Abstract
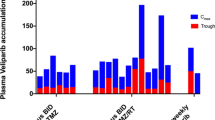
Temozolomide (TMZ) is an oral alkylating agent with proven antitumoral activity in preclinical and clinical studies in adults with high-grade glioma (HGG). However, only limited efficacy has been reported in children with HGG using the 5-day schedule. This study investigated the safety of administering TMZ to children and adolescents with brain tumors over an extended period. Extended schedules have been proven to overcome chemoresistance without any major toxicity. The toxicity of TMZ, administered at 70 mg/m2/day orally for 21 consecutive days every 28 days, was assessed in children with brain tumors. A total of 156 courses of TMZ were given to 17 patients (median age 12.5 years, range 1–17 years), who were recruited into the study. Eleven patients had progressive or relapsing disease, and six patients were newly diagnosed. In this cohort no cases of toxic death or nonhematological toxicity were reported. In comparison with the 5-day schedule, thrombocytopenia and neutropenia were noted to be less frequent. Grades 3 and 4 lymphopenia occurred in 10.8 and 22.4% of courses, respectively; among the lymphopenic patients there was one case of disseminated zoster (meningoencephalitis and cutaneous involvement), one case of rotavirus gastroenteritis, and two cases of herpetic stomatitis reported. The objective response rate was 11.8%. Overall, 82.3% of patients showed stable disease. The prolonged TMZ schedule appeared to be well tolerated, with few cases of neutropenia or thrombocytopenia recorded. Nevertheless, prolonged exposure to TMZ was associated with lymphopenia and may lead to a higher rate of viral infections.
Similar content being viewed by others
References
Newlands ES, Blackledge GR, Slack JA, Rustin GJ, Smith DB, Stuart NS (1992) Phase I trial of temozolomide. Br J Cancer 65:287–291
Yung WK, Prados MD, Yaya-Tur R et al (1999) Multicenter phase II trial of temozolomide in patients with anaplastic astrocytoma or anaplastic oligoastrocytoma at first relapse: Temodal Brain Tumor Group. J Clin Oncol 17:2762–2771
Lashford LS, Thiesse P, Jouvet A et al (2002) Temozolomide in malignant gliomas of childhood: a United Kingdom children’s cancer study group and French society for pediatric oncology intergroup study. J Clin Oncol 20:4684–4691
Verschuur AC, Grill J, Vassal G et al (2004) Temozolomide in pediatric high-grade glioma: a key for combination therapy ? Br J Cancer 91:425–429
Kuo DJ, Weiner HL, Wisoff J, Miller DC, Knopp EA, Finlay JL (2003) Temozolomide is active in childhood, progressive unresectable, low-grade gliomas. J Pediatr Hematol Oncol 25:372–378
Khaw SL, Coleman LT, Downie PA, Heath JA, Ashley DM (2007) Temozolomide in pediatric low-grade glioma. Pediatr Blood Cancer 49:808–811
Nicholson HS, Kretschmar CS, Krailo M et al (2007) Phase 2 study of temozolomide in children and adolescents with recurrent central nervous system tumors: a report from the Children’s Oncology Group. Cancer 110:1542–1550
Wick W, Platten M, Weller M (2009) New (alternative) temozolomide regimens for the treatment of glioma. Neuro-Oncology 11:69–79
Kong DS, Lee JI, Kim WS et al (2006) A pilot study of metronomic temozolomide treatment in patients with recurrent temozolomide-refractory glioblastoma. Oncol Rep 16:1117–1121
Kesari S, Schiff D, Drappatz J et al (2009) Phase II study of protracted daily temozolomide for low-grade gliomas in adults. Clin Cancer Res 15:330–337
Tolcher AW, Gerson SL, Denis L et al (2003) Marked inactivation of O6-alkylguanine-DNA alkyltransferase activity with protracted temozolomide schedules. Br J Cancer 88:1004–1011
Bertolini F, Paul S, Mancuso P et al (2003) Maximum tolerable dose and low-dose metronomic chemotherapy have opposite effects on the mobilization and viability of circulating endothelial progenitor cells. Cancer Res 63:4342–4346
Miller KD, Sweeney CJ, Sledge GW Jr (2001) Redefining the target: chemotherapeutics as antiangiogenics. J Clin Oncol 19:1195–1206
Zhou Q, Guo P, Wang X, Nuthalapati S, Gallo JM (2007) Preclinical pharmacokinetic and pharmacodynamic evaluation of metronomic and conventional temozolomide dosing regimens. J Pharmacol Exp Ther 321:265–275
Virrey JJ, Golden EB, Sivakumar W et al (2009) Glioma-associated endothelial cells are chemoresistant to temozolomide. J Neurooncol 95:13–22
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
http://ctep.cancer.gov/forms/CTCAE.pdf. Accessed January 1, 2004
Sterba J, Pavelka Z, Slampa P (2002) Concomitant radiotherapy and metronomic temozolomide in pediatric high-risk brain tumors. Neoplasma 49:117–120
Su YB, Sohn S, Krown SE et al (2004) Selective CD4+ lymphopenia in melanoma patients treated with temozolomide: a toxicity with therapeutic implications. J Clin Oncol 22:610–616
Ruggiero A, Cefalo G, Garré ML et al (2006) Phase II trial of temozolomide in children with recurrent high-grade glioma. J Neurooncol 77:89–94
Brock CS, Newlands ES, Wedge SR et al (1998) Phase I trial of temozolomide using an extended continuous oral schedule. Cancer Res 58:4363–4367
Khan RB, Raizer JJ, Malkin MG et al (2002) A phase II study of extended low-dose temozolomide in recurrent malignant gliomas. Neuro-Oncology 4:39–43
Stupp R, Dietrich P, Ostermann Kraljevic S et al (2002) Promising survival for patients with newly diagnosed glioblastoma multiforme treated with concomitant radiation plus temozolomide followed by adjuvant temozolomide. J Clin Oncol 20:1375–1382
Ganière V, Christen G, Bally F et al (2006) Listeria brain abscess, Pneumocystis pneumonia and Kaposi’s sarcoma after temozolomide. Nat Clin Pract Oncol 3:339–343
Baruchel S, Diezi M, Hargrave D et al (2006) Safety and pharmacokinetics of temozolomide using a dose-escalation, metronomic schedule in recurrent paediatric brain tumours. Eur J Cancer 42:2335–2342
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ridola, V., Barone, G., Lazzareschi, I. et al. Feasibility study of 21-day-on/7-day-off temozolomide in children with brain tumors. J Neurooncol 103, 147–153 (2011). https://doi.org/10.1007/s11060-010-0371-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-010-0371-x