Abstract
Understanding and controlling the sintering behaviour of gold nanoparticles is important in the field of ligand-protected nanoparticles for their use as precursors for thin film fabrication. Lowering the temperature of the sintering event of gold nanoparticles by facilitating desorption of the ligand through oxidation can provide compatibility of sintered gold nanoparticle thin films onto heat-sensitive substrates. Here we examine the processes by which 1-butanethiol-protected gold nanoparticles sinter under an ozone-rich environment. Upon heating, an ozone-rich environment significantly reduces the temperature of the sintering event when compared to sintering under ambient conditions. At room temperature, exposure to an ozone-rich environment induces sintering over a period of 2.5 h. Upon exposure to ozone, the surface-bound butanethiol ligands are oxidised to 1-butanesulfonic acid which facilitates sintering.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sintering of gold nanoparticles (AuNPs) is an attractive technique to form continuous, electrically conducting thin films [1, 2]. Using this method, low-cost printable electronic devices such as thin-film transistors [2], field effect transistors [3], and contacts [2, 4] can be fabricated. Formulations that sinter AuNPs at relatively low temperatures provide great flexibility and enable AuNP films to be applied to low melting point polymers or other heat-sensitive substrates. AuNPs can sinter if there is sufficient energy to overcome the activation energy barrier provided by the stabilizing ligands [5]. One method to lower the activation energy barrier, and thus the temperature of the sintering event (TSE), is to oxidise the capping ligands, which facilitates desorption from the gold surface.
The bond between gold and the thiolate stabilizing ligand may be described as a gold-thiyl interaction (where thiyls are species with the RS· structure) [6]. These interactions can be degraded upon exposure to an oxidiser, to UV light, or at elevated temperatures [7,8,9,10,11,12]. Alkanethiolate self-assembled monolayers (SAMs) on gold have been shown to oxidise to alkanesulfinates and alkanesulfides under ambient conditions [10, 13,14,15]. Scanning tunnelling microscope images of air-oxidised decanethiol SAMs on Au(111) shows that after a two-week exposure to ambient conditions, some conversion to decanesulfonate occurs [16]. Density functional theory calculations revealed that under oxidising conditions (surface oxide or ozone) thiolate groups on Au (111) surfaces might form sulfoxide derivatives (R2S = O), with sulfinate (RS(= O)O−) and sulfonate (RS(= O)2O−) derivatives formed if active oxygens are further supplied [17].
Of particular relevance to the current work, exposure of thiol-bound SAMs on gold to ozone has been shown to oxidise the sulphur atoms [11, 18, 19]. With regard to AuNPs, ozone can reduce the affinity of thiol-based ligands to the gold core in supported AuNPs, allowing for the removal of the ligands by washing with water [20]. X-ray photoelectron spectroscopy (XPS) analysis of the ozone-treated AuNPs revealed that the sulphur atoms had been oxidised upon exposure [20]. Previous work has also utilised nitrogen dioxide to oxidise the thiolate stabilizing ligands of AuNPs which subsequently sintered at room temperature [5]. In this work, we examine the sintering of thiol-stabilised AuNPs using ozone, a common and readily generated gas. We show that in an ozone-rich environment, the ligands surrounding AuNPs are oxidised and significantly reduce TSE to produce gold films. Importantly, we investigate the organic profile of the desorbed ligand to determine the processes that occur upon sintering. These findings shed a new light on advancing the use of gold films for heat sensitive substrates.
Experimental
General
1-Butanethiol, Sodium 1-butanesulfonate, tetraoctylammonium bromide, sodium borohydride, methanol, acetonitrile, deuterated chloroform (CDCl3) and deuterated dimethyl sulfoxide (DMSO-d6) were purchased from Sigma-Aldrich and used as received. Toluene (ChemSupplyAustralia) and chloroform (Rowe Scientific) were used as received. 1-Butanesulfonic acid [21] and dibutyl disulfide [22] were prepared by literature procedures. Tetrachloroauric acid [23], and butanethiol-capped AuNPs (BT@AuNPs) [2] was prepared using literature procedures. The AuNPs were characterised by scanning electron microscopy (SEM), transmission electron microscopes (TEM) and proton nuclear magnetic resonance spectroscopy (1H NMR). 1H NMR spectra were recorded using a Bruker NMR spectrometer operating at 400 MHz. Spectra were referenced using residual non-deuterated signals: DMSO-d6 (1H δ 2.49), CDCl3 (1H δ 7.26). Transmission electron microscopy (TEM) images were taken using a JEOL JEM-F200 FE-TEM operating at 200 kV and fitted with a Gatan Rio 1816–4 k × 4 k camera. The TEM samples were prepared by evaporating diluted nanoparticle solution on the carbon-coated copper grid. The images were analysed using ImageJ software (https://imagej.nih.gov/ij/). SEM was performed at facilities at Western Sydney University. A Zeiss Merlin field emission gun scanning electron microscope (FEGSEM) was utilised for imaging samples prepared on stubs. The FEGSEM was operated at 20 kV accelerating voltage in Hivac mode at a working distance of approximately 3 mm. Both secondary and in-lens secondary detectors were utilised for imaging. High-resolution mass-spectrometry (HRMS) was performed using an Agilent 6510 Q-TOF using flow injection and in positive ion mode for [M + H]+, or negative ion mode [M-H]− where specified.
Resistance measurements of AuNP films
Suspensions of BT@AuNPs in chloroform (0.5 mL of 10 mg/ml) were drop cast onto DropSens (Metrohm) interdigitated gold electrodes forming films of AuNPs of ~ 5 mm diameter, which were then heated within a modified Linkam THMS600 temperature control stage. A Linkam TMS 94 controller maintained a heating rate of 10 °C min−1 from room temperature to 250 °C. A Rigol DM3058E digital multimeter and a PT100 (RS PRO) RTD sensor, 2 mm × 5 mm Class B thermocouple measured the temperature on the gold electrode. A Rigol DM3068 digital multimeter (maximum resistance of 100 MΩ) measured the Electrical resistance of the electrode. A LabView program was used to interface with and control the multimeters, and to acquire the temperature and electrical resistance. Ozone was generated using a Hailea HLO-300 Ozonizer at 300 mg/h in a flow of 3.5 L/min.
Warning
Ozone was destructive to several electronic components including thermocouples when exposed for extended periods (up to 20 h).
Analysis of ozone-treated AuNPs
BT@AuNPs were placed in a 5-mL side arm tube attached to a condenser cooled to − 0.5 °C and fitted with a drying tube. The outflow from an ozone generator was passed through dry silica gel beads tightly packed in a condenser cooled to − 0.5 °C to remove moisture and then directed into the side arm tube for 48 h. Organic residues were then collected by rinsing the interior of the condenser and reaction tube first with CDCl3 (with sonication) and then DMSO-d6. The solutions were filtered through cellulose fibre (Kimwipe) to remove elemental gold and analysed using 1H NMR spectroscopy and HRMS.
Results and discussion
BT@AuNPs were synthesised using a two-phase Brust-Schiffrin method and had diameters of 3.4 (± 1.4) nm (measured using TEM, Figure S1).
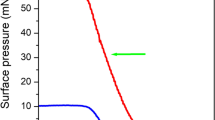
Films of AuNPs were prepared by drop-casting a suspension of BT@AuNPs in chloroform onto interdigitated gold electrodes. Upon heating at 10 °C/min in air, the films sintered at ~ 190 °C to form a conductive gold film. The sintering event is associated with a change in resistance from > 1 MΩ to < 100 Ω (Fig. 1). These results are consistent with our earlier studies on BT@AuNPs [24]. In contrast, heating of the films in an ozone-rich atmosphere caused the films to sinter at ~ 80 °C, which is significantly lower than the TSE of the AuNPs sintered in air.
The significant decrease in the TSE observed upon heating at 10 °C/min prompted experiments to examine the effect of exposure to ozone at room temperature. Films of metallic gold were formed from AuNP films exposed to a stream of ozone (Fig. 2). To probe this behaviour further, films of AuNPs were formed by drop-casting AuNP suspensions onto interdigitated electrodes and the resistance was measured upon exposure to ozone. Figure 3 shows the resistance of BT@AuNPs at room temperature upon exposure to air and an ozone-rich environment over 15 h. In our hands, and as reported by others [25], such AuNPs are stable and do not sinter over periods of at least months. Under an atmosphere of air, the resistance remained stable at 2.5 MΩ. In contrast, under an ozone atmosphere, the resistance of the BT@AuNPs decreased markedly to ~ 300 Ω after ~ 2.5 h. The slightly greater resistance of the room temperature sintered AuNPs (300 Ω) compared to the thermally sintered AuNPs (~ 15 Ω) may be attributed to residual organic material remaining after the decomposition of butanethiol (see below), leading to a less dense film [26].
Low temperature sintering has been reported in our earlier work using a chemically synthesised oxidant but the nature of the reactions leading to the sintering event was not explored [5]. Here we examine the residue surrounding the gold film formed after exposure to the ozone atmosphere using 1H NMR spectroscopy and HRMS data. After the reaction with ozone, which induced sintering, the reaction vessel was rinsed with CDCl3 and then DMSO-d6. The CDCl3 fraction contained very little material of which none could be characterised by 1H NMR spectroscopy. The DMSO-d6 fraction contained a significant amount of organic material.
The 1H NMR spectrum of the DMSO-d6 fraction contained signals that are consistent with the spectrum of 1-butanesulfonic acid (Figure S3). A triplet at 0.85 ppm is assigned to the CH3 group, a sextet and a quintet at 1.31 and 1.54 ppm, respectively, are assigned to the two central CH2 groups. A triplet at 2.43 ppm is assigned to the CH2 group adjacent to the SO3H group and is identical to a spectrum of synthesised 1-butanesulfonic acid. Importantly, we found no evidence of dibutyl disulfide formation upon ozone-induced sintering in either the 1H NMR spectrum (Figure S4) or HRMS. Upon sintering in air, hydrogen, nitrogen or argon atmospheres, surface-bound thiyl ligands leave exclusively as their corresponding disulfide compounds [24]. Other possible sulfur-containing compounds such as 1-butanethiol were excluded by comparison of the 1H NMR spectra (Figure S4).
The CDCl3 and DMSO-d6 fractions were both examined by mass spectrometry in positive and negative ion modes. In negative ion mode, the major peak was observed at m/z 137.028, which corresponds to the sulfonate ion with formula CH3(CH2)3SO3−. Other oxygen- and sulphur-containing compounds such as sulfoxide and sulfinates (105 and 121 m/z respectively) were not detected. These findings are consistent with work examining UV-induced photooxidation of thiol SAMs on gold (over various time periods) using XPS where only the corresponding sulfonate ions were detected [27].
Considering the resistance data together with post-sintering analysis, it is apparent that when the BT@AuNPs are exposed to an ozone-rich environment, the butanethiol ligands undergo oxidation to butanesulfonic acid thus facilitating desorption of the ligand from the gold surface and inducing the sintering event (Fig. 4).
SEM images were collected of thermally induced and ozone-induced sintered BT@AuNPs (Fig. 5) as well as pristine BT@AuNPs (Figure S2). The SEM images of the thermally induced, sintered gold films are consistent with previous reports, showing densification and large grain barriers [24]. SEM images of the room temperature ozone-induced sintered gold films show finer grain size with an agglomeration of particles and some residual material. Grains ranging from 600 to 1000 nm are apparent in the thermally induced sintered structures while the ozone-induced films have smaller grains ≤ 200 nm. In both cases, none of the original AuNPs were observed.
Conclusion
Conductive gold films have been prepared by sintering BT@AuNPs under an ozone-rich atmosphere. Resistance measurements of BT@AuNPs showed that exposure to the ozone-containing atmosphere during heating significantly reduced the TSE compared to sintering under ambient conditions by ~ 80 °C. Furthermore, electrically conductive gold films were formed at room temperature when BT@AuNPs were exposed to ozone for ~ 2.5 h.
Examination of the AuNPs post-sintering revealed that the butanethiyl ligands undergo oxidation to form the corresponding butanesulfonic acid, which is a poor stabilizing ligand. We found no evidence of dibutyl disulfide (the major product of sintering under ambient conditions) in the post-sintering residue, indicating that the oxidation process is further promoted by ozone. The gold films prepared by the new room temperature ozone-induced sintering process showed a different morphology to those sintered by thermal activation under ambient conditions (observed by SEM) with the former producing finer grain sizes.
Data availability
No datasets were generated or analysed during the current study.
References
Bishop PT, Ashfield LJ, Berzins A, Boardman A, Buche V, Cookson J, Gordon RJ, Salcianu C, Sutton PA (2010) Printed gold for electronic applications. Gold Bull 43:181–188
Wu Y, Li Y, Liu P, Gardner S, Ong BS (2006) Studies of gold nanoparticles as precursors to printed conductive features for thin-film transistors. Chemistry Mat 18:4627–4632
Presnova G, Presnov D, Krupenin V, Grigorenko V, Trifonov A, Andreeva I, Ignatenko O, Egorov A, Rubtsova M (2017) Biosensor based on a silicon nanowire field-effect transistor functionalized by gold nanoparticles for the highly sensitive determination of prostate specific antigen. Biosens Bioelectron 88:283–289
Huang D, Liao F, Molesa S, Redinger D, Subramanian V (2003) Plastic-compatible low resistance printable gold nanoparticle conductors for flexible electronics. J Electrochem Soc 150:G412–G417
Coutts MJ, Cortie MB, Ford MJ, McDonagh AM (2009) Rapid and controllable sintering of gold nanoparticle inks at room temperature using a chemical agent. J Phys Chem C 113:1325–1328
Reimers JR, Ford MJ, Halder A, Ulstrup J, Hush NS (2016) Gold surfaces and nanoparticles are protected by Au(0)-thiyl species and are destroyed when Au(I)-thiolates form. Proc Natl Acad Sci USA 113:E1424–E1433
Sotthewes K, Kap Ö, Wu H, Thompson D, Huskens J, Zandvliet HJW (2018) Ordering of Air-Oxidized Decanethiols on Au(111). J Phys Chem C 122(15):8430–8436
Garrell RL, Chadwick JE, Severance DL, McDonald NA, Myles DC (1995) Adsorption of sulfur containing molecules on gold: the effect of oxidation on monolayer formation and stability characterized by experiments and theory. J Am Chem Soc 117(46):11563–11571
Horn AB, Russell DA, Shorthouse LJ, Simpson TRE (1996) Ageing of alkanethiol self-assembled monolayers. J Chem Soc, Faraday Trans 92(23):4759–4762
Scott JR, Baker LS, Everett WR, Wilkins CL, Fritsch I (1997) Laser desorption fourier transform mass spectrometry exchange studies of air-oxidized alkanethiol self-assembled monolayers on gold. Anal Chem 69(14):2636–2639
Schoenfisch MH, Pemberton JE (1998) Air stability of alkanethiol self-assembled monolayers on silver and gold surfaces. J Am Chem Soc 120(18):4502–4513
Willey TM, Vance AL, van Buuren T, Bostedt C, Terminello LJ, Fadley CS (2005) Rapid degradation of alkanethiol-based self-assembled monolayers on gold in ambient laboratory conditions. Surf Sci 576(1):188–196
Li Y, Huang J, McIver RT Jr, Hemminger JC (1992) Characterization of thiol self-assembled films by laser desorption Fourier transform mass spectrometry. J Am Chem Soc 114(7):2428–2432
Tarlov MJ, Newman JG (1992) Static secondary ion mass spectrometry of self-assembled alkanethiol monolayers on gold. Langmuir 8(5):1398–1405
Burroughs JA, Hanley L (1993) Laser desorption ion trap mass spectrometry of self-assembled monolayers. J Am Soc Mass Spectrom 4(12):968–970
Kap Ö, Kabanov N, Tsvetanova M, Varlikli C, Klavsyuk AL, Zandvliet HJW, Sotthewes K (2020) Structural Stability of Physisorbed Air-Oxidized Decanethiols on Au(111). J Phys Chem C 124(22):11977–11984
Jang YH, Goddard WA III (2010) Oxidation of Thiol Anchor Groups in Molecular Junction Devices: A Density Functional Theory Study. J Phys Chem C 114(10):4646–4651
Poirier GE, Herne TM, Miller CC, Tarlov MJ (1999) Molecular-Scale Characterization of the Reaction of Ozone with Decanethiol Monolayers on Au(111). J Am Chem Soc 121(41):9703–9711
Zhang Y, Terrill RH, Tanzer TA, Bohn PW (1998) Ozonolysis is the primary cause of UV photooxidation of alkanethiolate monolayers at low irradiance. J Am Chem Soc 120(11):2654–2655
Elliott EW III, Glover RD, Hutchison JE (2015) Removal of thiol ligands from surface-confined nanoparticles without particle growth or desorption. ACS Nano 9(3):3050–3059
Smith K, Hou D (1996) A general and efficient method for the preparation of organic sulfonic acids by insertion of sulfur trioxide into the metal−carbon bond of organolithiums. J Org Chem 61(4):1530–1532
Shaabani A, Tavasoli-Rad F, Lee DG (2005) Potassium permanganate oxidation of organic compounds. Synth Commun 35(4):571–580
King SR, Massicot J, McDonagh A (2015) A straightforward route to tetrachloroauric acid from gold metal and molecular chlorine for nanoparticle synthesis. Metals 5:1454–1461
Summers PK, Angeloski A, Wuhrer R, Cortie MB, McDonagh AM (2023) The fate of organic species upon sintering of thiol-stabilised gold nanoparticles under different atmospheric conditions. Phys Chem Chem Phys 25(10):7170–7175
Brust M, Fink J, Bethell D, Schiffrin DJ, Kiely C (1995) Synthesis and reactions of functionalised gold nanoparticles. J Chem Soc Chem Commun 16:1655–1656
King SR, Shimmon S, Gentle AR, Westerhausen MT, Dowd A, McDonagh AM (2016) Remarkable thermal stability of gold nanoparticles functionalised with ruthenium phthalocyanine complexes. Nanotech 27(21):215702
Hutt DA, Leggett GJ (1996) Influence of adsorbate ordering on rates of UV photooxidation of self-assembled monolayers. J Phys Chem 100(16):6657–6662
Acknowledgements
We acknowledge the Advanced Materials Characterisation Facility (AMCF) of Western Sydney University for access to its instrumentation and staff. In particular, we thank Dr. Laurel George and Dr. Daniel Fanna. We thank Dr Thomas Lockwood (UTS) for assistance with mass spectrometry and the microstructural analysis unit (UTS) for assistance with TEM.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions This research is supported by an Australian Government Research Training Program Scholarship.
Author information
Authors and Affiliations
Contributions
Summers, P. K. and McDonagh A. M. contributed to the conception and design of the work. Summers P. K. performed the synthesis, sintering, NMR, SEM and TEM experiments and contributed to manuscript preparation. Wuhrer, R. assisted with SEM experiments and manuscript preparation. McDonagh A. M. contributed to synthesis and manuscript preparation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Summers, P.K., Wuhrer, R. & McDonagh, A.M. Electrically conductive gold films formed by sintering of gold nanoparticles at room temperature initiated by ozone. J Nanopart Res 26, 97 (2024). https://doi.org/10.1007/s11051-024-06012-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-024-06012-4