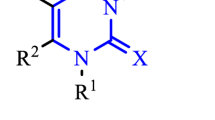
Abstract
Four series of heterocyclic compounds were obtained using Ugi-type multicomponent reactions (MCRs) with methyl 2-formylbenzoate as one of the starting materials. A facile and efficient one-pot procedure was suitable for all the MCRs under acidic conditions. This process provided access to four series of complex and potentially biologically active scaffolds.




Similar content being viewed by others
References
Dömling A (2006) Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem Rev 106:17–89. doi:10.1021/cr0505728
Dömling A, Ugi I (2000) Multicomponent reactions with isocyanides. Angew Chem Int Ed 39:3168–3210. doi:10.1002/1521-3773(20000915)39:18<3168:AID-ANIE3168>3.0.CO;2-U
Banfi L, Basso A, Guanti G, Riva R (2003) Passerini reaction—Amine Deprotection-Acyl Migration (PADAM): a convenient strategy for the solid-phase preparation of peptidomimetic compounds. Mol Divers 14:227–235. doi:10.1023/B:MODI.0000006778.42751.7f
Moliner FD, Bigatti M, Rosa CD, Banfi L, Riva R, Basso A (2014) Synthesis of triazolo-fused benzoxazepines and benzoxazepinones via Passerini reactions followed by 1,3-dipolar cycloadditions. Mol Divers 18:473–482. doi:10.1007/s11030-014-9530-x
Biggs-Houck JE, Younai A, Shaw JT (2010) Recent advances in multicomponent reactions for diversity-oriented synthesis. Curr Opin Chem Biol 14:371–382. doi:10.1016/j.cbpa.2010.03.003
Kalinski C, Lemoine H, Schmidt J, Burdack C, Kolb J, Umkehrer M, Ross G (2008) Multicomponent reactions as a powerful tool for generic drug synthesis. Synthesis 24:4007–4011. doi:10.1055/s-0028-1083239
Kalinski C, Umkehrer M, Weber L, Kolb J, Burdack C, Ross G (2010) On the industrial applications of MCRs: molecular diversity in drug discovery and generic drug synthesis. Mol Divers 14:513–522. doi:10.1007/s11030-010-9225-x
Hulme C, Gore V (2003) Multi-component reactions: emerging chemistry in drug discovery. From Xylocain to Crixivan. Curr Med Chem 10:51–80. doi:10.2174/0929867033368600
Dömling A, Wang W, Wang K (2012) Chemistry and biology of multicomponent reactions. Chem Rev 112:3083–3135. doi:10.1021/cr100233r
Harriman GCB (1997) Synthesis of small and medium sized 2,2-disubstituted lactams via the “intramolecular” three component Ugi reaction. Tetrahedron Lett 38:5591–5594. doi:10.1016/S0040-4039(97)01265-3
Zhang J, Jacobson A, Rusche JR, Herlihy W (1999) Unique structures generated by Ugi 3CC reactions using bifunctional starting materials containing aldehyde and carboxylic acid. J Org Chem 64:1074–1076. doi:10.1021/jo982192a
Hanusch-Kompa C, Ugi I (1998) Multi-component reactions 13: synthesis of \(\upgamma \)-lactams as part of a multiring system via Ugi-4-centre-3-component reaction. Tetrahedron Lett 39:2725–2728. doi:10.1016/S0040-4039(98)00428-6
Waller RW, Diorazio LJ, Taylor BA, Motherwell WB, Sheppard TD (2010) Isocyanide based multicomponent reactions of oxazolidines and related systems. Tetrahedron 66:6496–6507. doi:10.1016/j.tet.2010.05.083
Bachman M, Mann SE, Sheppard TD (2012) Rapid synthesis of highly functionalised a-amino amides and medium ring lactones using multicomponent reactions of amino alcohols and isocyanides. Org Biomol Chem 10:162–170. doi:10.1039/c1ob06534c
Borthwick AD, Liddle J, Davies DE, Exall AM, Hamlett C, Hickey DM, Mason AM, Smith IE, Nerozzi DF, Peace S, Pollard D, Sollis SL, Allen MJ, Woollard PM, Pullen MA, Westfall TD, Stanislaus DJ (2012) Pyridyl-2,5-diketopiperazines as potent, selective, and orally bioavailable oxytocin antagonists: synthesis, pharmacokinetics, and in vivo potency. J Med Chem 55:783–796. doi:10.1021/jm201287w
Gunawan S, Hulme C (2013) Bifunctional building blocks in the Ugi-azide condensation reaction: a general strategy toward exploration of new molecular diversity. Org Biomol Chem 11:6036–6039. doi:10.1039/C3OB40900G
Narhe BD, Tsai M, Sun C (2014) Rapid two-step synthesis of benzimidazo[\(1^\prime \),\(2^\prime \):1,5]pyrrolo[2,3-\(c\)]isoquinolines by a three-component coupling reaction. ACS Comb Sci 16:421–427. doi:10.1021/co500049r
Khoury K, Sinha MK, Nagashima T, Herdtweck E, Dömling A (2012) Efficient assembly of iminodicarboxamides by a “truly” four-component reaction. Angew Chem Int Ed 51:10280–10283. doi:10.1002/anie.201205366
Sinha MK, Khoury K, Herdtweck E, Dömling A (2013) Various cyclization scaffolds by a truly Ugi 4-CR. Org Biomol Chem 11:4792–4796. doi:10.1039/C3OB40523K
Xu Z, Ayaz M, Cappelli AA, Hulme C (2012) General one-pot, two-step protocol accessing a range of novel polycyclic heterocycles with high skeletal diversity. Acs Comb Sci 14:460–464. doi:10.1021/co300046r
Tanaka Y, Hasui T, Suginome M (2007) Acid-free, aminoborane-mediated Ugi-type reaction leading to general utilization of secondary amines. Org Lett 9:4407–4410. doi:10.1021/ol701570c
Suginome M, Tanaka Y, Hasui T (2006) Reductive amination of aldehydes using aminoboranes as iminium ion generators. Synlett 7:1047–1050. doi:10.1055/s-2006-939070
Xu Z, Shaw AY, Nichol GA, Cappelli P, Hulme C (2012) Applications of ortho-phenylisonitrile and ortho-\(N\)-Boc aniline for the two-step preparation of novel bis-heterocyclic chemotypes. Mol Divers 16:607–612. doi:10.1007/s11030-012-9374-1
Xu Z, Shaw AY, Dietrich JA, Cappelli P, Nichol G, Hulme C (2012) Facile, novel two-step syntheses of benzimidazoles, bis-benzimidazoles, and bis-benzimidazole-dihydroquinoxalines. Mol Divers 16:73–79. doi:10.1007/s11030-011-9354-x
Chen Z, Zhang J, Tang D, Xu Z (2014) Synthesis of fused benzimidazole-quinoxalinones via UDC strategy and following the intermolecular nucleophilic substitution reaction. Tetrahedron Lett 55:2742–2744. doi:10.1016/j.tetlet.2014.03.063
Song G, Li S, Yang Z, Yuan J, Wang M, Zhu J, Chen Z, Xu Z (2015) Microwave-assisted synthesis of fused piperazine-benzimidazoles via a facile, one-pot procedure. Tetrahedron Lett 56:4616–4618. doi:10.1016/j.tetlet.2015.06.035
Thompson MJ, Adams H, Chen B (2009) Development of a diversity-oriented approach to oxazole-5-amide libraries. J Org Chem 74:3856–3865. doi:10.1021/jo900425w
Azuaje J, El Maatougui A, Perez-Rubio JM, Coelho A, Fernandez F, Sotelo E (2013) Multicomponent assembly of diverse pyrazin-2(1H)-one chemotypes. J Org Chem 78:4402–4409. doi:10.1021/jo4003163
Shaw AY, Xu Z, Hulme C (2012) Ugi/Robinson-Gabriel reactions directed toward the synthesis of 2,4,5-trisubstituted oxazoles. Tetrahedron Lett 53:1998–2000. doi:10.1016/j.tetlet.2012.02.030
Ugi I, Steinbruckner C (1961) Isonitrile, II. Reaktion von isonitrilen mit carbonylverbindungen, aminen und stickstoffwasserstoffsäure. Chem Ber 94:734–742. doi:10.1002/cber.19610940323
Bienaymé H, Bouzid K (1998) Synthesis of rigid hydrophobic tetrazoles using an Ugi multi-component heterocyclic condensation. Tetrahedron Lett 39:2735–2738. doi:10.1016/S0040-4039(98)00283-4
Nixey T, Kelly M, Semin D, Hulme C (2002) Short solution phase preparation of fused azepine-tetrazoles via a UDC (Ugi/de-Boc/cyclize) strategy. Tetrahedron Lett 43:3681–3684. doi:10.1016/S0040-4039(02)00636-6
Marcos CF, Marcaccini S, Menchi G, Pepino R, Torroba T (2008) Studies on isocyanides: synthesis of tetrazolyl-isoindolinones via tandem Ugi four-component condensation/intramolecular amidation. Tetrahedron Lett 49:149–152. doi:10.1016/j.tetlet.2007.10.154
Acknowledgments
The authors thank the Chongqing Research Program of Basic Research and Frontier Technology (cstc2015jcyjA1328), the Scientific Research Foundation of Chongqing University of Arts and Sciences (Grant Nos. R2013XY01, R2013XY02), and SRF for ROCS, SEM. We would also like to thank Ms H.Z. Liu for obtaining the LC/MS and NMR data.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lei, J., Xu, ZG., Li, SQ. et al. Synthesis of isoindolin-1-one derivatives via multicomponent reactions of methyl 2-formylbenzoate and intramolecular amidation. Mol Divers 20, 859–865 (2016). https://doi.org/10.1007/s11030-016-9679-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-016-9679-6