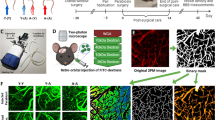
Kolmer cells (epiplexus macrophages) constitute the least studied population among the phagocytosing cells in the mammalian brain. We report here studies of the reactions of these cells to ischemic brain injury induced by brief termination of the blood supply in the basin of the middle cerebral artery. These studies used rats of spontaneously hypertensive strain SHR and normotensive WKY rats. Kolmer cells in normotensive WKY animals had small volumes of perinuclear cytoplasm with concentration of Iba-1 protein in the cortical layer and typical fine and sometimes tortuous processes. Moderate activation induced by increased arterial pressure in control SHR rats was accompanied by an increase in the volume of perinuclear cytoplasm and the appearance of numerous straight fine processes. Further activation induced by ischemia led to loss of processes, cells taking up a round shape. These structural rearrangements were accompanied by disappearance of the Iba-1 protein concentration gradient in the cytoplasm. This study also provided the first observation of the intranuclear accumulation of cytoplasmic Iba-1 protein in Kolmer cells, regardless of their activation, indicating that this protein is multifunctional within the cells. These results provide evidence that Kolmer cells are involve in the responses of the cerebral brain to injury.
Similar content being viewed by others
References
O. S. Alekseeva, O. V. Kirik, E. G. Gilerovich, and D. E. Korzhevskii, “Microglia of the Brain: Origin, Structure, Functions,” J. Evol. Biochem. Physiol., 55, 257–268 (2019).
T. Goldmann, P. Wieghofer, M. J. Jordao, et al., “Origin, fate and dynamics of macrophages at central nervous system interfaces,” Nat. Immunol., 17, No. 7, 797–805 (2016).
E. A. Ling, “Ultrastructure and origin of epiplexus cells in the telencephalic choroid plexus of postnatal rats studied by intravenous injection of carbon particles,” J. Anat, 129, No. 3, 479–492 (1979).
O. J. Tamega, L. F. Tirapelli, and S. Petroni, “Scanning electron microscopy study of the choroid plexus in the monkey (Cebus apella apella),” Arq. Neuropsiquiatr., 58, No. 3B, 820–825 (2000).
Y. Wan, Y. Hua, H. J. L. Garton, et al., “Activation of epiplexus macrophages in hydrocephalus caused by subarachnoid hemorrhage and thrombin,” CNS Neurosci. Ther., 25, No. 10, 1134–1141 (2019).
D. E. Korzhevskii and “The choroid plexus and structural organization of the blood-CSF barrier in humans,” Regionarn. Krovoobrashch. Mikrotsirkulyats., 2, No. 1, 5–14 (2003).
W. L. Maxwell, I. G. Hardy, C. Watt, et al., “Changes in the choroid plexus, responses by intrinsic epiplexus cells and recruitment from monocytes after experimental head acceleration injury in the non-human primate,” Acta Neuropathol., 84, No. 1, 78–84 (1992).
A. Peters and R. C. Swan, “The choroid plexus of the mature and aging rat: the choroidal epithelium,” Anat. Rec., 194, No. 3, 325–353 (1979).
A. Chamorro, A. Meisel, A. M. Planas, et al., “The immunology of acute stroke,” Nat. Dev. Neurol., 8, No. 7, 401–410 (2012).
C. Iadecola and J. Anrather, “The immunology of stroke: from mechanisms to translation,” Nat. Med., 17, No. 7, 796–808 (2011).
X. Y. Xiong, L. Liu, and Q. W. Yang, “Functions and mechanisms of microglia/macrophages in neuroinflammation and neurogenesis after stroke,” Prog. Neurobiol., 142, 23–44 (2016).
J. Pedragosa, A. Salas-Perdomo, M. Gallizioli, et al., “CNS-border associated macrophages respond to acute ischemic stroke attracting granulocytes and promoting vascular leakage,” Acta Neuropathol. Comm., 6, No. 1, 76 (2018).
M. Joukal, I. Klusakova, P. Solar, et al., “Cellular reactions of the choroid plexus induced by peripheral nerve injury,” Neurosci. Lett., 628, 73–77 (2016).
K. Okamoto and K. Aoki, “Development of a strain of spontaneously hypertensive rats,” Jpn. Circ. J., 27, 282–293 (1963).
D. A. Zhuravlev, “Models of arterial hypertension. Spontaneously hypertensive rats,” Arterial. Gipertenz., 15, No. 6, 721–723 (2009).
D. E. Korzhevskii, O. V. Kirik, A. E. Baisa, and T. D. Vlasov, “Simulation of unilateral ischemic injury to the striatal neurons inflicted by short-term occlusion of the middle cerebral artery,” Bull. Exp. Biol. Med., 147, No. 2, 255–256 (2009).
D. E. Korzhevskii, E. G. Sukhorukova, O. V. Kirik, and I. P. Grigorev, “Immunohistochemical demonstration of specific antigens in the human brain fixed in zinc-ethanol-formaldehyde,” Eur. J. Histochem., 59, No. 3, 233–237 (2015).
G. Paxinos and C. Watson, The Rat Brain in Stereotaxic Coordinates, Academic Press, San Diego (1998), 4th ed.
D. E. Korzhevskii and O. V. Kirik, “Brain microglia and microglial markers,” Neurosci. Behav. Physiol., 46, No. 3, 284–290 (2016).
M. D. Abramoff, P. J. Magalhaes, and S. J. Ram, “Image processing with ImageJ,” Biophotonics Internat., 11, No. 7, 36–42 (2004).
K. Ohsawa, Y. Imai, H. Kanazawa, et al., “Involment of Iba1 in membrane ruffling and phagocytosis of macrophages/microglia,” J. Cell Sci., 133, No. 17, 3073–3084 (2000).
K. Ohsawa, Y. Imai, Y. Sasaki, and S. Kohsaka, “Microglia/macrophages- specific protein Iba1 binds to fimbrin and enhances its actin-bundling activity,” J. Neurochem., 88, No. 4, 844–856 (2004).
H. Al-Sarraf and L. Philip, “Effect of hypertension on the integrity of blood brain and blood CSF barriers, cerebral blood flow and CSF secretion in the rat,” Brain Res., 975, No. 1–2, 179–188 (2003).
E. A. Ling, C. Y. Tseng, and W. C. Wong, “An electron microscopical study of the epiplexus and supraependymal cells in the prenatal rat brain following a maternal injection of 6-aminonicotinamide,” J. Anat, 140, 119–129 (1985)
C. Gu, X. Hao, J. Li, et al., “Effects of minocycline on epiplexus macrophage activation, choroid plexus injury and hydrocephalus development in spontaneous hypertensive rats,” J. Cereb. Blood Flow Metab., 39, No. 10, 1936–1948 (2019).
A. R. Patel, R. Ritzel, L. D. McCullough, and F. Liu, “Microglia and ischemic stroke: a double-edged sword,” Int. J. Physiol. Pathophysiol. Pharmacol., 5, No. 2, 73–90 (2013).
D. Ito, K. Tanaka, S. Suzuki, et al., “Enhanced expression of Iba1, ionized calcium-binding adapter molecule 1, after transient focal cerebral ischemia in rat brain,” Stroke, 32, No. 5, 1208–1215 (2001).
D. E. Korzhevskii, O. V. Kirik, E. G. Sukhorukova, and M. A. Syrtsova, “Microglia of the human substantia nigra,” Med. Akad. Zh., 14, No. 4, 68–72 (2014).
D. E. Korzhevskii, O. V. Kirik, O. S. Alekseeva, et al., “Intranuclear accumulation of Iba-1 protein in microgliocytes in the human brain,” Morfologiya, 149, No. 2, 73–76 (2016); Neurosci. Behav. Physiol., 47, No. 4, 435–437 (2017).
D. E. Korzhevskii, I. P. Grigor’ev, V. V. Gusel’nikova, et al., “Immunohistochemical markers for neurobiology,” Med. Akad. Zh., 19, No. 4, 7–24 (2019).
J. O. Schulze, C. Quedenau, and Y. Roske, “Structural and functional characterization of human Iba proteins,” FEBS J., 275, No. 18, 4627–4640 (2008).
L. M. Berglund, O. Kotova, P. Osmark, et al., “NFAT regulates the expression of AIF-1 and IRT-1: yin and yang splice variants of neointima formation and atherosclerosis,” Cardiovasc. Res., 93, No. 3, 414–423 (2012).
P. S. Subramaniam, M. M. Green, J. Larkin, et al., “Nuclear translocation of IFN-gamma is an intrinsic requirement for its biologic activity and can be driven by a heterologous nuclear localization sequence,” J. Interferon Cytokine Res., 21, No. 11, 951–959 (2001).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 107, No. 2, pp. 177–186, February, 2021.
Rights and permissions
About this article
Cite this article
Kirik, O.V., Tsyba, D.L., Alekseeva, O.S. et al. Changes in Kolmer Cells in SHR Rats after Cerebral Ischemia. Neurosci Behav Physi 51, 1148–1152 (2021). https://doi.org/10.1007/s11055-021-01174-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-021-01174-3