Abstract
Carbon dioxide storage technologies are needed not only to store the carbon captured in the emissions of hard-to-abate sectors but also for some carbon dioxide removal technologies requiring a final and permanent storage of CO2. The pace and scale of geological CO2 storage deployment have fallen short of expectations, and there is a growing interest in ocean-based CO2 storage options. As complementary to geological storage, buffered accelerated weathering of limestone (BAWL) has been proposed to produce a buffered ionic solution at seawater pH, derived from the reaction in seawater between a CO2 stream and a micron-sized powder of calcium carbonate (CaCO3), within a long tubular reactor. The addition of calcium hydroxide to buffer the unreacted CO2 before the discharge in seawater is also envisaged. BAWL avoids the risks of CO2 degassing back into the atmosphere and does not induce seawater acidification. This work presents a mass and energy balance and preliminary cost analysis of the technology for different configurations of discharge depth (100, 500, 3,000 m), pipeline length (10, 25, 100 km) and diameter of CaCO3 particles (1, 2, 10 µm) fed in the tubular reactor. The total energy consumption to capture and store 1 t of CO2 generated by a steam-methane reforming (SMR) process ranges from 1.3 to 2.2 MWh. The CO2 released from the CaCO3 calcination to produce the buffering solution leads to a total CO2 storage requirement 43–85% higher than the CO2 derived by SMR. The total cost to capture and store 1 t of CO2 from SMR is estimated in the range 142–189 €.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Carbon capture and storage (CCS) is one of the mitigation technologies proposed to limit global warming. It consists of separating CO2 from the flue gas produced in power plants or other industrial sources and, subsequently, storing it underground (Bui et al. 2018). The most advanced technology is geological storage, which can take place in deep saline formations, depleted oil and gas reservoirs, and unminable coal seams. This storage option has several drawbacks, such as the long time required to qualify a suitable site, the uneven geographical distribution of the storage locations and the deep uncertainty over the sustainable injection rate at any given site (IEA 2014; Lane et al. 2021).
An alternative method for the capture and storage of CO2 in the form of bicarbonates was proposed by Rau and Caldeira (1999) and Caldeira and Rau (2000). The method is called accelerated weathering of limestone (AWL) and consists of bringing particles of mineral carbonate and water into direct contact with a CO2-rich waste gas effluent from an industrial source. In AWL, CO2 will react with water forming carbonic acid, which reacts with the carbonate solid to produce bicarbonate ions (HCO3−) in solution according to reaction (1). The AWL technology accelerates a natural process that would occur over the course of millennia (Archer 2005).
However, AWL requires high amounts of water and very large reactors. Due to an incomplete reaction, there may also be unwanted side effects, such as degassing of CO2 and acidification of seawater. To overcome these limitations, an evolution of this method, called buffered accelerated weathering of limestone (BAWL), has been proposed by Caserini et al. (2021), who discussed its chemical basis, as well as its main advantages and limitations.
The BAWL process is designed to produce a buffered ionic calcium bicarbonate solution at the seawater pH. In BAWL, a CO2-rich flux is first mixed with the micronized particles of carbonate mineral and seawater in order to form a calcium carbonate (CaCO3)-rich slurry. This slurry is then injected inside a dissolution reactor (DR), i.e., a pipeline installed on the seafloor, where the carbonate mineral is completely dissolved into an ionic solution on its way towards the deep sea. A second pipeline brings a suspension of slaked lime, i.e. calcium hydroxide (Ca(OH)2), into a buffering reactor (BR), which corresponds to the final part of the DR, in order to buffer the unreacted CO2, bringing its pH back to the seawater value. Caserini et al. (2021) showed that the storage of 1 t of CO2 requires approximately 2,000 m3 of seawater, 1.35 t of CaCO3, and 0.4 t of Ca(OH)2 when the discharge of the ionic solution occurs at 3,000 m depth.
Starting from the chemical modelling of the process (Caserini et al. 2021), this paper analyses the mass and energy balances and the costs of the application of BAWL to the capture and storage of CO2 from the flue gas of an existing industrial source. To this aim, we consider different configurations (DR length, final discharge depth) and working parameters (limestone particle diameter, amount of CaCO3 and Ca(OH)2) to evaluate advantages and disadvantages and their influence on the feasibility of BAWL as a large-scale CO2 capture and storage option.
2 Buffered accelerated weathering of limestone
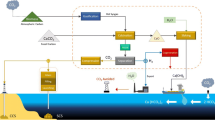
The BAWL process can be applied to capture and store CO2 coming from several industrial sources (e.g., cement or steel-making plants) located on a coastline. The BAWL application considered in this paper aims to capture and store CO2 from the flue gas of steam-methane reformer (SMR), a commercial technology available to produce hydrogen, and the electric calcination, which is a phase of the BAWL technology itself. As shown in Fig. 1, the process is composed of the following sections:
-
A limestone mill (MIL) to grind limestone rock to micron size level
-
An electric calciner (CAL) to produce calcium oxide
-
A slaker (SLA) to produce calcium hydroxide
-
A mixer (MIX), where a CO2-rich gas is mixed with the micronized carbonate mineral and seawater in the desired proportions to form a carbonate-rich acid slurry
-
A dissolution reactor (DR), where calcium carbonate is completely dissolved
-
A buffering reactor (BR), where a proper quantity of calcium hydroxide is added to the main ionic solution to buffer the unreacted CO2
-
A pipeline (SL) for transporting slaked lime to the BR
Reducing the amount of CaCO3 or the diameter of the ground CaCO3 allows for shorter DR pipe and lower depth, increasing the amount of Ca(OH)2 required for buffering the remaining CO2. This leads to many possible configurations characterized by different energy consumption, rate of limestone dissolution and technological challenges (i.e., for pipeline installation in very deep seawater). To evaluate the trade-off, three BAWL configurations, shown in Table 1, are evaluated in the present paper. Configuration A has been considered in Caserini et al. (2021).
2.1 Comminution
The first section of the plant (MIL) is dedicated to the comminution of pure limestone to obtain finely ground calcium carbonate particles for usage in DR, and bigger grains for calcination. Limestone is a sedimentary carbonate rock mainly formed by the mineral calcite. Other minerals such as dolomite or aragonite could also be used, but the analysis of the pros and cons of their use goes beyond the scope of the present work.
The energy required to bring the quarried rock down to a micronized size depends on the specific size to be achieved, the mineralogy of the raw material, and the way with which the size reduction process is carried out. Based on the model provided by Strefler et al. (2018), the energy required for the production of the limestone powder fed to the calciner (diameter 74 µm, starting from 0.5 m quarried stones) is assessed at 45 MJ t−1. The same model has been used to assess the energy requirements for the comminution to 10 µm (456 MJ t−1) and 2 µm (1,444 MJ t−1). For the comminution to 1 µm, the value of 2,002 MJ t−1 has been taken from Rinder and Hagke (2021), since the model by Strefler et al. (2018) leads to an energy consumption outside the range of values proposed by other authors (Moosdorf et al. 2014; Wang and Forssberg 2007) .
2.2 Electric calciner and slaker
An air-tight electric calciner (CAL) produces calcium oxide according to the following high-temperature endothermic reaction:
Electric calciners are today available at the commercial scale from different suppliers and different technologies, i.e., indirect electrically heated rotary kiln up to 400 kg h−1 (IBU-tec 2021), electrically heated conveyors up to 350 kg h−1 (ETIA Ecotechnologies 2021) or flash calcination up to 10,000 kg h−1 (Calix 2021; Leilac Project 2021). The energy consumption of the different technologies is similar, since the energy required for the calcination reaction (2) is largely the main component in each technology.
The calcium oxide produced by the calciner is then transformed into calcium hydroxide by a slaker, adding water according to the following exothermic reaction:
The slaker is fed with the hot (850 °C) CaO released from the calciner; the water used for the slaking cools down the slaked lime slightly above 100 °C. Thus, the steam generated by the slaking process is fed to the calciner to generate a modified atmosphere inside the calciner; this lowers the calcination temperature (Basu et al. 2011) and allows an easier capture of the CO2 downstream through the condensation of the water vapor. The thermal efficiency of the slaker is assumed 90%. Although the slaking process is well known, and slakers are available at the commercial scale, an innovation is the integration of the slaker with the electric calciner in order to minimize energy consumption, which is assessed at 2.4 GJ per ton of CaCO3. Typically, the average fuel consumption of a conventional calcination process is between 2.3 and 3.14 GJ per tonne of CaCO3 (EuLA 2014).
2.3 CO2 source
BAWL could work with different CO2 sources, provided that the impurities in the CO2 stream are limited, to avoid their transfer during the scrubbing and the risk of marine pollution. The CO2 content in the flue gas of an industrial source has a wide composition range, from 3 to 4%vol of a natural gas combined cycle gas turbine (IEA 2014) to 25%vol of a cement plant (IEA 2008). The present paper considers a flue gas from a modular SMR for hydrogen production available at the commercial scale (HyGear 2019), with a CO2 content of 14.1%vol and delivered at 1 bara (absolute pressure).
2.4 Scrubbing and mixing
The CO2 released by the calcination process and the CO2-rich flue gas coming from the external SMR source is scrubbed by seawater and limestone into the MIX, before entering the DR. Scrubbing CO2 with water is a well-known method for capturing CO2 from flue gases (Rau et al. 2007; IEA 2000; Carbonreuse 2021). A bubble generator placed on the bottom of the column produces fine up-flow bubbles that mix with a descending flow of water. A bubble-type absorption column is preferred to a packed bed scrubber, since it is more suitable for capturing a large amount of CO2 (Tier et al. 2014).
A commercial water scrubbing system (CarbonReUse Finland Oy 2021) with a CO2 capture efficiency > 95% using 2,000 m3H2O/tCO2 as dilution water is considered. See Sect. 3 and SM for more details about the amount of water required. The gases, mainly composed by N2, and not absorbed by seawater, are released in a controlled way outside the MIX from its upper part, along with the small percentage of CO2 (neglected in the following calculation) that is not captured in the column. The addition of the micronized CaCO3 is done downstream of the water scrubbing.
Considering a residence time of the water flow inside the MIX of at least 90 s, necessary for CO2 hydration in seawater (Zeebe et al. 1999), a height of the bubble column of at least 10 m is considered, with a total volume of the MIX ranging from approx. 29,000 to 48,000 m3, depending on the configuration.
The CO2-rich gas is delivered to the BAWL plant at 1 bara and a compressor increases its pressure from 1 bara to the pressure at the injection point of the MIX.
2.5 Dissolution reactor
The resulting flow is sent into the DR, where the slurry flow encounters a progressively increasing hydrostatic pressure that enhances the solubility of the carbonate minerals (Zeebe and Westbroek 2003; Dong et al. 2018), according to the previous reaction (1).
The DR is a co-flow moving fluidized bed reactor composed of micron-sized particles of limestone kept in a fully turbulent flow in order to avoid their sedimentation. The length of the DR allows the residence time needed for the dissolution reaction and the pressure of several bars, depending on the configuration. Consequently, the carbonate mineral is completely dissolved into an ionic solution on its way to the sea, before the end of the DR, and a full ionic solution is discharged into the sea. The amounts of carbonate mineral and dissolved CO2 have been calculated using the software PHREEQC Version 3, developed by the US Geological Survey (USGS 2020; Parkhurst and Appelo 2013) and designed to carry out a wide variety of aqueous geochemical calculations. We used the default database (“PHREEQC.DAT”) for the ion activities and reaction rates. Further details about the script can be found in Section SM3 of Supplementary Material. They depend on the quantity of the dilution water, the size of the carbonate particles, the residence time, and the pressure along all the DR. The non-reacting carbonate impurities will be continuously discharged into the sea at the end of the DR, eliminating the need of backwashing and cleaning typical of commercial limestone reactors.
The DR could be manufactured in plastic materials like HDPE (high-density polyethylene), using commercial pipe manufacturing technologies that allow the installation of modular and standardized DRs. A commercial HDPE pipe (AGRU 2021) has been considered.
Diameter, length, final discharging depth, and flow velocity in the pipeline varies in the three configurations (Table 2), assuming for the assessment of the dissolution a straight inclined DR pipe with a fixed standard dimension ratio (SDR, i.e., the ratio between pipe outside diameter and pipe wall thickness). Although the DR pipe will be installed on seafloors that will have different profiles in real applications, the differences in the dissolution kinetics with the case of an average slope are of minor importance. A flow velocity greater than 1.2 m s−1 inside the DR and the SL pipeline has been assumed to avoid the settling of particles.
More details about methods adopted in the hydraulic calculations can be found in Section SM1 of Supplementary Material.
2.6 Buffering reactor and SL pipeline
The residual acidity of the ionic solution at the end of the DR due to the unreacted CO2 is neutralized in the buffering reactor (BR) by an appropriate quantity of calcium hydroxide produced by the slaker, according to the following reaction:
where all ionic reactants and products have been written in fully dissociated form. The main objective of the DR is to maximize the amount of dissolved CaCO3 in order to minimize the use of Ca(OH)2 in the BR. The BR is the last portion of the DR pipe, where the slaked lime is injected and mixed with the ionic solution. The slaked lime will be transported in the form of a slurry through the SL pipe and injected into the BR. By means of one or more nozzles and static mixers, an even mixing is achieved in a short time (15–20 s) in order to avoid any precipitation of calcium carbonate. The BR is thus approximately 30 m long. The BR should be equipped with sensors to measure turbidity, pH, alkalinity, and hardness of the final buffered ionic solution before being discharged into the sea, to dose the appropriate quantity of calcium hydroxide.
The SL pipeline, carrying the ionic solution of Ca(OH)2 from the slaker to the input of the BR, has an internal diameter of about 0.9 m, with small variations in the three configurations related to the velocity.
Similar to the DR pipeline, a flow velocity higher than 1.2 m s−1 is also maintained. Details about hydraulic calculations can be found in Section SM2 of Supplementary Material.
3 Mass and energy balance
Calculations of mass and energy balance are referred to the capture and storage of 1 t of CO2 in flue gas with a 14.1%vol CO2 content coming from an external source, i.e., a SMR plant, as well as the calcination of CaCO3 included in the BAWL. The amount of CaCO3 dissolved in the DR, unreacted CO2 at the end of the DR, total CaCO3 and Ca(OH)2 needed, calculated with the software PHREEQC (USGS 2020; Parkhurst and Appelo 2013) considering the methodology and the calcite dissolution kinetics presented in Caserini et al. (2021) are shown in Table 3.
Table 3 also shows the total CO2 stored in the three configurations (see Sect. 2 for details), assuming that the net CO2 to be stored coming from SMR is always 100,000 t year−1. The production of CO2 internal to the BAWL process, from slaked lime production, implies the need to store an additional amount of CO2, ranging between 43% (case A) and 85% (case C).
Overall, to store 1 tCO2 coming from SMR and the calcination of limestone, there is the need for about 1.5–1.8 t of limestone, depending on the configuration, and 2000 t of seawater.
The amount of CO2 dissolved through reaction with CaCO3 varies between 48% in case A and 18% in case C, implying that a larger amount of Ca(OH)2 for buffering the remaining CO2 is required in the latter.
The energy and mass balance of the process in configuration B is shown in Fig. 2.
Although such results could be used for a more detailed analysis of the efficiency of the process, i.e., based on a life cycle assessment (LCA) approach, a preliminary carbon balance of the process can be estimated by considering the main CO2 emission related to the input used in the process such as the consumption of electricity. Since the LCOE (levelized cost of energy) of new solar and wind installations is already lower than the fossil alternatives (Lazard 2020), it is possible to assume that the new BAWL plants will be powered by renewable sources with a low carbon footprint (IPCC 2011), so that the CO2 penalty of the process is similar to what has been shown in Table 3. On the contrary, if electric energy comes from a mixed renewable/fossil system, the implied carbon emission for electricity consumption should be considered. For example, assuming the average carbon intensity of electricity in the European system in 2020 equal to 231 tCO2eq GWh−1 (EEA 2022), the additional CO2 penalty would range between 30% (case A) and 50% (case C) of the CO2 coming from SMR (Table 4).
4 Preliminary cost analysis
Capital (CAPEX) and Operating Expenditure (OPEX) for each phase and component of BAWL are presented below for the three configurations.
The costs for the equipment used in the different process phases have been derived from either models available in the literature or data provided by specialized suppliers, as shown in Table 5. Specifically, the CAPEX of the limestone mill, electric calciner, and slaker is calculated using market quotations, whereas for the remaining components, i.e., mixer, pumps, and compressor, the criteria indicated by Sinnott and Towler (2009a) have been assumed. In the DR and SL pipelines, calculations regarding the amount of ballast and HDPE have been done following the procedure recommended by the Plastic Pipe Institute (PPI 2008). Except for HPDE pipelines, CAPEX of each piece of equipment has been multiplied by a cautious factor (3.6 for a mixed fluids-solids processing plant), known as Lang factor, to take into account the cost of installation (Lang 1948).
OPEX is broken into two typical categories, i.e., variable and fixed costs. The former is due to the utilization of raw material and electricity; the latter includes labor, maintenance, supervision, contingency and others. The Consumer Price Index Inflation Calculator of the US Bureau of Labor Statistics (USBLS 2021) has been used to adjust the original prices to 2021, and an exchange rate of 1.05 US$ €−1 has been assumed.
The model provided by Camm (1993) has been used to assess all the expenditures involved to carry out the typical operations of a mine, i.e., drilling, blasting, material handling and hauling, labor, and other. Specifically, on the basis of a large open pit mine model and assuming a mine capacity of at least 20,000 t day−1, a cost of ~3€ € for a ton of extracted raw material is assessed, including financial costs. This has been derived considering a discount rate of 4% and 40 years as the total investment period. Costs of processing other than simple extraction have not been included.
The majority of the BAWL components are powered by electricity, whose cost depends on the location where the plant is installed. A cost of 31 € MWh−1 has been assumed based on the average LCOE from unsubsidized wind and solar energy (utility-scale) provided by Lazard (2020).
Limestone transport (1.5 € t−1) is calculated considering an average supply distance of 500 km and an average specific cost of 0.003 € t−1 km−1 (Harvey 2008; Langer et al. 2009; Rau et al. 2007).
The personnel cost is calculated considering 5 shifts of 2 workers with an hourly cost of 28.5 € h−1, which is the average labor cost in 2020 in the European Union (Statista 2022c). The supervision cost is calculated as 25% of the personnel cost.
The other typical costs of an industrial operation, such as the property tax, rent of land, general plant overheads, allocated environmental charges, license fees, and capital charges, are omitted in this analysis and could be better analyzed in further studies considering any specific BAWL installation case. The total cost of BAWL is calculated considering the shares due to debt financing and repayment (DFR) over 100 years for pipelines and over 40 years for other equipment. Costs of inside battery limits investment (ISBL), off-site costs (OSBL), and engineering and construction management (ECM) have been calculated according to Sinnott and Towler (2009d).
The costs assessed for the three configurations are shown in Table 6.
The total cost for the storage of 1 t of CO2 coming from SMR is assessed between 142 € tCO2−1 (case B) and 189 € tCO2−1 (case A). The cost referred to 1 t of CO2 stored in seawater as bicarbonates through the DR (thus including the CO2 generated by the process itself) varies between 132 € tCO2-1 in case A, 92 € tCO2−1 in case B and 84 € tCO2−1 in case C.
5 Discussion
The analysis presented in this paper allows to understand the pros and cons of different technological choices for the BAWL process. The installation of the BAWL in shallow waters (i.e., 100 m) and the use of short pipelines (i.e., 10 km) reduce the dissolution of CaCO3 inside the DR (50% reduction from case A, 20% in case C). This implies the need of more slaked lime to buffer the remaining CO2 (+ 51% in case C compared to case A), more electricity (+ 40% in case C compared to case A) and additional OPEX (+ 30% in case C compared to case A). On the other hand, the installation in deeper waters (i.e., 3,000 m) with longer pipelines (i.e., 100 km) favors CaCO3 dissolution, lowers the operative costs (around 30% reduction in A compared to C, mainly due to the cost of electricity and limestone) but increases the technical challenges and the capital expenditure related to the installation of the pipelines (about 17 times higher in case A compared to C).
Configuration B provides the lowest total cost (142 € per 1 t of CO2 coming from SRM processes), due to intermediate CAPEX and OPEX. Indeed, such a solution represents a compromise in terms of electricity (169 GWh year−1 against 130 GWh year−1 and 218 GWh year−1 of case A and C, respectively), OPEX (93 € per tonne of CO2 derived by SMR, against 79 € and 111 €) and the capital expenditures mostly related to the construction and installation of DR and BR pipelines (35 M€ against 173 M€ and 10 M€). Moreover, the annual production of calcium hydroxide required in this case amounts to \(\sim\) 90 kt year-1 against \(\sim\) 70 kt year−1 and \(\sim\) 141 kt year−1 of case A and C, respectively. Lastly, the total storage cost in configuration C is only ~ 10% higher than in case B, within the uncertainty of the evaluation.
The positive aspects of BAWL as a CO2 capture and storage technology are its relatively small modular size, i.e., about 100,000 t year−1, as well as the use of very widely spread and well-known inputs such as limestone, seawater, and renewable electric energy. Being modular, BAWL can be installed close to a single emission point located on the coastline, whereas geological CCS is typically based on very large projects connecting clusters of emitters (such as the Northern Lights project).
The installation of long DR pipelines on the seafloor minimizes the onshore footprint caused by the huge reactors needed in the AWL technology as initially proposed by Rau and Caldeira (1999).
The BAWL technology releases an alkaline “CO2-equilibrated” solution into the sea, substantially reducing the risk of degassing CO2 or acidifying the sea. This is especially true for discharge at high depths (case A). The discharge in shallow waters (case C) could theoretically imply enhanced biotic CaCO3 precipitation with CO2 release, but also an increase in primary productivity with CO2 consumption (Gore et al. 2019). Further research is needed to assess if biotic precipitation could reduce the efficiency of BAWL in the case of a discharge in shallow waters. Very few studies have investigated the local effect on the biota that could arise from alkalinity addition and the consequent increase of calcite and aragonite saturation state at the discharge point, and since other factors as light and temperature play a significant role.
The local impact of the discharge of a high alkalinity ionic solution can be minimized by choosing a suitable site where the continuous mixing with fresh seawater guarantees its strong dilution. The location of the discharge should be far from enclosed coastal areas and possibly within off-shore currents in order to limit local alterations of the carbonate chemistry and to avoid negative impacts on the biota (Kirchner et al. 2020). Dilution is also useful to avoid or minimize the risk of CO2 degassing, allowing the enriched seawater to mix in a certain time with the surrounding seawater without overcoming the aragonite saturation state (Ωar) value of 5, identified by Hartmann et al. (2022) as a threshold for abiotic precipitation of carbonate.
To increase the overall CO2 benefits of BAWL, the electricity used should derive from renewable sources. The high energy consumption of the technology could be a limiting factor for its development and should rely on specific strategies to develop cheap decarbonized renewable electricity built for the purpose. In the future energy system, characterized by a very high share of decarbonized electric sources (wind and solar in particular), grinding and calcination could be also operated in batch mode only during specific time frames, i.e., off-peak hours (i.e., from 10 p.m. to 8 a.m. on weekdays and all day on weekends and holidays) when power demand is at its lowest, or hours when renewables are working at their highest capacity. This strategy could limit the wastage of renewable electricity not needed by the energy system.
BAWL could be used as a CO2 capture and storage option for hydrogen production from the SMR process, a pivotal energy carrier for the decarbonization of transport and other hard-to-abate sectors (Davis et al. 2018). Assuming a CO2 production of 18.1 t for 1 t of H2 generated by SMR (IEAGHG 2017), the energy consumption for storing CO2 with BAWL is about 31 MWh t H2−1 in case B (26 and 41 in case A and C, respectively). This is substantially lower than the average energy consumption for producing “green” H2 with electrolyzers (51–58 MWh t H2−1, IRENA 2018).
Although in the first phase BAWL could be used for storing the CO2 produced by a SMR process, in the long run the H2 production plants using SMR could be decommissioned and substituted by electric calciners. This strategy allows to produce slaked lime, to store the produced CO2 in the BAWL system, to use the decarbonized slaked lime for atmospheric carbon dioxide removal through ocean liming.
6 Conclusions
In the BAWL process, the discharge of the alkaline solution at the same pH as the surrounding seawater allows permanent CO2 storage in the form of bicarbonates (Caldeira and Rau 2000), without acidifying the seawater and avoiding the risk of CO2 degassing. This study concludes that the capture and storage of CO2 from an external Steam-Methane Reformer source could be carried out with different configurations, associated with different limestone, electricity consumptions, and costs.
This deployment strategy is very well suited for capturing and storing CO2 from new modular “blue” hydrogen production units, instead of installing tailor-made CCS at industrial sites. The infrastructure of the BAWL could be conveniently used for the generation of CO2 negative emissions when the main aim of climate restoration will be the removal of CO2 from the atmosphere.
The estimated cost of the captured and stored CO2 with BAWL is comparable with the forecast of large-scale geological CCS projects (Gassnova 2020). The strengths of the CO2 capture and storage using BAWL rely on its modularity and on very abundant and well-distributed raw materials: renewable energy, seawater, and calcium carbonate.
Further studies should be done in order to assess the environmental impacts through the LCA methodology and the potential impacts on marine ecosystems of discharging large amounts of alkaline water into the sea.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
AGRU (2021) XXL piping system. Retrieved on December 2021 from https://agruamerica.com/wp-content/uploads/2020/03/Pro_grossrohr_en-WEBSC.pdf
Archer D (2005) Fate of Fossil Fuel CO2 in Geologic Time. J Geophys Res: Oceans 110 (C9). https://doi.org/10.1029/2004JC002625
Basu P, Achayra B, Dutta A (2011) Study of calcination - carbonation of calcium carbonate in different fluidizing mediums for chemical looping gasification in circulating fluidized beds in "10th International Conference on Circulating Fluidized Beds and Fluidization Technology - CFB-10", T. Knowlton, PSRI Eds, ECI Symposium Series. https://dc.engconfintl.org/cfb10/36
Bui M, Adjimam CS, Bardow A, Anthony EJ, Boston A, Brown S et al (2018) Carbon capture and storage (CCS): the way forward. Energy Environ Sci 5(11):1062–1176. https://doi.org/10.1039/C7EE02342A
Caldeira K, Rau G (2000) Accelerating carbonate dissolution to sequester carbon dioxide in the ocean: geochemical implications. Geophys Res Lett 27(2):225–228. https://doi.org/10.1029/1999GL002364
Calix (2021) Reducing CO2 Emissions by Project LEILAC. Available at: www.calix.global/industries/cement-and-lime/. Retrieved in March, 2021
Camm, TW (1993) Simplified cost models for prefeasibility mineral evaluations. Bureau of Mines Information Circular 9298. Available at: https://pubs.er.usgs.gov/publication/70138816. Retrieved in March 2022.
Carbonreuse (2021) Carbonreuse CO2 process. Our process. Available at: https://www.carbonreuse.fi/our-solution-1. Retrieved in May, 2022.
CarbonReUse Finland Oy (2021) Budget offer. Personal Communication. Website of the supplier: www.carbonreuse.fi
Caserini S, Cappello G, Righi D et al (2021) Buffered accelerated weathering of limestone for storing CO2 chemical background. Int J Greenh Gas Control 112:103517. https://doi.org/10.1016/j.ijggc.2021.103517
Davis SJ, Lewis NS, Shaner M, Aggarwal S, Arent D et al (2018) Net-zero emissions energy systems. Science 360(6396):eaas9793. https://doi.org/10.1126/science.aas9793
Dong S, Subhas A, Rollins N, Naviaux J, Adkins J, Berelson W (2018) A kinetic pressure effect on calcite dissolution in seawater. Geochim Cosmochim Acta 238:411–423. https://doi.org/10.1016/j.gca.2018.07.015
EEA (2022) Greenhouse gas emission intensity of electricity generation in Europe. www.eea.europa.eu/ims/greenhouse-gas-emission-intensity-of-1
ETIA Ecotechnologies (2021) Spirajoule: electrically heated screw conveyor. Available at: https://etia-group.com/our-products/spirajoule/. Retrieved in May, 2022
EuLA (2014) A competitive and efficient lime industry. cornestone for a sustainable Europe. Technical report. European Lime Association. Available on: https://www.eula.eu/wp-content/uploads/2019/02/A-Competitive-and-Efficient-Lime-Industry-Technical-report-by-Ecofys_0.pdf. Retrieved in May, 2022
Gassnova (2020) Potential for reduced costs for carbon capture, transport and storage value chains (CCS). Report No.: 2019–1092, Rev. 2. Available at: https://ccsnorway.com/app/uploads/sites/6/2020/07/Report-Cost-reduction-curves-for-CCS-Gassnova-version-2b-1.pdf. Retrieved in May, 2022
Gore S, Renforth P, Perkins R (2019) The potential environmental response to increasing oceanalkalinity for negative emissions. Mitig Adapt Strateg Glob Change 24(7):1191–1211. https://doi.org/10.1007/s11027-018-9830-z
Hartmann J, Suitner N, Lim C, Schneider J, Marín-Samper L, Arístegui J, Renforth P, Taucher J and Riebesell U (2022) Stability of alkalinity in ocean alkalinity enhancement (OAE) approaches – consequences for durability of CO2 storage. Biogeosci 20(4):781–802. https://doi.org/10.5194/bg-20-781-2023
Harvey D (2008) Mitigating the atmospheric CO2 increase and ocean acidification by adding limestone powder to upwelling regions. J Geophys Res Atmos 113:C4. https://doi.org/10.1029/2007JC004373
HyGear (2019) Hy.Gen on-site hydrogen generation system. Cost-effective steam methane reforming. Available at: https://hygear.com/wp-content/uploads/2019/06/HYGEAR_HyGen_May19.pdf. Retrieved in May, 2022.
IBU-tec (2021) Fact sheet. Indirectly heated Rotary Kilns. Available at: https://www.ibu-tec.com/fileadmin/user_upload/Download_PDF/Datenblaetter_Fact_sheets/IBU-tec_Indirectly_Fired_Rotary_Kilns.pdf. Retrieved in March, 2021.
IEA (2000) Capture of CO2 using water scrubbing. Report Number PH3/26. International Energy Agency. Available at: https://ieaghg.org/docs/General_Docs/Reports/PH3_26%20Water%20scrubbing.pdf. Retrieved in May, 2022.
IEA (2008) CO2 capture in the cement industry. International Energy Agency. Technical study. Report Number 2008/3. Available at: https://ieaghg.org/docs/General_Docs/Reports/2008-3.pdf. Retrieved in May, 2022.
IEA (2014) CCS 2014 What Lies in Store for the CCS? International Energy Agency. Technology report — June 2014. Available at: https://www.iea.org/reports/what-lies-in-store-for-ccs. Retrieved in May, 2022
IEAGHG (2017) Techno - economic evaluation of SMR based standalone (merchant) hydrogen plant with CCS. Technical Report 2017–2
IMS (2021) Integrity Municipal Systems, LLC. Personal Communication
IPCC (2011) Summary for Policymakers. In: IPCC Special Report on Renewable Energy Sources and Climate Change Mitigation [O Edenhofer R. Pichs‐Madruga Y, Sokona K, Seyboth P, Matschoss S, Kadner T, Zwickel P, Eickemeier G, Hansen S, Schlömer C, von Stechow (eds)], Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
IRENA (2018) Hydrogen from renewable power: technology outlook for the energy transition. Available at: https://www.irena.org/publications/2018/sep/hydrogen-from-renewable-power
JDKILN (2021) Juda Kiln - 200 t/d - 3 production lines - EPC Project. Available at: https://www.jdkiln.com/juda-kiln-200td-3-production-lines-epc-project-product/. Retrieved in December 2021.
Kirchner J, Lettmann K, Schnetger B, Wolff JO, Brumsack HJ (2020) Carbon Capture via accelerated weathering of limestone: modelling local impacts on the carbonate chemistry of the southern North Sea. Int J Greenhouse Gas Control 92:1–10. https://doi.org/10.1016/j.ijggc.2019.102855
Lane J, Greig C, Garnett A (2021) Uncertain storage prospects create a conundrum for carbon capture and storage ambitions. Nat Clim Chang 11:925–936. https://doi.org/10.1038/s41558-021-01175-7
Lang HJ (1948) Simplified approach to preliminary cost estimates. Chemical Engineering, June: 112–113.
Langer WH, Rau GH, Caldeira K et al. (2009) Accelerated weathering of limestone for CO2 mitigation opportunities for the stone and cement industries. Mining Engineering 61, 2: 310– 315. Available at: https://pubs.er.usgs.gov/publication/70037039. Retrieved in May, 2022.
Lawrence A, Thollander P, Andrei M, Karlsson M (2019) Specific energy consumption/use (SEC) in energy management for improving energy efficiency in industry: meaning, usage and differences. Energies 12:2. https://doi.org/10.3390/en12020247
Lazard (2020) Levelized cost of energy, levelized cost of storage, and levelized cost of hydrogen 2020. Available at: www.lazard.com/perspective/lcoe2020. Retrieved in May 2022.
Leilac Project (2021) The core technology - Direct Separation. Available at: https://www.project-leilac.eu/the-core-technology. Retrieved in May, 2022.
Moosdorf N, Renforth P, Hartmann J (2014) Carbon dioxide efficiency of terrestrial enhanced weathering. Environ Sci Technol 48(9):4809–4816. https://doi.org/10.1021/es4052022
Parkhurst DL and Appelo CAJ (2013) Description of input and examples for PHREEQC version 3—a computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. US Geological Survey Techniques and Methods, Book 6, Chapter A43 497. http://pubs.usgs.gov/tm/06/a43
PPI (2008) Plastic Pipe Institute. Second edition handbook of pe pipe 2008. Chapter 10 Marine Installation. Available at: https://www.charterplastics.com/handbook-of-pe-pipe. Retrieved in May, 2022.
Rau G, Caldeira K (1999) Enhanced carbonate dissolution: a means of sequestering waste CO2 as ocean bicarbonate. Energy Convers Manage 40:1803–1813. https://doi.org/10.1016/S0196-8904(99)00071-0
Rau G, Knauss K, Langer W, Caldeira K (2007) Reducing energy-related CO2 emissions using accelerated weathering of limestone. Energy 32:1471–1477. https://doi.org/10.1016/j.energy.2006.10.011
Rau G, Caldeira K, Knauss KG, Downs B, Sarv H (2001) Enhanced carbonate dissolution as a means of capturing and sequestering carbon dioxide. First National Conference on Carbon Sequestration, Washington DC (US), May 14–17, 2001
Rinder T, von Hagke C (2021) The influence of particle size on the potential of enhanced basalt weathering for carbon dioxide removal - insights from a regional assessment. J Clean Prod 315:128178. https://doi.org/10.1016/j.jclepro.2021.128178
SCM (2020) Shanghai Clirik Machinery CO. LTD. 6 Sets of HGM125L - 70 t/d - Micro Powder Grinding Mill Plant. Personal communication
Sinnott and Towler (2008b). Chapter 6 “Cost and Project Evaluation”. Table 6.6. In: Chemical Engineering Design. Principles, Practice and Economics of Plant and Process Design. 2nd edition. Butterworth-Heinemann, USA. p. 320
Sinnott and Towler (2008c). Chapter 6 “Cost and Project Evaluation”. Table 6.6. In: Chemical Engineering Design. Principles, Practice and Economics of Plant and Process Design. 2nd edition. Butterworth-Heinemann, USA. p. 319
Sinnott and Towler (2008d). Chapter 6 ‘Cost and Project Evaluation”. In: Chemical Engineering Design. Principles, Practice and Economics of Plant and Process Design. 2nd edition. Butterworth-Heinemann, USA. pp. 299–301.
Sinnott GP, Towler RK (2009a) Chemical engineering design: principles, practice, and economics of plant and process design. 2nd edition. Butterworth-Heinemann, USA. Available at: https://www.academia.edu/37151834/Chemical_Engineering_Design_Principles_Practice_and_Economics_of_Plant_and_Process_Design. Retrieved in May, 2022.
Statista (2022a) Price of high-density polyethylene worldwide from 2017 to 2020 with estimated figures for 2021 to 2022a. Available at: https://www.statista.com/statistics/1171074/price-high-density-polyethylene-forecast-globally/. Retrieved in March 2022a
Statista (2022b) Cement prices in the United States from 2010 to 2021 (in U.S. dollars per metric ton). Available at: https://www.statista.com/statistics/219339/us-prices-of-cement/. Retrieved in March 2022b
Statista (2022c) Average hourly labor cost in selected European countries in 2020. Available at: https://www.statista.com/statistics/1211601/hourly-labor-cost-in-europe/#:~:text=The%20average%20hourly%20labor%20cost,to%206.5%20Euros%20in%20Bulgaria. Retrieved in May, 2022c.
Strefler J, Amann T, Bauer N, Kriegel E, Hartmann J (2018) Potential and costs of carbon dioxide removal by enhanced weathering of rocks. Environ Res Lett 13(3):034010. https://doi.org/10.1088/1748-9326/aaa9c4
Tier S, Suomalainen M, Onarheim K (2014) Pre-evaluation of a new process for capture of CO2 using water. VTT research report n. 04035–14. Available at: https://publications.vtt.fi/julkaisut/muut/2014/VTT-R-04035-14.pdf. Retrieved in May, 2022.
USBLS (2021) CPI Inflation Calculator. U.S. Bureau of Labor Statistics. Available at: https://www.bls.gov/data/inflation_calculator.htm. Retrieved in December 2021.
USGS (2020) PHREEQC Version 3. United States Geological Survey. Retrieved from www.usgs.gov/software/phreeqc-version-3.
Wang Y, Forssberg E (2007) Enhancement of energy efficiency for mechanical production of fine and ultra-fine particles in comminution. China Particuology 5(3):193–201. https://doi.org/10.1016/j.cpart.2007.04.003
Zeebe RE, Westbroek P (2003) A simple model for the CaCO3 saturation state of the ocean: the “Strangelove”, the “Neritan”, and the “Cretan” Ocean. Geochem Geophys Geosyst 4:12. https://doi.org/10.1029/2003GC000538
Zeebe RE, Wolf-Gladrow DA, Jansen H (1999) On the time required to establish chemical and isotopic equilibrium in the carbon dioxide system in seawater. Mar Chem 65:135–153. https://doi.org/10.1016/S0304-4203(98)00092-9
Funding
Open access funding provided by Politecnico di Milano within the CRUI-CARE Agreement. The research was carried out within the Desarc-Maresanus project (www.desarc-maresanus.net), which received the financial support of Amundi SGR SpA.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: SC and GC. Chemical background and calculations through PHREEQC: SV and GR. Mass and energy balance and hydraulic calculations: SC, GC, SDM and FC. Cost and energy analyses: GC and SDM. First draft of the manuscript: SC and SDM. Final review: MG. All authors critically contributed to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
De Marco, S., Varliero, S., Caserini, S. et al. Techno-economic evaluation of buffered accelerated weathering of limestone as a CO2 capture and storage option. Mitig Adapt Strateg Glob Change 28, 17 (2023). https://doi.org/10.1007/s11027-023-10052-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11027-023-10052-x