cis-regulatory elements with other botanical models, supporting a strategy of cross-taxon application of drought-responsive genes, regulatory cues, and physiological-genetic information.





Similar content being viewed by others
References
Abe H, Urao T, Ito T, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell 15:63–78. doi:10.1105/tpc.006130
AbuQamar S, Chen X, Dhawan R, Bluhm B, Salmeron J, Lam S, Dietrich RA, Mengiste T (2006) Expression profiling and mutant analysis reveals complex regulatory networks involved in Arabidopsis response to Botrytis infection. Plant J 48:28–44. doi:10.1111/j.1365-313X.2006.02849.x
Acevedo-Hernandez GJ, Leon P, Herrera-Estrella LR (2005) Sugar and ABA responsiveness of a minimal RBCS light-responsive unit is mediated by direct binding of ABI4. Plant J 43:506–519. doi:10.1111/j.1365-313X.2005.02468.x
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang A, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. doi:10.1093/nar/25.17.3389
Behel D (2001) TVA research on coal combustion by-products: uses and environmental impacts International ash utilization symposium. Center for Applied Energy Research, University of Kentucky, Kentucky
Bevilacqua A, Fiorenza MT, Mangia F (2000) A developmentally regulated GAGA box-binding factor and Sp1 are required for transcription of the hsp701 gene at the onset of mouse zygotic genome activation. Development 127:1541–1551
Bieri S, Mauch S, Shen QH, Peart J, Devoto A, Casais C, Ceron F, Schulze S, Steinbiss HH, Shirasu K, Schultz-Lefert P (2004) Rar1 positively controls steady state levels of barley MLA resistance proteins and enables sufficient MLA6 accumulation for effective resistance. Plant Cell 16:3480–3495. doi:10.1105/tpc.104.026682
Boudsocq M, Lauriere C (2005) Osmotic signaling in Plants Multiplel pathways mediated by emerging kinase families. Plant Physiol 138:1185–1194. doi:10.1104/pp.105.061275
Cheadle C, Vawter MP, Freed WJ, Becker KG (2003) Analysis of microarray data using Z-score transformation. J Mol Diagn 4:73–81
Deuschle K, Funck D, Forlani G, Stransky H, Biehl A, Leister D, Graaff E, Kunze R, Frommer WB (2004) The role of delta-pyrroline-5-carboxylate dehydrogenase in proline degradation. Plant Cell 16:3413–3425. doi:10.1105/tpc.104.023622
Dunn MA, White AJ, Vural S, Hughes MA (1998) Identification of promoter elements in a low-temperature-responsive gene (blt49) from barley (Hordeum vulgare L). Plant Mol Biol 38:551–564. doi:10.1023/A:1006098132352
Eisen MB, Spellman PT, Brown PO, Botstein D (1998) Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 95:14863–14868. doi:10.1073/pnas.95.25.14863
Ensyn Technologies (1997) Determining the suitability of coastal Bermudagrass and poultry litter as feedstocks for the bio-oil process. Ensyn Technologies New Jersey Inc, Toms River, NJ
Finkelstein RR, Lynch TJ (2000) The Arabidopsis abscisic acid response gene ABI5 encodes a basic leucine zipper transcription factor. Plant Cell 12:599–609
Fujita M, Fujita Y, Noutoshi Y, Takahashi F, Narusaka Y, Yamaguchi-Shinozaki K, Shinozaki K (2006) Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Curr Opin Plant Biol 9:436–442. doi:10.1016/j.pbi.2006.05.014
Gantzer CJ, Anderson SH, Thompson AL, Brown JR (1990) Estimating soil erosion after 100 years of cropping on Sanborn Field. J Soil Water Conserv 45:641–644
Gorantla M, Babu PR, Reddy Lachagari VB, Feltus FA, Paterson AH, Reddy AR (2005) Functional genomics of drought stress response in rice: transcript mapping of annotated unigenes of an indica rice (Oryza sativa L cv Nagina 22). Curr Sci 89:496–514
Gromoff ED, Schroda M, Oster U, Beck C (2006) Identification of a plastid response element that acts as an enhancer within the Chlamydomonas HSP70A promoter. Nucleic Acids Res 34:4767–4779. doi:10.1093/nar/gkl602
Groth D, Lehrach H, Hennig S (2004) GOblet: a platform for Gene Ontology annotation of anonymous sequence data. Nucleic Acids Res 32:W313–W317. doi:10.1093/nar/gkh406
Gygi SP, Rochon Y, Franza BR, Aebersold R (1999) Correlation between protein and mRNA abundance in yeast. Mol Cell Biol 19:1720–1730
Harris MA, Clark J, Ireland A, Lomax J, Ashburner M, Foulger R, Eilbeck K, Lewis S, Marshall B, Mungall C, Richter J, Rubin GM, Blake JA, Bult C, Dolan M, Drabkin H, Eppig J, Hill DP, Ni L, Ringwald M, Balakrishnan R, Cherry J, Christie KR, Costanzo MC, Dwight SS, Engel S, Fisk DG, Hirschman JE, Hong EL, Nash RS, Sethuraman A, Theesfeld CL, Botstein D, Dolinski K, Feierbach B, Berardini T, Mundodi S, Rhee S, Apweiler R, Barrell D, Camon E, Dimmer E, Lee V, Chisholm R, Gaudet P, Kibbe W, Kishore R, Schwarz EM, Sternberg P, Gwinn M, Hannick L, Wortman J, Berriman M, Wood V, de la Cruz N, Tonellato P, Jaiswal P, Seigfried T, White R (2004) The Gene Ontology (GO) database and informatics resource. Nucleic Acids Res 32:D258–D261. doi:10.1093/nar/gkh066
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database. Nucleic Acids Res 27:279–300. doi:10.1093/nar/27.1.297
Huang B, Duncan RR, Carrow RN (1997) Drought-resistance mechanisms of seven warm-season turfgrasses under surface soil drying: I shoot response. Crop Sci 37:1858–1863
Hwang I, Chen H, Sheen J (2002) Two-component signal transduction pathways in Arabidopsis. Plant Physiol 129:500–515. doi:10.1104/pp.005504
Ingram J, Bartels D (1996) The molecular basis of dehydration tolerance in plants. Annu Rev Plant Biol 44:377–403
Ishitani M, Rao I, Wenzl P, Beebe S, Tohme J (2004) Integration of genomics approach with traditional breeding towards improving abiotic stress adaptation: drought and aluminum toxicity as case studies. Field Crops Res 90:35–45. doi:10.1016/j.fcr.2004.07.004
Jauhar PP (1993) Cytogenetics of the Festuca-Lolium complex: relevance to breeding. Springer, Berlin
Kawaguchi R, Girke T, Bray EA, Bailey-Serres J (2004) Differential mRNA translation contributes to gene regulation under non-stress and dehydration stress conditions in Arabidopsis thaliana. Plant J 38:823–839. doi:10.1111/j.1365-313X.2004.02090.x
Kim SY, Chung H, Thomas TL (1997) Isolation of a novel class of bZIP transcription factors that interact with ABA-responsive and embryo-specification elements in the Dc3 promoter using a modified yeast one-hybrid system. Plant J 11:1237–1251. doi:10.1046/j.1365-313X.1997.11061237.x
Kim C, Jang CS, Kamps TL, Robertson JS, Feltus FA, Paterson AH (2008) Transcrioptome analysis of leaf tissue from Cynodon dactylon L. by a normalized cDNA library. Funct Plant Biol 35:585–594. doi:10.1071/FP08133
Lu H, Rate DN, Song JT, Greenberg JT (2003) ACD6, a novel ankyrin protein, is a regulator and an effector of salicylic acid signaling in the arabidopsis defense response. Plant Cell 15:2408–2420. doi:10.1105/tpc.015412
Martinez-Hernandez A, Lopez-Ochoa L, Arguello-Astorga G, Herrera-Estrella L (2002) Functional properties and regulatory complexity of a minimal RBCS light-responsive unit activated by phytochrome, cryptochrome, and plastid signals. Plant Physiol 128:1223–1233. doi:10.1104/pp.010678
McQueen-Mason S, Durachko DM, Cosgrove DJ (1992) Two endogenous proteins that induce cell wall extension in plants. Plant Cell 4:1425–1433
Mohler V, Klahr A, Wenzel G, Schwarz G (2002) A resistance gene analog useful for targeting disease resistance genes against different pathogens on group 1S chromosomes of barley, wheat and rye. Theor Appl Genet 105:364–368. doi:10.1007/s00122-002-0919-z
Moser LE, Hoveland CS (1996) Cool-season grass overview. Crop Science Society of America, Madison, WI
Munns R, Passioura JB, Guo J, Chazen O, Cramer GR (2000) Water relations and leaf expansion: importance of time scale. J Exp Bot 51:1495–1504. doi:10.1093/jexbot/51.350.1495
Nanjo T, Fujita M, Seki M, Kato T, Tabata S, Shinozaki K (2003) Toxicity of free proline revealed in an Arabidopsis T-DNA-tagged mutant deficient in proline dehydrogenase. Plant Cell Physiol 44:541–548. doi:10.1093/pcp/pcg066
Newton RP, Brenton AG, Smith CJ, Dudley E (2004) Plant proteome analysis by mass spectrometry: principles, problems, pitfalls and recent developments. Phytochemistry 65:1449–1485. doi:10.1016/j.phytochem.2004.04.015
Ozturk ZN, Talame V, Deyholos M, Michalowski CB, Galbraith DW, Gozukirmizi N, Tuberosa R, Bohnert HJ (2002) Monitoring large-scale changes in transcript abundance in drought-and salt-stressed barley. Plant Mol Biol 48:551–573. doi:10.1023/A:1014875215580
Randall GW, Mulla D (2001) Nitrate nitrogen in surface water as influenced by climatic conditions and agricultural practices. J Environ Qual 20:337–344
Ribaut J-M, Ragot M (2007) Marker-assisted selection to improve drought adaptation in maize: the backcross approach, perspectives, limitations, and alternatives. J Exp Bot 58:351–360. doi:10.1093/jxb/erl214
Ribaut J-M, Banziger M, Hoisington D (2002) Genetic dissection and plant improvement under abiotic stress conditions: drought tolerance in maze as an example. JIRCAS Working Rep 23:85–92
Rouhier N, Gelhaye E, Jacquot JP (2004) Plant glutaredoxins still mysterious reducing systems. Cell Mol Life Sci 61:1266–1277. doi:10.1007/s00018-004-3410-y
Rzewuski G, Sauter M (2002) The novel rice (Oryza sativa L) gene OsSbf1 encodes a putative member of the Na+/bile acid symporter family. J Exp Bot 53:1991–1993. doi:10.1093/jxb/erf053
Sangwan I, O’Brian MR (2002) Identification of a soybean protein that interacts with GAGA element dinucleotide repeat DNA. Plant Physiol 129:1788–1794. doi:10.1104/pp.002618
Santi L, Wang Y, Stile MR, Berendzen K, Wanke D, Roig C, Pozzi C, Muller K, Muller J, Rohde W, Salamini F (2003) The GA octodinucleotide repeat binding factor BBR participates in the transcriptional regulation of the homeobox gene Bkn3. Plant J 34:813–826. doi:10.1046/j.1365-313X.2003.01767.x
Satoh R, Nakashima K, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2002) ACTCAT, a novel cis-acting element for proline- and hypoosmolarity-responsive expression of the ProDH gene encoding proline dehydrogenase in Arabidopsis. Plant Physiol 130:709–719. doi:10.1104/pp.009993
Stone SL, Williams LA, Farmer LM, Vierstra RD, Callis J (2006) KEEP ON GOING, a RING E3 ligase essential for Arabidopsis growth and development, is involved in abscisic acid signaling. Plant Cell 18:3415–3428. doi:10.1105/tpc.106.046532
Suprunova T, Krugman T, Distelfeld A, Fahima T, Nevo E, Korol A (2007) Identification of a novel gene (Hsdr4) involved in water-stress tolerance in wild barley. Plant Mol Biol 64:17–34. doi:10.1007/s11103-006-9131-x
Talame V, Ozturk ZN, Bohnert HJ, Tuberosa R (2007) Barley transcript profiles under dehydration shock and drought stress treatments: a comparative analysis. J Exp Bot 58:229–240. doi:10.1093/jxb/erl163
Tanaka R, Oster U, Kruse E, Rudiger W, Grimm B (1999) Reduced activity of geranylgeranyl reductase leads to loss of chlorophyll and tocopherol and to partially geranylgeranylated chlorophyll in transgenic tobacco plants expressing antisense RNA for geranylgeranyl reductase. Plant Physiol 120:695–704. doi:10.1104/pp.120.3.695
Verslues PE, Sharp RE (1999) Proline accumulation in maize (Zea mays L) primary roots at low water potentials II metabolic source of increased proline deposition in the elongation zone. Plant Physiol 119:1349–1360. doi:10.1104/pp.119.4.1349
Wang Z, Hopkins A, Mian MAR (2001) Forage and turf grass biotechnology. Crit Rev Plant Sci 20:573–619. doi:10.1016/S0735-2689(01)80005-8
Welcker C, Boussuge B, Bencivenni C, Ribaut J-M, Tardieu F (2007) Are source and sink strengths genetically linked in maize plants subjected to water deficit? A QTL study of the responses of leaf growth and of Anthesis-Silking Interval to water deficit. J Exp Bot 58:339–349. doi:10.1093/jxb/erl227
Wu Y, Meeley RB, Cosgrove DJ (2001) analysis and expression of the α-expansin and β-expansin gene families in maize. Plant Physiol 126:222–232. doi:10.1104/pp.126.1.222
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol 57:781–803. doi:10.1146/annurev.arplant.57.032905.105444
Zheng B, Halperin T, Hruskova-Heidingsfeldova O, Adam Z, Clarke AK (2002) Characterization of chloroplast Clp proteins in Arabidopsis: localization, tissue specificity and stress responses. Physiol Plant 114:92–101. doi:10.1034/j.1399-3054.2002.1140113.x
Zheng J, Zhao J, Tao Y, Wang J, Liu Y, Fu J, Jin Y, Gao P, Zhang J, Bai Y, Wang G (2004) Isolation and analysis of water stress induced genes in maize seedlings by subtractive PCR and cDNA macroarray. Plant Mol Biol 55:807–823
Zhou J, Wang X, Jiao Y, Qin Y, Liu X, He K, Chen C, Ma L, Wang J, Xiong L, Zhang Q, Fan L, Deng XW (2007) Global genome expression analysis of rice in response to drought and high-salinity stress in shoot, flag leaf, and panicle. Plant Mol Biol 63:591–608. doi:10.1007/s11103-006-9111-1
Acknowledgments
This work was supported by a grant from the United States Golf Association (USGA).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, C., Lemke, C. & Paterson, A.H. Functional dissection of drought-responsive gene expression patterns in Cynodon dactylon L.. Plant Mol Biol 70, 1–16 (2009). https://doi.org/10.1007/s11103-009-9453-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-009-9453-6