Abstract
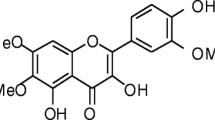
Oxidative stress (OS) is well established as a major event in Alzheimer’s disease (AD) pathology. One of the mostly-researched classes of antioxidants to manage with overwhelming OS include flavonoids. This study was aimed to investigate the protective effect of A. congolensis extract (HEEAC) on AlCl3-mediated AD-like OS and assess the contribution of its antioxidant flavonoid contents. Female Wistar (250–300 g) rats received orally 50 mg/Kg bw of AlCl3, followed one hour later by doses (150 or 300 mg/kg) of HEEAC or vitamin E at 100 mg/kg daily for eight consecutive weeks. OS related biomarkers were evaluated at the end of treatment. To assess the contribution of flavonoid contents to its activity, HEEAC was fractioned using solvent of varying polarities. Flavonoid-rich extracts obtained were tested for their antioxidant capacity. AlCl3 administration significantly lowered antioxidant enzymes (catalase, glutathione peroxidase) and aconitase levels, reduced total thiol and thiol protein levels and increased lipid peroxidation and protein oxidation levels in brain. When co-administrated with HEEAC at 150 mg/kg, all of these OS related biomarkers were significantly moderated. The efficacity of the extract was significantly higher than vitamin E. Flavonoid-rich fractions extracted mainly n-butanol fraction show strong antioxidant activity, which can be considered as the major antioxidant fraction of this plant. HEEAC protect brain cells against oxidative damage induced by AlCl3, specifically through the strong antioxidant property of its n-butanol flavonoid-rich fraction, which may be a promising agent for preventing oxidative damage in AD.
Similar content being viewed by others
Data availability
The data generated and analysed during the current study are available from the corresponding author on request.
Code availability
Not applicable.
References
Aiyegoro O, Okoh A (2010) Preliminary phytochemical screening and in vitro antioxidant activities of the aqueous extract of Helichrysum longifolium DC. BMC 10:21. https://doi.org/10.1186/1472-6882-10-21
Aksenov MY, Aksenova MV, Butterfield DA, Geddes JW, Markesbery WR (2001) Protein oxidation in the brain in Alzheimer’s disease. Neuroscience 103:373–383
Arslan J, Jamshed H, Qureshi H (2020) Early detection and prevention of Alzheimer’s disease: role of oxidative markers and natural antioxidants. Front Aging Neurosci 12:231. https://doi.org/10.3389/fnagi.2020.00231
Assob JCN, Njouendou AJ, Nkeng PEA, Badami SM, Chouna JR, Verapur VP, Typpewamy BD, Wanji S (2014) In vitro screening of antioxidant properties of ten cameroonian medicinal plants. J Adv Biol 2(3):171–177
Auti ST, Kulkarni YA (2019) Neuroprotective effect of cardamom oil against aluminum induced neurotoxicity in rats. Front Neurol 10:399. https://doi.org/10.3389/fneur.2019.00399
Awa FM, Uzoegwwu PN, Ifeonu P, Oyugi JO, Rutherford J, Yao X, Fehrmann F, Fowke KR, Eze MO (2012) Free radical scavaging activity, phenolic contents and cytotoxicity of selected Nigerian medicinal plants. Food Chem 14(2):1137–1143
Beinert H, Kennedy MC (1993) Aconitase, a two-faced protein: enzyme and iron regulatory factor. FASEB J 7:1442–1449
Bekkara F, Jay M, Viricel MR (1998) Distribution of phenolic compounds within seed and seedlings of two Vicia fabacvs differing in their seed tannin content, and study of their seed and root phenolic exudations. Plant Soil 203:27–36
Cakatay U, Kayali R, Uzun H (2008) Relation of plasma protein oxidation parameters and paraoxonase activity in the ageing population. Clin Experim Med 8:51–57
Chang YT, Chang WN, Tsai NW, Huang CC, Kung CT, Su YJ, Lin WC, Cheng BC, Su CM, Chiang YF, Lu CH (2014) The roles of biomarkers of oxidative stress and antioxidant in Alzheimer’s disease: a systematic review. BioMed Res Int 2014:14. https://doi.org/10.1155/2014/182303
Cheignon C, Tomas M, Bonnefont-Rousselot D, Faller P, Hureau C, Collin F (2018) Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biol 14:450–464
Cummings J, Aisen PS, DuBois B, Frölich L, Jack CR Jr, Jones RW, Morris JC, Raskin J, Dowsett SA, Scheltens P (2016) Drug development in Alzheimer’s disease: the path to 2025. Alzheimer’s Res Ther 8:39. https://doi.org/10.1186/s13195-016-0207-9
Di Paolo M, Papi L, Gori F, Turillazzi E (2019) Natural products in neurodegenerative diseases: a great promise but an ethical challenge. Int J Mol Sci 20:5170. https://doi.org/10.3390/ijms20205170
Djiokeng PG, Ngoumen NDJ, Youovop FJ, Mbong AM-A, Ngondi JL, Oben JE (2014) Neuroprotective effect of Baillonella toxisperma Pierre on the oxidative stress status in an experimental animal model of Alzheimer’s disease. JAB 5:3
Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95
Dua A, Gupta SK, Mittal A, Ritu M (2012) A study of antioxidant properties and antioxidant compounds of cumin (Cuminum cyminum). IJPBA 3(5):1110–1116
Dysken MW, Sano M, Asthana S, Vertrees JE, Pallaki M, Llorente M, Love S, Schellenberg GD, McCarten JR, Malphurs J, Prieto S, Chen P, Loreck DJ, Trapp G, Bakshi RS, Mintzer JE, Heidebrink JL, Vidal-Cardona A, Arroyo LM, Cruz AR, Zachariah S, Kowall NW, Chopra MP, Craft S, Thielke S, Turvey CL, Woodman C, Monnell KA, Gordon K, Tomaska J, Segal Y, Peduzzi PN, Guarino PD (2014) Effect of vitamin E and memantine on functional decline in Alzheimer disease: the TEAM-AD VA cooperative randomized trial. JAMA - J Am Med Assoc 311:33–44
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Meth Enzymol 105:114–121
Fokou A (2006) Chemical investigation of three plants used in cameroonian traditional medicine: Maesopsiseminii (Rhamnaceae), Autranella congolensis (Sapotaceae) and Pentadesma grandifolia (Guttiferae)”, Thesis, Bielefeld University
Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL, Bohr VA (2019) Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 15:565–581. https://doi.org/10.1038/s41582-019-0244-7
Huang WJ, Zhang X, Chenhuang WW (2016) Role of oxidative stress in Alzheimer’s disease. Biomed Rep 4:519–522
Kennedy MC, Emptage MH, Dreyerand JL, Beinert H (1983) The role of iron in the activation–inactivation of aconitase. J Biol Chem 258:11098–11105
Krebs HA, Holzach O (1952) The conversion of citrate into cis-aconitate and isocitrate in the presence of aconitase. Biochem J 52(3):527–528
Kumar V, Bal A, Gill KD (2009) Aluminium-induced oxidative DNA damage recognition and cell-cycle disruption in different regions of rat brain. Toxicol 264:137–144
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG, Ahn BW, Shaltiel S, Stadtman ER (1990) Determination of carbonyl content in oxidatively modified proteins. Meth Enzymol 186:464–478
Liu Z, Zhang A, Sun H, Han Y, Konga L, Wang X (2017) Two decades of new drug discovery and development for Alzheimer’s disease. RSC Adv 7:6046–6058
Lowry OH, Rosenberg NJ, Fany AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. JBC 193:265–275
Mangialasche F, Baglioni M, Cecchetti R, Kivipelto M, Ruggiero C, Piobbico D, Kussmaul L, Monastero R, Brancorsini S, Mecocci P (2015) Lymphocytic mitochondrial aconitase activity is reduced in Alzheimer’s disease and mild cognitive impairment. J Alzheimer’s dis 44:649–660
Minotti G, Aust SD (1987) An investigation into the mechanism of citrate-Fe2+-dependent lipid peroxidation. Free Radic Biol Med 3(6):379–387
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biochem 247(10):3170–3175
National Research Council (1985) A guide for the care and use of laboratory animals. A report of the Institute of Laboratory Animal Research Committee on Care and Use of Laboratory Animals. Washington DC: National Academy Press. National Institute of Health Publication No. 85–23; 2000
Ngoumen NDJ, Ngondi JL, Oben JE (2020) Effect of Autranella congolensis on lipid profile of rat’s brain with experimentally induced Alzheimer’s disease. J Food Res 9(4):60–70
Nguedia JCA, Njouendou AJ, Nkeng PEA, Chouna JR, Badami SM, Verapur VP, Typpewamy BD, Wanji S (2014) Protective effect of Autranella congolensis and Sapium ellipticum stem bark extracts against hepatotoxicity induced by thioacetamide. Archives 2(39):38–47
Pohl F, Kong T, Lin P (2018) The potential use of plant natural products and plant extracts with antioxidant properties for the prevention/treatment of neurodegenerative diseases: in vitro vivo and clinical trials. Molecules 23:3283. https://doi.org/10.3390/molecules23123283
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269:337–341
Pritam P, Deka R, Bhardwaj A, Srivastava R, Kumar D, Jha AK, Jha NK, Villa C, Jha SK (2022) Antioxidants in Alzheimer’s disease: current therapeutic significance and future prospects. Biology 11:212. https://doi.org/10.3390/biology11020212
Sanchez-Moreno C, Larrauri JA, Saura-Calixto F (1999) Free radical scavenging capacity of selectes red, rosé and white wines. J Sci Food Agric 79:1301–1304
Sedlak J, Lindsay RH (1968) Estimation of total protein-bound, and non-protein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Sies H, Berndt C, Jones DP (2017) Oxidative stress. Annu Rev of Biochem 86:715–748
Sinha K (1984) Colorimetric assay of catalase. Anal Biochem 47(2):389–394
Sreejayan N, Rao MNA (1997) Nitric oxide scavenging by curcuminoids. J Pharmacol 49:105–107
Sun JS, Tsuang YW, Chen JJ, Yang YS, Lu FJ (1998) An ultra-weak chemiluminescence study on oxidative stress in rabbits following acute thermal injury. Burrus 24:225–231
Tonniesa E, Trushinaa E (2017) Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimer’s Dis 57:1105–1121
Wang Y, Yang J, Yi J (2012) Redox sensing by proteins: oxidative modifications on cysteines and the consequent events. Antioxid Redox Signal 16:649–657
Xie L, Deng Z, Zhang J, Dong H, Wang W, Xing B, Liu X (2022) Comparison of flavonoid O-Glycoside, C-Glycoside and their aglycones on antioxidant capacity and metabolism during in vitro digestion and in vivo. Foods 11:882
Yagi K (1976) Simple fluorometric assay for lipoperoxyde in blood plasma. Biochem Med 15(2):212–216
Ye H, Shaw IC (2019) Food flavonoid ligand structure/estrogen receptor-α affinity relationships—toxicity or food functionality? Food Chem Toxicol 129:328–336
Zeka K, Ruparelia K, Arroo RRJ, Budriesi R, Micucci M (2017) Flavonoids and their metabolites: Prevention in cardiovascular diseases and diabetes. Diseases 5:19. https://doi.org/10.3390/diseases5030019
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Ngoumen Ngassa Dany Joël, Ella Fils Armand, Ambamba Akamba Bruno Dupon and Nanhah Kamga Jules Vidal. The first draft of the manuscript was written by Ngoumen Ngassa Dany Joël, Mandob Enyegue Damaris, Fonkoua Martin, Ngondi Judith Laure and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Animal Ethics Committee of the Faculty of Sciences, University of Yaounde 1, Yaounde, Cameroon and in accordance with the principles of the “Guide to the Care and Use of Experimental Animals (Committee on Care and Use of Laboratory Animals, 1985)’’.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ngoumen, D.J.N., Mandob, D.E., Ella, F.A. et al. Flavonoid-enrich extract of Autranella congolensis (Sapotaceae) protects against aluminium chloride-mediated Alzheimer’s disease-like oxidative stress in rat through the antioxidant properties. Metab Brain Dis 38, 1025–1034 (2023). https://doi.org/10.1007/s11011-022-01142-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01142-x