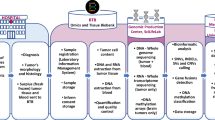
Abstract
Over the past decade we have witnessed a rapid increase in our understanding of the molecular characteristics of pediatric central nervous system (CNS) tumors. Studies that utilize genomic sequencing have revealed a heterogeneous group of genetic drivers in pediatric CNS tumors including point mutations, gene fusions, and copy number alterations. This manuscript provides an overview of somatic genomic alterations in the most common pediatric CNS tumors including low grade gliomas, high grade gliomas, medulloblastomas, and ependymomas. Additionally, we will discuss the need and opportunity for genomic and clinical data sharing through the children’s brain tumor network and other international initiatives.

Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Collins FS, McKusick VA (2001) Implications of the Human Genome Project for medical science. JAMA 285(5):540–544
Ashley EA (2016) Towards precision medicine. Nat Rev Genet 17(9):507–522
DeWitt JC, Mock A, Louis DN (2017) The 2016 WHO classification of central nervous system tumors: what neurologists need to know. Curr Opin Neurol 30(6):643–649
Louis DN et al (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131(6):803–820
Louis DN et al (2021) The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol 23(8):1231–1251
Ater JL et al (2012) Randomized study of two chemotherapy regimens for treatment of low-grade glioma in young children: a report from the Children’s Oncology Group. J Clin Oncol 30(21):2641–2647
Margol AS et al (2018) A comparative analysis of clinicopathological features and survival among early adolescents/young adults and children with low-grade glioma: a report from the Children’s Oncology Group. J Neurooncol 140(3):575–582
Zhang J et al (2013) Whole-genome sequencing identifies genetic alterations in pediatric low-grade gliomas. Nat Genet 45(6):602–612
Jones DT et al (2009) Oncogenic RAF1 rearrangement and a novel BRAF mutation as alternatives to KIAA1549:BRAF fusion in activating the MAPK pathway in pilocytic astrocytoma. Oncogene 28(20):2119–2123
Cin H et al (2011) Oncogenic FAM131B-BRAF fusion resulting from 7q34 deletion comprises an alternative mechanism of MAPK pathway activation in pilocytic astrocytoma. Acta Neuropathol 121(6):763–774
Horbinski C (2012) Something old and something new about molecular diagnostics in gliomas. Surg Pathol Clin 5(4):919–939
Lassaletta A et al (2017) Therapeutic and Prognostic Implications of BRAF V600E in Pediatric Low-Grade Gliomas. J Clin Oncol 35(25):2934–2941
Del Bufalo F et al (2018) BRAF V600E Inhibitor (Vemurafenib) for BRAF V600E Mutated Low Grade Gliomas. Front Oncol 8:526
Hargrave DR et al (2019) Efficacy and safety of dabrafenib in pediatric patients with BRAF V600 mutation–positive relapsed or refractory low-grade glioma: results from a phase I/IIa study. Clin Cancer Res 25(24):7303–7311
Maraka S, Janku F (2018) BRAF alterations in primary brain tumors. Discov Med 26(141):51–60
Fangusaro J et al (2019) Selumetinib in paediatric patients with BRAF-aberrant or neurofibromatosis type 1-associated recurrent, refractory, or progressive low-grade glioma: a multicentre, phase 2 trial. Lancet Oncol 20(7):1011–1022
Karajannis MA et al (2014) Phase II study of sorafenib in children with recurrent or progressive low-grade astrocytomas. Neuro Oncol 16(10):1408–1416
Sievert AJ et al (2013) Paradoxical activation and RAF inhibitor resistance of BRAF protein kinase fusions characterizing pediatric astrocytomas. Proc Natl Acad Sci USA 110(15):5957–5962
Wright KD, Fine ZM, Aspri E, Kieran T, Chi MW (2018) S, LGG-26. Type II braf inhibitor TAK-580 shows promise for upcoming clinal trial as evidenced by single patient IND study. Neuro Oncol 20(2):i110
Johnson A et al (2017) Comprehensive Genomic Profiling of 282 Pediatric Low- and High-Grade Gliomas Reveals Genomic Drivers, Tumor Mutational Burden, and Hypermutation Signatures. Oncologist 22(12):1478–1490
Helfferich J et al (2016) Neurofibromatosis type 1 associated low grade gliomas: A comparison with sporadic low grade gliomas. Crit Rev Oncol Hematol 104:30–41
Mackay A et al (2018) Molecular, Pathological, Radiological, and Immune Profiling of Non-brainstem Pediatric High-Grade Glioma from the HERBY Phase II Randomized Trial. Cancer Cell 33(5):829–842 e5
Hargrave D, Bartels U, Bouffet E (2006) Diffuse brainstem glioma in children: critical review of clinical trials. Lancet Oncol 7(3):241–248
Mackay A et al (2017) Integrated molecular meta-analysis of 1,000 pediatric high-grade and diffuse intrinsic pontine glioma. Cancer Cell 32(4):520-537 e5
Sturm D et al (2012) Hotspot mutations in H3F3A and IDH1 define distinct epigenetic and biological subgroups of glioblastoma. Cancer Cell 22(4):425–437
Schwartzentruber J et al (2012) Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 482(7384):226–231
Wisoff JH et al (1998) Current neurosurgical management and the impact of the extent of resection in the treatment of malignant gliomas of childhood: a report of the Children’s Cancer Group trial no. CCG-945. J Neurosurg 89(1):52–59
Cohen KJ et al (2011) Temozolomide in the treatment of high-grade gliomas in children: a report from the Children’s Oncology Group. Neuro Oncol 13(3):317–323
Williams MJ et al (2017) Therapeutic Targeting of Histone Modifications in Adult and Pediatric High-Grade Glioma. Front Oncol 7:45
Anastas JN et al (2019) Re-programing Chromatin with a Bifunctional LSD1/HDAC Inhibitor Induces Therapeutic Differentiation in DIPG. Cancer Cell 36(5):528–544 e10
Capdevielle C et al (2020) HDAC inhibition induces expression of scaffolding proteins critical for tumor progression in pediatric glioma: focus on EBP50 and IRSp53. Neuro Oncol 22(4):550–562
Mayr L et al (2020) Cerebrospinal fluid penetration and combination therapy of entrectinib for disseminated ROS1/NTRK-fusion positive pediatric high-grade glioma. J Personal Med 10(4):290
Gambella A et al (2020) NTRK fusions in central nervous system tumors: a rare, but worthy target. Int J Mol Sci 21(3):753
Torre M et al (2020) Molecular and clinicopathologic features of gliomas harboring NTRK fusions. Acta Neuropathol Commun 8(1):107
Korshunov A et al (2015) Integrated analysis of pediatric glioblastoma reveals a subset of biologically favorable tumors with associated molecular prognostic markers. Acta Neuropathol 129(5):669–678
Souweidane MM et al (2018) Convection-enhanced delivery for diffuse intrinsic pontine glioma: a single-centre, dose-escalation, phase 1 trial. Lancet Oncol 19(8):1040–1050
Cacciotti C et al (2020) Immune checkpoint inhibition for pediatric patients with recurrent/refractory CNS tumors: a single institution experience. J Neurooncol 149(1):113–122
Guerreiro Stucklin AS et al (2019) Alterations in ALK/ROS1/NTRK/MET drive a group of infantile hemispheric gliomas. Nat Commun 10(1):4343
Clarke M et al (2020) Infant High-Grade Gliomas Comprise Multiple Subgroups Characterized by Novel Targetable Gene Fusions and Favorable Outcomes. Cancer Discov 10(7):942–963
Marinoff AE et al (2017) Rethinking childhood ependymoma: a retrospective, multi-center analysis reveals poor long-term overall survival. J Neurooncol 135(1):201–211
Pajtler KW et al (2015) Molecular Classification of Ependymal Tumors across All CNS Compartments, Histopathological Grades, and Age Groups. Cancer Cell 27(5):728–743
Arabzade A et al (2021) ZFTA-RELA Dictates Oncogenic Transcriptional Programs to Drive Aggressive Supratentorial Ependymoma. Cancer Discov 11(9):2200–2215
Pajtler KW et al (2017) The current consensus on the clinical management of intracranial ependymoma and its distinct molecular variants. Acta Neuropathol 133(1):5–12
Northcott PA et al (2012) Molecular subgroups of medulloblastoma. Expert Rev Neurother 12(7):871–884
Cavalli FMG et al (2017) Intertumoral heterogeneity within medulloblastoma subgroups. Cancer Cell 31(6):737-754 e6
Kumar R, Liu APY, Northcott PA (2019) Medulloblastoma genomics in the modern molecular era. Brain Pathol 30(3):679–690
Yeo KK et al (2019) Prognostic significance of molecular subgroups of medulloblastoma in young children receiving irradiation-sparing regimens. J Neurooncol 145(2):375–383
Cho YJ et al (2011) Integrative genomic analysis of medulloblastoma identifies a molecular subgroup that drives poor clinical outcome. J Clin Oncol 29(11):1424–1430
Ellison DW et al (2011) Medulloblastoma: clinicopathological correlates of SHH, WNT, and non-SHH/WNT molecular subgroups. Acta Neuropathol 121(3):381–396
Ellison DW et al (2011) Definition of disease-risk stratification groups in childhood medulloblastoma using combined clinical, pathologic, and molecular variables. J Clin Oncol 29(11):1400–1407
Fattet S et al (2009) Beta-catenin status in paediatric medulloblastomas: correlation of immunohistochemical expression with mutational status, genetic profiles, and clinical characteristics. J Pathol 218(1):86–94
Aldosari N et al (2002) MYCC and MYCN oncogene amplification in medulloblastoma. A fluorescence in situ hybridization study on paraffin sections from the Children’s Oncology Group. Arch Pathol Lab Med 126(5):540–544
Pfister S et al (2009) Outcome prediction in pediatric medulloblastoma based on DNA copy-number aberrations of chromosomes 6q and 17q and the MYC and MYCN loci. J Clin Oncol 27(10):1627–1636
Robinson GW et al (2015) Vismodegib exerts targeted efficacy against recurrent sonic hedgehog-subgroup medulloblastoma: results from phase ii pediatric brain tumor consortium studies PBTC-025B and PBTC-032. J Clin Oncol 33(24):2646–2654
Piñeros M et al (2021) Scaling Up the Surveillance of Childhood Cancer: A Global Roadmap. J Natl Cancer Inst 113(1):9–15
Ijaz H et al (2019) Pediatric high grade glioma resources from the children’s brain tumor tissue consortium (Cbttc). Neuro Oncol 22(1):163–165
Capper D et al (2018) DNA methylation-based classification of central nervous system tumours. Nature 555(7697):469–474
Vassal G et al (2014) Challenges for children and adolescents with cancer in Europe: the SIOP-Europe agenda. Pediatr Blood Cancer 61(9):1551–1557
Funding
No funding was used for completion of this study.
Author information
Authors and Affiliations
Contributions
All authors have contributed significantly to the design and writing of this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest or competing interests to disclose.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kaneva, K., Goldman, S. Review of the genomic landscape of common pediatric CNS tumors and how data sharing will continue to shape this landscape in the future. Mol Biol Rep 48, 7537–7544 (2021). https://doi.org/10.1007/s11033-021-06811-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06811-1