Abstract
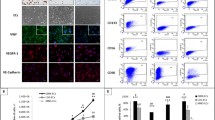
Temozolomide is considered the standard of care and drug of choice for the treatment of initially diagnosed malignant gliomas. Although well tolerated, temozolomide still has limited clinical efficacy. Following drug treatment, patient prognosis still remains poor; tumor recurrence is almost universal. We hypothesized that this lack of effectiveness with temozolomide is because this drug does not target the glioma microenvironment, which is highly vascular in malignant gliomas. To test this hypothesis we analyzed the effects of temozolomide on the tumor vasculature in vitro and in vivo. We found that this drug did not affect the viability or proliferation rate of endothelial cells isolated from human glioma specimens, although temozolomide was highly cytotoxic to the glioma cell lines U87MG and U251. Furthermore, temozolomide did not inhibit the migration of these glioma-associated endothelial cells, a key mechanism responsible for tumor angiogenesis. In in vivo studies, using the intracranial glioma mouse model, temozolomide did not cause a pronounced effect on microvessel density. Our findings show that temozolomide has no apparent effect on the glioma vascular microenvironment. Thus combination therapy with anti-vascular agents may enhance temozolomide effectiveness as glioma therapeutic protocol.





Similar content being viewed by others
References
Dehdashti AR, Hegi ME, Regli L et al (2006) New trends in the medical management of glioblastoma multiforme: the role of temozolomide chemotherapy. Neurosurg Focus 20:E6. doi:10.3171/foc.2006.20.4.3
Stupp R, Hegi ME, van den Bent MJ et al (2006) Changing paradigms—an update on the multidisciplinary management of malignant glioma. Oncologist 11:165–180. doi:10.1634/theoncologist.11-2-165
Nagasubramanian R, Dolan ME (2003) Temozolomide: realizing the promise and potential. Curr Opin Oncol 15:412–418. doi:10.1097/00001622-200311000-00002
Fukumura D, Jain RK (2007) Tumor microvasculature and microenvironment: targets for anti-angiogenesis and normalization. Microvasc Res 74(2–3):72–84. doi:10.1016/j.mvr.2007.05.003
Charalambous C, Hofman FH, Chen TC (2005) Functional and phenotypic differences between glioblastoma multiforme-derived and normal human brain endothelial cells. J Neurosurg 102:699–705
Ramjeesingh R, Leung R, Siu CH (2003) Interleukin-8 secreted by endothelial cells induces chemotaxis of melanoma cells through the chemokine receptor CXCR1. FASEB J 17(10):1292–1294
Jones J, Berkhoff S, Weich E et al (2007) Transient downregulation of beta 1 integrin subtypes on kidney carcinoma cells is induced by mechanical contact with endothelial cell membranes. J Cell Mol Med 11(4):826–838. doi:10.1111/j.1582-4934.2007.00071.x
Virrey JJ, Guan S, Li W, Schönthal AH, Chen TC, Hofman FM (2008) Increased surviving expression confers chemoresistance to tumor-associated endothelial cells. Am J Pathol 173(2):575–585
Charalambous C, Virrey JJ, Kardosh A, Jabbour MN, Qazi-Abdullah L, Pen L, Zidovetzki R, Schönthal AH, Chen TC, Hofman FM (2007) Glioma-associated endothelial cells show evidence of replicative senescence. Exp Cell Res 313:1192–1202. doi:10.1016/j.yexcr.2006.12.027
Chrzanowska-Wodnicka M, Kraus AE, Gale D et al (2008) Defective angiogenesis, endothelial cell migration, proliferation, and MAPK signaling in Rap1b-deficient mice. Blood 111(5):2647–2656. doi:10.1182/blood-2007-08-109710
Toschi E, Bacigalupo I, Strippoli R (2006) HIV-1 Tat regulates endothelial cell cycle progression via activation of the Ras/ERK MAPK signaling pathway. Mol Biol Cell 17(4):1985–1994. doi:10.1091/mbc.E05-08-0717
Charalambous C, Pen LB, Su YS, Milan J, Chen TC, Hofman FM (2005) Interleukin-8 differentially regulates migration of tumor-associated and normal human brain endothelial cells. Cancer Res 65:10347–10354. doi:10.1158/0008-5472.CAN-05-0949
Gately S, Li WW (2004) Multiple roles of COX-2 in tumor angiogenesis: a target for antiangiogenic therapy. Semin Oncol 31(2 Suppl 7):22–29
Raut CP, Nawrocki S, Lashinger LM et al (2004) Celecoxib inhibits angiogenesis by inducing endothelial cell apoptosis in human tumor pancreatic xenografts. Cancer Biol Ther 3(12):1217–1224
Son MJ, Kim JS, Kim MH et al (2006) Combination treatment with temozolomide and thalidomide inhibits tumor growth and angiogenesis in an orthotopic glioma model. Int J Oncol 28(1):53–59
Agarwala SS, Kirkwood JM (2000) Temozolomide, a novel alkylating agent with activity in the central nervous system, may improve the treatment of advanced metastatic melanoma. Oncologist 5(2):144–151. doi:10.1634/theoncologist.5-2-144
Kim JT, Kim JS, Ko KW et al (2006) Metronomic treatment of temozolomide inhibits tumor cell growth through reduction of angiogenesis and augmentation of apoptosis in orthotopic models of gliomas. Oncol Rep 16(1):33–39
Shaked Y, Kerbel RS (2007) Antiangiogenic strategies on defense: on the possibility of blocking rebounds by the tumor vasculature after chemotherapy. Cancer Res 67(15):7055–7058. doi:10.1158/0008-5472.CAN-07-0905
Batista LF, Roos WP, Christmann M et al (2007) Differential sensitivity of malignant glioma cells to methylating and chloroethylating anticancer drugs: p53 determines the switch by regulating xpc, ddb2, and DNA double-strand breaks. Cancer Res 67(24):11886–11895. doi:10.1158/0008-5472.CAN-07-2964
Natsume A, Wakabayashi T, Ishii D et al (2008) A combination of IFN-beta and temozolomide in human glioma xenograft models: implication of p53-mediated MGMT downregulation. Cancer Chemother Pharmacol 61(4):653–659. doi:10.1007/s00280-007-0520-x
Plowman J, Waud WR, Koutsoukos SD et al (1994) Preclinical antitumor activity of temozolomide in mice: efficacy against human brain tumor xenografts and synergism with 1, 3-bis(2-chloroethyl)-1-nitrosourea. Cancer Res 54(14):3793–3799
Miyagami M, Katayama Y (2005) Angiogenesis of glioma: evaluation of ultrastructural characteristics of microvessels and tubular bodies (Weibel–Palade) in endothelial cells and immunohistochemical findings with VEGF and p53 protein. Med Mol Morphol 38:36–42. doi:10.1007/s00795-004-0273-0
Pyrko P, Schönthal AH, Hofman FH, Chen TCC, Lee AS (2007) The unfolded protein response regulator GRP78/BiP as a novel target for increasing chemosensitivity in malignant gliomas. Cancer Res 67(20):9809–9816. doi:10.1158/0008-5472.CAN-07-0625
Hegi ME, Diserens AC, Gorlia T et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003. doi:10.1056/NEJMoa043331
Ma J, Waxman DJ (2008) Combination of antiangiogenesis with chemotherapy for more effective cancer treatment. Mol Cancer Ther 7(12):3670–3684. doi:10.1158/1535-7163.MCT-08-0715
Kang SG, Kim JS, Park K et al (2006) Combination celecoxib and temozolomide in C6 rat glioma orthotopic model. Oncol Rep 15(1):7–13
Riva M, Imbesi F, Beghi E, Galli C (2007) Temozolomide and thalidomide in the treatment of glioblastoma multiforme. Anticancer Res 27(2):1067–1071
Ku GY, Krol G, Ilson DH (2007) Successful treatment of leptomeningeal disease in colorectal cancer with a regimen of bevacizumab, temozolomide, and irinotecan. J Clin Oncol 25(13):e14–e16. doi:10.1200/JCO.2006.10.3317
Acknowledgments
We would like to thank the members of the Glioma Research Group at the Keck School of Medicine, University of Southern California for insightful discussions. This work was funded by the Whittier Foundation, USC/Norris Cancer Center and the Sounder Foundation.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Virrey, J.J., Golden, E.B., Sivakumar, W. et al. Glioma-associated endothelial cells are chemoresistant to temozolomide. J Neurooncol 95, 13–22 (2009). https://doi.org/10.1007/s11060-009-9891-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-009-9891-7