Abstract
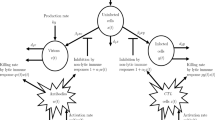
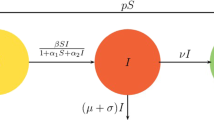
A hepatitis B or C virus (HBV or HCV) epidemic model with intra- and extra-hepatic coinfection, immune delay and saturation incidence, as well as antiviral therapy is proposed in this paper. The existence of equilibria (infection-free, immune-free and immune-activated), the basic reproduction numbers, i.e., \(R_{0}\), \(R_{1}\), are given respectively, by which the criteria on (local and global) stability of above equilibria are established. Furthermore, if the immune delay \(\tau >\tau _{0}\), both the existence of subcritical (supercritical) Hopf bifurcation on the immune-activated equilibrium \(E^{*}\), and the stability of bifurcating periodic solutions are obtained. Finally, the theoretical results are demonstrated by numerical simulations. We derive that the immune delay and intra- and extra-hepatic coinfection have significant influence on the transmission of HBV/HCV, could cause more complicated dynamics at \(E^{*}\) from stability to unstablity untill bifurcation, which greatly increases the difficulty of disease control. While effective antiviral therapy could evidently decrease the spread of HBV/HCV.

















Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Organisation, W.H.: Hepatitis Pandemic. https://www.who.int/health-topics/hepatitis. Retrieved: 15/10/2022
Bonhoeffer, S., May, R.M., Shaw, G.M., et al.: Virus dynamics and drug therapy. Proc. Natl. Acad. Sci. U. S. A. 94(13), 6971–6976 (1997). https://doi.org/10.1073/pnas.94.13.6971
Nowak, M., Bonhoeffer, S., Hill, A., et al.: Viral dynamics in hepatitis B infection. Proc. Natl. Acad. Sci. U. S. A. 93(9), 4398–4402 (1996). https://doi.org/10.1073/pnas.93.9.4398
Wang, K.F., Wang, W.D., Pang, H., et al.: Complex dynamic behavior in a viral model with delayed immune response. Physica D 226(2), 197–208 (2007). https://doi.org/10.1016/j.physd.2006.12.001
Mehmood, M., Hamid, M., Ashraf, S., et al.: Galerkin time discretization for transmission dynamics of HBV with non-linear saturated incidence rate. Appl. Math. Comput. 410(1), 1–18 (2021). https://doi.org/10.1016/j.amc.2021.126481
Pal, S., Anal, C.: Dynamics of the interaction of plankton and planktivorous fish with delay. Cogent Math. 2, 1–27 (2015). https://doi.org/10.1080/23311835.2015.1074337
Yang, H., Wei, J.J.: Analyzing global stability of a viral model with general incidence rate and cytotoxic T lymphocytes immune response. Nonlinear Dyn. 82(1–2), 713–722 (2015). https://doi.org/10.1007/s11071-015-2189-8
Guidotti, L.G., Ishikawa, T., Hobbset, M., et al.: Intracellular inactivation of the hepatitis B virus by cytotoxic T lymphocytes. Immunity 4(1), 25–36 (1996). https://doi.org/10.1016/S1074-7613(00)80295-2
Seeger, C., Mason, W.S.: Molecular biology of hepatitis B virus infection. Virology 479, 672–686 (2015). https://doi.org/10.1016/j.virol.2015.02.031
Nowak, M.A., Bangham, C.R.M.: Population dynamics of immune responses to persistent virus. Science 272, 74–79 (1996). https://doi.org/10.1126/science.272.5258.74
Canabarro, A.A., Glerial, I.M., Lyra, M.L.: Periodic solutions and chaos in a non-linear model for the delayed cellular immune response. Phys. A 342(1–2), 234–241 (2004). https://doi.org/10.1016/j.physa.2004.04.083
Bertoletti, A., Ferrari, C.: Kinetics of the immune response during HBV and HCV infection. Hepatology 38(1), 4–13 (2003). https://doi.org/10.1053/jhep.2003.50310
Major, M.E., Mihalik, K., Fernandez, J., et al.: Long-term follow-up of chimpanzees inoculated with the first infectious clone for hepatitis C virus. J. Virol. 73(4), 3317–3325 (1999). https://doi.org/10.1128/JVI.73.4.3317-3325.1999
Tian, X.H., Xu, R.: Global stability and Hopf bifurcation of an HIV-1 infection model with saturation incidence and delayed CTL immune response. Appl. Math. Comput. 237, 146–154 (2014). https://doi.org/10.1016/j.amc.2014.03.091
Ebert, D., Zschokke-Rohringer, C.D., Carins, H.J.: Does effects and density dependent regulation of two microparasites of Daphnia magna. Oecologia 122(2), 200–209 (2000). https://doi.org/10.1007/PL00008847
Song, X., Neumann, A.U.: Global stability and periodic solution of the viral dynamics. J. Math. Anal. Appl. 329(1), 281–297 (2007). https://doi.org/10.1016/j.jmaa.2006.06.064
Berenguer, M., Prieto, M., Juan, F.S., et al.: Contribution of donor age to the recent decrease in patient survival among HCV-infected liver transplant recipients. Hepatology 36(1), 202–210 (2002). https://doi.org/10.1053/jhep.2002.33993
Tsiang, M., Rooney, J., Toole, J., et al.: Biphasic clearance kinetics of hepatitis B virus from patients during adefovir dipivoxil therapy. Hepatology 29(6), 1863–1869 (1999). https://doi.org/10.1002/hep.510290626
Pontisso, P., Poon, M.C., Tiollais, P., et al.: Detection of hepatitis B virus DNA in mononuclear blood cells. Brit. Med. J. 288(6430), 1563–1566 (1984). https://doi.org/10.1136/bmj.288.6430.1563
Pontisso, P., Vidalino, L., Quarta, S., Gatta, A.: Biological and clinical implications of HBV infection in peripheral blood mononuclear cells. Autoimmun. Rev. 8(1), 13–17 (2008). https://doi.org/10.1016/j.autrev.2008.07.016
Lanford, R.E., Michaels, M.G., Chavez, D., et al.: Persistence of extrahepatic hepatitis B virus DNA in the absence of detectable hepatic replication in patients with baboon liver transplants. J. Med. Virol. 46(3), 207–212 (1995). https://doi.org/10.1002/jmv.1890460307
Mason, A., Theal, J., Bain, V., et al.: Hepatitis B virus replication in damaged endothelial tissues of patients with extrahepatic disease. Am. J. Gastroenterol. 100(4), 972–976 (2005). https://doi.org/10.1111/j.1572-0241.2005.41308.x
Cacoub, P., Asselah, T.: Hepatitis B virus infection and extra-hepatic manifestations: a systemic disease. Am. J. Gastroenterol. 117(2), 253–263 (2022). https://doi.org/10.14309/ajg.0000000000001575
Inoue, J., Ueno, Y., Kogure, T., et al.: Analysis of the full-length genome of hepatitis B virus in the serum and cerebrospinal fluid of a patient with acute hepatitis B and transverse myelitis. J. Clin. Virol. 41(4), 301–304 (2008). https://doi.org/10.1016/j.jcv.2008.01.002
Kamiza, A.B., Su, F.H., Wang, W.C., et al. Chronic hepatitis infection is associated with extrahepatic cancer development: A nationwide population-based study in Taiwan. BMC Cancer 16, (2016). https://doi.org/10.1186/s12885-016-2918-5
Rong, Q.F., Huang, J., Su, E.B., et al., Infection of hepatitis B virus in extrahepatic endothelial tissues mediated by endothelial progenitor cells, Virol. J. 4, (2007). https://doi.org/10.1186/1743-422X-4-36
Zheng, X.Y., Wei, R.B., Tang, L., et al.: Meta-analysis of combined therapy for adult hepatitis B virus-associated glomerulonephritis. World J. Gastroenterol. 18(8), 821–832 (2012). https://doi.org/10.3748/wjg.v18.i8.821
Galun, E., Offensperger, W.B., Weizacker, F., et al.: Human non-hepatocytes support hepadnaviral replication and virion production. J. Gen. Viro. 73, 173–178 (1992). https://doi.org/10.1099/0022-1317-73-1-173
Wodarz, D.: Hepatitis C virus dynamics and pathology: the role of CTL and antibody responses. J. Gen. Virol. 84, 1743–1750 (2003). https://doi.org/10.1099/vir.0.19118-0
Cui, J.A., Pang, J.H., Hui, J.: The importance of immune responses in a model of hepatitis B virus. Nonlinear Dyn. 67(1), 723–734 (2011). https://doi.org/10.1007/s11071-011-0022-6
Dhar, M., Samaddar, S., Bhattacharya, P.: Modeling the effect of non-cytolytic immune response on viral infection dynamics in the presence of humoral immunity. Nonlinear Dyn. 98(1), 637–655 (2019). https://doi.org/10.1007/s11071-019-05219-8
Qesmi, R., Wu, J., Wu, J.H., et al.: Influence of backward bifurcation in a model of hepatitis B and C viruses. Math. Biosci. 224(2), 118–125 (2010). https://doi.org/10.1016/j.mbs.2010.01.002
Carrozzo, M., Quadri, R., Latorre, P., et al.: Molecular evidence that the hepatitis C virus replicates in the oral mucosa. J. Hepatol. 37(3), 364–369 (2002). https://doi.org/10.1016/S0168-8278(02)00183-6
Zhu, Q., Guo, J.T., Seeger, C.: Replication of hepatitis C virus subgenomes in nonhepatic epithelial and mouse hepatoma cells. J. Virol. 77(17), 9204–9210 (2003). https://doi.org/10.1128/JVI.77.17.9204-9210.2003
De Vita, S., De Re, V., Sansonno, D., et al.: Gastric mucosa as an additional extrahepatic localization of hepatitis C virus: viral detection in gastric low-grade lymphoma associated with autoimmune disease and in chronic gastritis. Hepatology 31(1), 182–189 (2000). https://doi.org/10.1002/hep.510310127
Dahari, H., Feliu, A., Garcia-Retortillo, M., et al.: Second hepatitis C replication compartment indicated by viral dynamics during liver transplantation. J. Hepatol. 42(4), 491–498 (2005). https://doi.org/10.1016/j.jhep.2004.12.017
Hu, X.L., Li, J.Q., Feng, X.M.: Threshold dynamics of a HCV model with virus to cell transmission in both liver with CTL immune response and the extrahepatic tissue. J. Biol. Dyn. 15(1), 19–34 (2021). https://doi.org/10.1080/17513758.2020.1859632
Webmed: Combination Antiviral Therapy for Hepatitis C. http://www.webmd.com/hepatitis/combination-antiviral-therapy-for-hepatitis-c
Webmed, Hepatitis Health Center: Hepatitis B medications. http://www.webmd.com/hepatitis/hepb-guide/hepatitis-b-medications
Ribeiro, R., Lo, A., Perelson, A.: Dynamics of hepatitis B virus infection. Microbes Infect. 4(8), 829–835 (2002). https://doi.org/10.1016/S1286-4579(02)01603-9
Smith, H.L.: Monotone Dynamical Systems: An Introduction to the Theory of Competitive and Cooperative Systems. American Mathematical Society, Providence (1995)
LaSalle, J.P.: The stability of dynamical systems. In: Regional Conference Series in Applied Mathematics, SIAM, Philadelphia (1976)
Kuang, Y.: Delay Differential Equations with Application in Population Dynamics. Academic Press Inc, Boston, MA (1993)
Hassard, B.D., Kazarinoff, N.D., Wan, Y.: Theory and Application of Hopf Bifurcation. Cambridge University Press, Cambridge (1981)
Song, Y., Wei, J.: Bifurcation analysis for Chen’s system with delayed feedback and its application to control of chaos. Chaos Solit. Fract. 22(1), 75–91 (2004). https://doi.org/10.1016/j.chaos.2003.12.075
Ciupe, S., Rebeiro, R., Nelson, P., et al.: Modeling the mechanisms of acute hepatitis B virus infection. J. Theor. Biol. 247(1), 23–35 (2007). https://doi.org/10.1016/j.jtbi.2007.02.017
Hews, S., Eikenberry, S., Nagy, J., et al.: Rich dynamics of a hepatitis B viral infection model with logistic hepatocyte growth. J. Math. Biol. 60(4), 573–590 (2010). https://doi.org/10.1007/s00285-009-0278-3
Ji, Y., Min, L., Zheng, Y., et al.: A viral infection model with periodic immune response and nonlinear CTL response. Math. Comput. Simul. 80(12), 2309–2316 (2010). https://doi.org/10.1016/j.matcom.2010.04.029
Prakash, M., Rakkiyappan, R., Manivannan, A.: Stability and bifurcation analysis of hepatitis B-type virus infection model. Math. Meth. Appl. Sci. 44(8), 6462–6481 (2021). https://doi.org/10.1002/mma.7198
Song, X.Y., Wang, S.L., Dong, J.: Stability properties and Hopf bifurcation of a delayed viral infection model with lytic immune response. J. Math. Anal. Appl. 373(2), 345–355 (2011). https://doi.org/10.1016/j.jmaa.2010.04.010
Steven, H.S.: Nonlinear Dynamics and Chaos. Boca Raton London, New York (2015)
Ito, H., Yoshioka, K., Ukai, K., et al.: The fluctuations of viral load and serum alanine aminotransferase levels in chronic hepatitis C. Hepatol. Res. 30(1), 11–17 (2004). https://doi.org/10.1016/j.hepres.2004.06.002
Ortiz, G.M., Hu, J., Goldwitz, J.A., et al.: Residual viral replication during antiretroviral therapy boosts human immunodeficiency virus type 1-specific CD8\(^{+}\) T-cell responses in subjects treated early after infection. J. Virol. 76(1), 411–415 (2002). https://doi.org/10.1128/JVI.76.1.411-415.2002
Wang, Y., Liu, X.N.: Stability and Hopf bifurcation of a within-host chikungunya virus infection model with two delays. Math. Comput. Simul. 138, 31–48 (2017). https://doi.org/10.1016/j.matcom.2016.12.011
Funding
This work was supported by the the Natural Science Foundation of Xinjiang Province, People’s Republic of China (2022D01E41), the National Natural Science Foundation of China (Grant Nos. 12261087, 11861065, 12262035), the Open Project of Key Laboratory of Applied Mathematics of Xinjiang Uygur Autonomous Region, China (Grant No. 2021D04014), the Scientific Research Programmes of Colleges in Xinjiang, People’s Republic of China (Grant No. XJEDU2021I002, XJEDU2021Y001), the Postgraduate Research and Innovation Program of Xinjiang Uygur Autonomous Region, China (Grant No. XJ2022G020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing of interests
The authors have no conflicts to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A: The details in Sect. 3.2
Appendix B: The details in Sect. 3.3
Appendix C: The details in Sect. 5.1
Appendix D: The details in Sect. 6
We have obtained that the model (6) experiences Hopf bifurcation at the equilibrium \(E^{*}\) for \(\tau =\tau _{0}\), and \(\pm i\omega _{0}\) is the pure imaginary root corresponding to the characteristic equation at \(E^{*}\).
Let \(\widetilde{T_{l}}(t)=T_{l}(t)-T_{l}^{*}\), \(\widetilde{I_{l}}(t)=I_{l}(t)-I_{l}^{*}\), \(\widetilde{T_{e}}(t)=T_{e}(t)-T_{e}^{*}\), \(\widetilde{I_{e}}(t)=I_{e}(t)-I_{e}^{*}\), \(\widetilde{V}(t)=V(t)-V^{*}\), \(\widetilde{Z}(t)=Z(t)-Z^{*}\), and replace \(\widetilde{T_{l}}\), \(\widetilde{I_{l}}\), \(\widetilde{T_{e}}\), \(\widetilde{I_{e}}\), \(\widetilde{V}\), \(\widetilde{Z}\), with \(T_{l}\), \(I_{l}\), \(T_{e}\), \(I_{e}\), V, Z.
Then we define \(\tau =\tau _{0}+\mu \), \(t=s\tau \), \(T_{l}(s\tau )=\overline{T_{l}}(s)\), \(I_{l}(s\tau )=\overline{I_{l}}(s)\), \(T_{e}(s\tau )=\overline{T_{e}}(s)\), \(I_{e}(s\tau )=\overline{I_{e}}(s)\), \(V(s\tau )=\overline{V}(s)\), \(Z(s\tau )=\overline{Z}(s)\), and still denote \(\overline{T_{l}}\), \(\overline{I_{l}}\), \(\overline{T_{e}}\), \(\overline{I_{e}}\), \(\overline{V}\), \(\overline{Z}\) as \(T_{l}\), \(I_{l}\), \(T_{e}\), \(I_{e}\), V, Z. The linear term at the right of the transformed model is sorted out through calculation is (D1),
where
And the nonlinear term at the right of the transformed model is (D2),
where
and
Then the model (6) could be written as a functional differential equation in \(C =C([-1, 0],\ R_{+}^{6})\).
For \(\phi (\theta )=(\phi _{1}(\theta ), \phi _{2}(\theta ), \phi _{3}(\theta ), \phi _{4}(\theta ), \phi _{5}(\theta ), \phi _{6}(\theta ))^{T}\in C\), define operator \(L_{\mu }\phi =(\tau _{0}+\mu )\mathbb {M}_{1}\phi (0)+(\tau _{0}+\mu )\mathbb {M}_{2}\phi (-1)\), where
According to the Riesz representation theorem, there is a matrix value function \(\eta (\theta , \mu ):[-1, 0]\rightarrow R^{6}\), such that
then we have
For \(\phi \in C([-1, 0],\ R_{+}^{6})\), we define
and
Therefore, model (6) is equivalent to abstract differential equation (D6)
Next, we can discuss the Eq. (D6).
For \(\varphi \in C^{1}([0,1], (R^{6})^{*})\), we can define the adjoint operator,
and define the bilinear inner product as (D8)
where \(\eta (\theta )=\eta (\theta ,0)\), and the conjugate operator of \(A(\mu )\) is \(A^{*}\). Based on the above discussion, \(\pm i\omega _{0}\tau _{0}\) is the eigenvalue of A(0) and \(A^{*}\). Then we will calculate the eigenvectors of A and \(A^{*}\) about \(i\omega _{0}\tau _{0}\) and \(-i\omega _{0}\tau _{0}\).
Suppose that \(q(\theta )\) and \(q^{*}(s)\) are the eigenvectors of \(A(\mu _{0})\) and \(A^{*}\) corresponding to \(i\omega _{0}\tau _{0}\) and \(-i\omega _{0}\tau _{0}\) respectively, and \(\langle q^{*}(s), q(\theta )\rangle =1\).
When \(\theta \ne 0\), according to the definition of A(0), easily that \(q(\theta )=q(0)e^{i\tau _{0}\omega _{0}\theta }\) and q(0) satisfies the following equation,
where I is the identity matrix, and \(q(0)=(q_{1}, q_{2}, q_{3}, q_{4}, q_{5}, q_{6})^{T}\), calculation shows that
where \(M_{1}=Q_{1}+d_{l}\), \(M_{2}=Q_{3}+d_{e}\). Similarly, \(q^{*}\) satisfies the following equation
we can further get
Let \(q^{*}(s)=Dq^{*}e^{i\tau _{0}\omega _{0}s}\), we demand to use \(\langle q^{*}(s), q(\theta )\rangle =1\) to determine the formula of D,
Thus, we can choose D as
at the same time, we can easily obtain \(\langle q^{*}(s),\overline{q}(\theta )\rangle =0\).
According to the algorithm in [52] and the similar discussion in [45], we can calculate the corresponding coefficients,
Further, we solve the expressions of \(W_{20}(\theta )\) and \(W_{11}(\theta )\),
and \(E_{1}\) \(E_{2}\) can be calculated as follows,
where \(H_{20}(0)=g_{20}(\overline{q^{*}}(0))^{-1}\), \(H_{11}(0)=g_{11}(\overline{q^{*}}(0))^{-1}\).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, B., Zhang, Y., Sang, Y. et al. Stability and Hopf bifurcation on an immunity delayed HBV/HCV model with intra- and extra-hepatic coinfection and saturation incidence. Nonlinear Dyn 111, 14485–14511 (2023). https://doi.org/10.1007/s11071-023-08580-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-023-08580-x