Abstract
Background
Alzheimer’s disease (AD), the most common neurodegenerative disease, is charactered by these accepted pathological features, such as β-amyloid (Aβ) plaques outside the neurons and neurofibrillary tangles inside the neurons. In recent years, several studies have demonstrated that pyroptosis is associated with the development of AD process. However, whether Aβ25–35 induces pyroptosis is still unclear. Thioredoxin-1 (Trx-1), an intracellular multifunctional protein, showed neuroprotective roles by inhibiting the neurotoxicity of Aβ, attenuating the apoptosis of brain neurons and improving the spatial learning and memory ability in AD models. Whether Trx-1 could inhibit pyroptosis in AD needs to be further investigated.
Methods and results
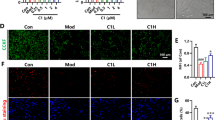
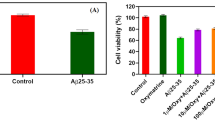
In the present study, MTT assay was employed to detected the viability. Western blotting was employed to detect the protein levels. Enzyme linked immunosorbent assay was used to examine the intracellular and extracellular levels of IL-18 and IL-1β. Chronic Aβ25–35 treatment remarkedly compromised the viability of PC12 cells, increased the expression of NOD-like receptor pyrin domain containing 1 (NLRP-1), caspase-1 and gasdermin D (GSDMD), and promoted the extracellular release of interleukin (IL)-18 and IL-1β. Simultaneously, Aβ25–35 treatment also significantly reduced the intracellular protein levels of Trx-1. Pharmacological inhibition of Trx-1 activity further decreased the cell viability, activated the NLRP-1/caspase-1/GSDMD pyroptotic pathway, and exacerbated the extracellular release of IL-18 and IL-1β.
Conclusions
These data suggest that Trx-1 may play a potential inhibitory effect on Aβ25–35-induced pyroptosis.




Similar content being viewed by others
References
Jia L, Quan M, Fu Y, Zhao T, Li Y, Wei C, Tang Y, Qin Q, Wang F, Qiao Y, Shi S, Wang YJ, Du Y, Zhang J, Zhang J, Luo B, Qu Q, Zhou C, Gauthier S, Jia J, Group for the Project of Dementia Situation in C (2020) Dementia in China: epidemiology, clinical management, and research advances. Lancet Neurol 19:81–92. https://doi.org/10.1016/S1474-4422(19)30290-X
Li J, Du Q, Li N, Du S, Sun Z (2021) Alpiniae oxyphyllae Fructus and Alzheimer’s disease: an update and current perspective on this traditional Chinese medicine. Biomed Pharmacother 135:111167. https://doi.org/10.1016/j.biopha.2020.111167
Harrison TM, Du R, Klencklen G, Baker SL, Jagust WJ (2021) Distinct effects of beta-amyloid and tau on cortical thickness in cognitively healthy older adults. Alzheimers Dement 17:1085–1096. doi: https://doi.org/10.1002/alz.12249
Xia Y, Wang ZH, Zhang J, Liu X, Yu SP, Ye KX, Wang JZ, Ye K, Wang XC (2020) C/EBPbeta is a key transcription factor for APOE and preferentially mediates ApoE4 expression in Alzheimer’s disease. Mol Psychiatry doi. https://doi.org/10.1038/s41380-020-00956-4
Edler MK, Sherwood CC, Meindl RS, Hopkins WD, Ely JJ, Erwin JM, Mufson EJ, Hof PR, Raghanti MA (2017) Aged chimpanzees exhibit pathologic hallmarks of Alzheimer’s disease. Neurobiol Aging 59:107–120. doi: https://doi.org/10.1016/j.neurobiolaging.2017.07.006
Calsolaro V, Edison P (2016) Neuroinflammation in Alzheimer’s disease: current evidence and future directions. Alzheimers Dement 12:719–732. https://doi.org/10.1016/j.jalz.2016.02.010
Shi J, Gao W, Shao F (2017) Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem Sci 42:245–254. https://doi.org/10.1016/j.tibs.2016.10.004
Xue Y, Enosi Tuipulotu D, Tan WH, Kay C, Man SM (2019) Emerging activators and regulators of inflammasomes and pyroptosis. Trends Immunol 40:1035–1052. https://doi.org/10.1016/j.it.2019.09.005
Wang K, Sun Q, Zhong X, Zeng M, Zeng H, Shi X, Li Z, Wang Y, Zhao Q, Shao F, Ding J (2020) Structural mechanism for GSDMD targeting by autoprocessed caspases in pyroptosis. Cell 180:941-955e20. https://doi.org/10.1016/j.cell.2020.02.002
Ding J, Wang K, Liu W, She Y, Sun Q, Shi J, Sun H, Wang DC, Shao F (2016) Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 535:111–116. doi: https://doi.org/10.1038/nature18590
Tan MS, Tan L, Jiang T, Zhu XC, Wang HF, Jia CD, Yu JT (2014) Amyloid-beta induces NLRP1-dependent neuronal pyroptosis in models of Alzheimer’s disease. Cell Death Dis 5:e1382. doi: https://doi.org/10.1038/cddis.2014.348
Zhang T, Guan B, Tan S, Zhu H, Ren D, Li R, Xiao L (2021) Bushen huoxue acupuncture inhibits NLRP1 inflammasome-mediated neuronal pyroptosis in SAMP8 mouse model of Alzheimer’s disease. Neuropsychiatr Dis Treat 17:339–346. https://doi.org/10.2147/NDT.S279304
Shen H, Han C, Yang Y, Guo L, Sheng Y, Wang J, Li W, Zhai L, Wang G, Guan Q (2021) Pyroptosis executive protein GSDMD as a biomarker for diagnosis and identification of Alzheimer’s disease. Brain Behav 11:e02063. doi: https://doi.org/10.1002/brb3.2063
Zhao N, Sun C, Zheng M, Liu S, Shi R (2019) Amentoflavone suppresses amyloid beta1-42 neurotoxicity in Alzheimer’s disease through the inhibition of pyroptosis. Life Sci 239:117043. https://doi.org/10.1016/j.lfs.2019.117043
Han C, Yang Y, Guan Q, Zhang X, Shen H, Sheng Y, Wang J, Zhou X, Li W, Guo L, Jiao Q (2020) New mechanism of nerve injury in Alzheimer’s disease: beta-amyloid-induced neuronal pyroptosis. J Cell Mol Med 24:8078–8090. https://doi.org/10.1111/jcmm.15439
Kaminsky YG, Marlatt MW, Smith MA, Kosenko EA (2010) Subcellular and metabolic examination of amyloid-beta peptides in Alzheimer disease pathogenesis: evidence for Abeta(25-35). Exp Neurol 221:26–37. https://doi.org/10.1016/j.expneurol.2009.09.005
Zhou J, Wang C, Wu J, Fukunaga A, Cheng Z, Wang J, Yamauchi A, Yodoi J, Tian H (2020) Anti-allergic and anti-inflammatory effects and molecular mechanisms of thioredoxin on respiratory system diseases. Antioxid Redox Signal 32:785–801. https://doi.org/10.1089/ars.2019.7807
Granger DN, Kvietys PR (2015) Reperfusion injury and reactive oxygen species: The evolution of a concept. Redox Biol 6:524–551. https://doi.org/10.1016/j.redox.2015.08.020
Zeng XS, Jia JJ, Ma LF (2015) Gensenoside Rb1 protects rat PC12 cells from oxidative stress-induced endoplasmic reticulum stress: the involvement of thioredoxin-1. Mol Cell Biochem 410:239–246. https://doi.org/10.1007/s11010-015-2557-1
Masutani H, Bai J, Kim YC, Yodoi J (2004) Thioredoxin as a neurotrophic cofactor and an important regulator of neuroprotection. Mol Neurobiol 29:229–242. https://doi.org/10.1385/MN:29:3:229
Chen W, Zeng X, Luo F, Lv T, Zhou X, Bai J (2014) The decreased expression of thioredoxin-1 in brain of mice with experimental autoimmune myasthenia gravis. Neuromuscul Disord 24:726–735. https://doi.org/10.1016/j.nmd.2014.05.001
Zeng XS, Zhou XS, Luo FC, Jia JJ, Qi L, Yang ZX, Zhang W, Bai J (2014) Comparative analysis of the neuroprotective effects of ginsenosides Rg1 and Rb1 extracted from Panax notoginseng against cerebral ischemia. Can J Physiol Pharmacol 92:102–108. doi: https://doi.org/10.1139/cjpp-2013-0274
Jia JJ, Geng WS, Wang ZQ, Chen L, Zeng XS (2019) The role of thioredoxin system in cancer: strategy for cancer therapy. Cancer Chemother Pharmacol 84:453–470. https://doi.org/10.1007/s00280-019-03869-4
Zeng XS, Geng WS, Wang ZQ, Jia JJ (2020) Morphine addiction and oxidative stress: the potential effects of thioredoxin-1. Front Pharmacol 11:82. https://doi.org/10.3389/fphar.2020.00082
Zeng XS, Jia JJ, Kwon Y, Wang SD, Bai J (2014) The role of thioredoxin-1 in suppression of endoplasmic reticulum stress in Parkinson disease. Free Radic Biol Med 67:10–18. https://doi.org/10.1016/j.freeradbiomed.2013.10.013
Jia J, Zeng X, Xu G, Wang Z (2021) The Potential Roles of Redox Enzymes in Alzheimer’s Disease: Focus on Thioredoxin. ASN Neuro 13:1759091421994351. doi: https://doi.org/10.1177/1759091421994351
Wang CY, Xu Y, Wang X, Guo C, Wang T, Wang ZY (2019) Dl-3-n-butylphthalide inhibits NLRP3 inflammasome and mitigates Alzheimer’s-like pathology via Nrf2-TXNIP-TrX axis. Antioxid Redox Signal 30:1411–1431. https://doi.org/10.1089/ars.2017.7440
Lane CA, Hardy J, Schott JM (2018) Alzheimer’s disease. Eur J Neurol 25:59–70. doi: https://doi.org/10.1111/ene.13439
Hodson R (2018) Alzheimer’s disease. Nature 559:S1. https://doi.org/10.1038/d41586-018-05717-6
Jeong S (2017) Molecular and cellular basis of neurodegeneration in Alzheimer’s disease. Mol Cells 40:613–620. https://doi.org/10.14348/molcells.2017.0096
Yang L, Guo Y, Huang M, Wu X, Li X, Chen G, Li Y, Bai J (2018) Thioredoxin-1 protects spinal cord from demyelination induced by methamphetamine through suppressing endoplasmic reticulum stress and inflammation. Front Neurol 9:49. https://doi.org/10.3389/fneur.2018.00049
Lovell MA, Xie C, Gabbita SP, Markesbery WR (2000) Decreased thioredoxin and increased thioredoxin reductase levels in Alzheimer’s disease brain. Free Radic Biol Med 28:418–427. https://doi.org/10.1016/s0891-5849(99)00258-0
Akterin S, Cowburn RF, Miranda-Vizuete A, Jimenez A, Bogdanovic N, Winblad B, Cedazo-Minguez A (2006) Involvement of glutaredoxin-1 and thioredoxin-1 in beta-amyloid toxicity and Alzheimer’s disease. Cell Death Differ 13:1454–1465. https://doi.org/10.1038/sj.cdd.4401818
Acknowledgements
This study was supported by the Natural Science Foundation of Zhejiang Province (LQ22H090003), Natural Science Foundation of Henan Province (202300410332), Sci-Tech Planning Project of Jiaxing (2021AY30001) and Research Foundation for Advanced Talents of Jiaxing University (CD70520017 and CD70520018).
Author information
Authors and Affiliations
Contributions
X-SZ and J-JJ designed the article contents. J-JJ, X-HZ and G-TX performed the experiments and analyzed the data. X-SZ and LL wrote the original paper. X-SZ and LL revised the paper. All authors reviewed the paper and approved the submitted version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics Approval and Consent to Participate
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jia, J., Zhang, X., Xu, G. et al. Thioredoxin-1 inhibits amyloid-β25–35-induced activation of NLRP1/caspase-1/GSDMD pyroptotic pathway in PC12 cells. Mol Biol Rep 49, 3445–3452 (2022). https://doi.org/10.1007/s11033-022-07177-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-022-07177-8