Abstract
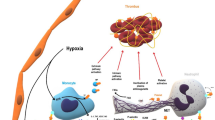
Neutrophil extracellular traps (NETs) are network-like structures released by activated neutrophils. They consist mainly of double-stranded DNA, histones, and neutrophil granule proteins. Continuous release of NETs in response to external stimuli leads to activation of surrounding platelets and monocytes/macrophages, resulting in damage to endothelial cells (EC) and vascular smooth muscle cells (VSMC). Some clinical trials have demonstrated the association between NETs and the severity and prognosis of atherosclerosis. Furthermore, experimental findings have shed light on the molecular mechanisms by which NETs contribute to atherogenesis. NETs play a significant role in the formation of atherosclerotic plaques. This review focuses on recent advancements in the understanding of the relationship between NETs and atherosclerosis. It explores various aspects, including the formation of NETs in atherosclerosis, clinical trials investigating NET-induced atherosclerosis, the mechanisms by which NETs promote atherogenesis, and the translational implications of NETs. Ultimately, we aim to propose new research directions for the diagnosis and treatment of atherosclerosis.





Similar content being viewed by others
Data Availability
Not applicable.
Abbreviations
- NETs:
-
Neutrophil extracellular traps
- dsDNA:
-
Double-stranded DNA
- citH3:
-
Citrullinated histone H3
- MPO:
-
Myeloperoxidase
- NE:
-
Neutrophil elastase
- ApoB:
-
Apolipoprotein B
- LPS:
-
Lipopolysaccharide
- PMA:
-
Phorbol-12 myristate-13 acetate
- ox-LDL:
-
Oxidized low-density lipoprotein
- mtDNA:
-
Mitochondrial DNA
- C5aR1:
-
Complement C5a receptor 1
- STAT3:
-
Signal transducers and activators of transcription family 3
- ROS:
-
Reactive oxygen species
- VDAC:
-
Voltage-dependent ion channel
- cGAS:
-
Cyclic GMP-AMP synthesis
- PAD4:
-
Peptidylarginine deiminase 4
- Nox:
-
NADPH oxidase
- Ang II:
-
Angiotensin II
- ATR1:
-
Angiotensin type 1 receptor
- PKC:
-
Protein kinase C
- CXCL:
-
Chemokine (C-X-C motif) ligand
- CXCR:
-
Chemokine (C-X-C motif) receptor
- TLR:
-
Toll-like receptor
- IRAK1/4:
-
Interleukin-1 receptor-associated kinase 1/4
- Cl− :
-
Chloride ions
- SGK1:
-
Serum- and glucose-dependent protein kinase 1
- Ca2+ :
-
Calcium ion
- Arg:
-
Arginine
- IRGM:
-
Immune-related GTPase family M protein
- cPLA2:
-
Cytosolic phospholipase A2
- cfDNA:
-
Cell-free DNA
- STEMI:
-
ST-segment elevation myocardial infarction
- TCC:
-
Terminal complement complex
- TXA2:
-
Thromboxane A2
- PSGL-1:
-
P-selectin glycoprotein ligand-1
- ICAM:
-
Intercellular adhesion molecule
- LFA-1:
-
Lymphocyte function-associated antigen 1
- CCL:
-
Chemokine (C–C motif) ligand
- vWF:
-
Von Willebrand Factor
- NLRP3:
-
NOD-like receptor protein 3
- IL:
-
Interleukin
- MMP:
-
Matrix metalloproteinase
- EGFR:
-
Epidermal growth factor receptor
- HOCI:
-
Hypochlorous acid
- HOSCN:
-
Hypothiocyanous acid
- TNF:
-
Tumor necrosis factor
- MCP-1:
-
Monocyte chemoattractant protein-1
- EC:
-
Endothelial cells
- EC-MVs:
-
Endothelial-derived microvesicles
- VCAM:
-
Vascular cell adhesion molecule
- ABCA1:
-
ATP binding cassette transporter A1
- LCAT:
-
Lecithin cholesterol acyltransferase
- ApoA1:
-
Apolipoprotein A1
- eNOS:
-
Endothelial nitric oxide synthase
- TF:
-
Tissue factor
- VSMC:
-
Vascular smooth muscle cells
- RAGE:
-
Receptors for advanced glycosylation end products
- sGC:
-
Soluble guanylyl cyclase
- ETs:
-
Extracellular traps
- tACPA:
-
Therapeutic anti-citrullinated protein antibody
References
Libby P, Buring JE, Badimon L, Hansson GK, Deanfield J, Bittencourt MS et al (2019) Atherosclerosis. Nat Rev Dis Primers 5:56. https://doi.org/10.1038/s41572-019-0106-z
Libby P (2021) The changing landscape of atherosclerosis. Nature 592:524–533. https://doi.org/10.1038/s41586-021-03392-8
Wolf D, Ley K (2019) Immunity and Inflammation in Atherosclerosis. Circ Res 124:315–327. https://doi.org/10.1161/CIRCRESAHA.118.313591
Soehnlein O, Libby P (2021) Targeting inflammation in atherosclerosis - from experimental insights to the clinic. Nat Rev Drug Discov 20:589–610. https://doi.org/10.1038/s41573-021-00198-1
Adamstein NH, MacFadyen JG, Rose LM, Glynn RJ, Dey AK, Libby P et al (2021) The neutrophil-lymphocyte ratio and incident atherosclerotic events: analyses from five contemporary randomized trials. Eur Heart J 42:896–903. https://doi.org/10.1093/eurheartj/ehaa1034
Castanheira F, Kubes P (2019) Neutrophils and NETs in modulating acute and chronic inflammation. Blood 133:2178–2185. https://doi.org/10.1182/blood-2018-11-844530
Mortensen JH, Sinkeviciute D, Manon-Jensen T, Domislović V, McCall K, Thudium CS et al (2022) A specific calprotectin neo-epitope [CPa9-HNE] in serum from inflammatory bowel disease patients is associated with neutrophil activity and endoscopic severity. J Crohns Colitis 16:1447–1460. https://doi.org/10.1093/ecco-jcc/jjac047
Döring Y, Soehnlein O, Weber C (2017) Neutrophil extracellular traps in atherosclerosis and atherothrombosis. Circ Res 120:736–743. https://doi.org/10.1161/CIRCRESAHA.116.309692
van der Windt DJ, Sud V, Zhang H, Varley PR, Goswami J, Yazdani HO et al (2018) Neutrophil extracellular traps promote inflammation and development of hepatocellular carcinoma in nonalcoholic steatohepatitis. Hepatology 68:1347–1360. https://doi.org/10.1002/hep.29914
Criqui MH, Matsushita K, Aboyans V, Hess CN, Hicks CW, Kwan TW et al (2021) Lower extremity peripheral artery disease: contemporary epidemiology, management gaps, and future directions: a scientific statement from the american heart association. Circulation 144:e171–e191. https://doi.org/10.1161/CIR.0000000000001005
Borén J, Chapman MJ, Krauss RM, Packard CJ, Bentzon JF, Binder CJ et al (2020) Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 41:2313–2330. https://doi.org/10.1093/eurheartj/ehz962
Duan Y, Gong K, Xu S, Zhang F, Meng X, Han J (2022) Regulation of cholesterol homeostasis in health and diseases: from mechanisms to targeted therapeutics. Signal Transduct Target Ther 7:265. https://doi.org/10.1038/s41392-022-01125-5
Di Angelantonio E, Gao P, Pennells L, Kaptoge S, Caslake M, Thompson A et al (2012) Lipid-related markers and cardiovascular disease prediction. JAMA 307:2499–2506. https://doi.org/10.1001/jama.2012.6571
Björnson E, Adiels M, Taskinen MR, Burgess S, Rawshani A, Borén J et al (2023) Triglyceride-rich lipoprotein remnants, low-density lipoproteins, and risk of coronary heart disease: a UK Biobank study. Eur Heart J 44:4186–4195. https://doi.org/10.1093/eurheartj/ehad337
Richardson TG, Sanderson E, Palmer TM, Ala-Korpela M, Ference BA, Davey SG et al (2020) Evaluating the relationship between circulating lipoprotein lipids and apolipoproteins with risk of coronary heart disease: a multivariable Mendelian randomisation analysis. PLoS Med 17:e1003062. https://doi.org/10.1371/journal.pmed.1003062
Zhang Y, Pletcher MJ, Vittinghoff E, Clemons AM, Jacobs DJ, Allen NB et al (2021) Association between cumulative low-density lipoprotein cholesterol exposure during young adulthood and middle age and risk of cardiovascular events. JAMA Cardiol 6:1406–1413. https://doi.org/10.1001/jamacardio.2021.3508
Libby P, Hansson GK (2019) From focal lipid storage to systemic inflammation: JACC review topic of the week. J Am Coll Cardiol 74:1594–1607. https://doi.org/10.1016/j.jacc.2019.07.061
Kong P, Cui ZY, Huang XF, Zhang DD, Guo RJ, Han M (2022) Inflammation and atherosclerosis: signaling pathways and therapeutic intervention. Signal Transduct Target Ther 7:131. https://doi.org/10.1038/s41392-022-00955-7
Vallejo J, Cochain C, Zernecke A, Ley K (2021) Heterogeneity of immune cells in human atherosclerosis revealed by scRNA-Seq. Cardiovasc Res 117:2537–2543. https://doi.org/10.1093/cvr/cvab260
Ridker PM (2016) A test in context: high-sensitivity C-reactive protein. J Am Coll Cardiol 67:712–723. https://doi.org/10.1016/j.jacc.2015.11.037
Abbate A, Toldo S, Marchetti C, Kron J, Van Tassell BW, Dinarello CA (2020) Interleukin-1 and the inflammasome as therapeutic targets in cardiovascular disease. Circ Res 126:1260–1280. https://doi.org/10.1161/CIRCRESAHA.120.315937
Kim CW, Oh ET, Park HJ (2021) A strategy to prevent atherosclerosis via TNF receptor regulation. FASEB J 35:e21391. https://doi.org/10.1096/fj.202000764R
Ridker PM, Rane M (2021) Interleukin-6 signaling and anti-interleukin-6 therapeutics in cardiovascular disease. Circ Res 128:1728–1746. https://doi.org/10.1161/CIRCRESAHA.121.319077
Ketelhuth D, Lutgens E, Bäck M, Binder CJ, Van den Bossche J, Daniel C et al (2019) Immunometabolism and atherosclerosis: perspectives and clinical significance: a position paper from the Working Group on Atherosclerosis and Vascular Biology of the European Society of Cardiology. Cardiovasc Res 115:1385–1392. https://doi.org/10.1093/cvr/cvz166
Kim HW, Shi H, Winkler MA, Lee R, Weintraub NL (2020) Perivascular adipose tissue and vascular perturbation/atherosclerosis. Arterioscler Thromb Vasc Biol 40:2569–2576. https://doi.org/10.1161/ATVBAHA.120.312470
Gu P, Hui X, Zheng Q, Gao Y, Jin L, Jiang W et al (2021) Mitochondrial uncoupling protein 1 antagonizes atherosclerosis by blocking NLRP3 inflammasome-dependent interleukin-1β production. Sci Adv 7:eabl4024. https://doi.org/10.1126/sciadv.abl4024
Antoniades C, Tousoulis D, Vavlukis M, Fleming I, Duncker DJ, Eringa E et al (2023) Perivascular adipose tissue as a source of therapeutic targets and clinical biomarkers. Eur Heart J 44:3827–3844. https://doi.org/10.1093/eurheartj/ehad484
Meyers S, Crescente M, Verhamme P, Martinod K (2022) Staphylococcus aureus and neutrophil extracellular traps: the master manipulator meets its match in immunothrombosis. Arterioscler Thromb Vasc Biol 42:261–276. https://doi.org/10.1161/ATVBAHA.121.316930
Liew PX, Kubes P (2019) The neutrophil’s role during health and disease. Physiol Rev 99:1223–1248. https://doi.org/10.1152/physrev.00012.2018
Ravindran M, Khan MA, Palaniyar N (2019) Neutrophil extracellular trap formation: physiology, pathology, and pharmacology. Biomolecules. https://doi.org/10.3390/biom9080365
Papayannopoulos V (2018) Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol 18:134–147. https://doi.org/10.1038/nri.2017.105
Chen JL, Tong Y, Zhu Q, Gao LQ, Sun Y (2022) Neutrophil extracellular traps induced by Porphyromonas gingivalis lipopolysaccharide modulate inflammatory responses via a Ca2+-dependent pathway. Arch Oral Biol 141:105467. https://doi.org/10.1016/j.archoralbio.2022.105467
Wang Z, Zhu D, Zhang Y, Xia F, Zhu J, Dai J et al (2023) Extracellular vesicles produced by avian pathogenic Escherichia coli (APEC) activate macrophage proinflammatory response and neutrophil extracellular trap (NET) formation through TLR4 signaling. Microb Cell Fact 22:177. https://doi.org/10.1186/s12934-023-02171-6
Liao TL, Liu HJ, Chen DY, Tang KT, Chen YM, Liu PY (2023) SARS-CoV-2 primed platelets-derived microRNAs enhance NETs formation by extracellular vesicle transmission and TLR7/8 activation. Cell Commun Signal 21:304. https://doi.org/10.1186/s12964-023-01345-4
Unger L, Skoluda S, Backman E, Amulic B, Ponce-Garcia FM, Etiaba CN et al (2023) Candida albicans induces neutrophil extracellular traps and leucotoxic hypercitrullination via candidalysin. EMBO Rep 24:e57571. https://doi.org/10.15252/embr.202357571
Döring Y, Libby P, Soehnlein O (2020) Neutrophil extracellular traps participate in cardiovascular diseases: recent experimental and clinical insights. Circ Res 126:1228–1241. https://doi.org/10.1161/CIRCRESAHA.120.315931
Li P, Li M, Lindberg MR, Kennett MJ, Xiong N, Wang Y (2010) PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps. J Exp Med 207:1853–1862. https://doi.org/10.1084/jem.20100239
Arroyo R, Khan MA, Echaide M, Pérez-Gil J, Palaniyar N (2019) SP-D attenuates LPS-induced formation of human neutrophil extracellular traps (NETs), protecting pulmonary surfactant inactivation by NETs. Commun Biol 2:470. https://doi.org/10.1038/s42003-019-0662-5
Bonnekoh H, Scheffel J, Wu J, Hoffmann S, Maurer M, Krause K (2019) Skin and systemic inflammation in schnitzler’s syndrome are associated with neutrophil extracellular trap formation. Front Immunol 10:546. https://doi.org/10.3389/fimmu.2019.00546
Dömer D, Walther T, Möller S, Behnen M, Laskay T (2021) Neutrophil extracellular traps activate proinflammatory functions of human neutrophils. Front Immunol 12:636954. https://doi.org/10.3389/fimmu.2021.636954
Kiss MG, Binder CJ (2022) The multifaceted impact of complement on atherosclerosis. Atherosclerosis 351:29–40. https://doi.org/10.1016/j.atherosclerosis.2022.03.014
Biros E, Reznik JE, Moran CS (2022) Role of inflammatory cytokines in genesis and treatment of atherosclerosis. Trends Cardiovasc Med 32:138–142. https://doi.org/10.1016/j.tcm.2021.02.001
Liu L, Shi Z, Ji X, Zhang W, Luan J, Zahr T et al (2022) Adipokines, adiposity, and atherosclerosis. Cell Mol Life Sci 79:272. https://doi.org/10.1007/s00018-022-04286-2
La Sala L, Prattichizzo F, Ceriello A (2019) The link between diabetes and atherosclerosis. Eur J Prev Cardiol 26:15–24. https://doi.org/10.1177/2047487319878373
Shemiakova T, Ivanova E, Grechko AV, Gerasimova EV, Sobenin IA, Orekhov AN (2020) Mitochondrial dysfunction and dna damage in the context of pathogenesis of atherosclerosis. Biomedicines. https://doi.org/10.3390/biomedicines8060166
Wang Y, Wang W, Wang N, Tall AR, Tabas I (2017) Mitochondrial oxidative stress promotes atherosclerosis and neutrophil extracellular traps in aged mice. Arterioscler Thromb Vasc Biol 37:e99–e107. https://doi.org/10.1161/ATVBAHA.117.309580
Chen Y, Li X, Lin X, Liang H, Liu X, Zhang X et al (2022) Complement C5a induces the generation of neutrophil extracellular traps by inhibiting mitochondrial STAT3 to promote the development of arterial thrombosis. Thromb J 20:24. https://doi.org/10.1186/s12959-022-00384-0
Kim J, Gupta R, Blanco LP, Yang S, Shteinfer-Kuzmine A, Wang K et al (2019) VDAC oligomers form mitochondrial pores to release mtDNA fragments and promote lupus-like disease. Science 366:1531–1536. https://doi.org/10.1126/science.aav4011
Lood C, Blanco LP, Purmalek MM, Carmona-Rivera C, De Ravin SS, Smith CK et al (2016) Neutrophil extracellular traps enriched in oxidized mitochondrial DNA are interferogenic and contribute to lupus-like disease. Nat Med 22:146–153. https://doi.org/10.1038/nm.4027
Liu L, Mao Y, Xu B, Zhang X, Fang C, Ma Y et al (2019) Induction of neutrophil extracellular traps during tissue injury: involvement of STING and Toll-like receptor 9 pathways. Cell Prolif 52:e12579. https://doi.org/10.1111/cpr.12579
Chrysanthopoulou A, Gkaliagkousi E, Lazaridis A, Arelaki S, Pateinakis P, Ntinopoulou M et al (2021) Angiotensin II triggers release of neutrophil extracellular traps, linking thromboinflammation with essential hypertension. JCI Insight. https://doi.org/10.1172/jci.insight.148668
Menegazzo L, Ciciliot S, Poncina N, Mazzucato M, Persano M, Bonora B et al (2015) NETosis is induced by high glucose and associated with type 2 diabetes. Acta Diabetol 52:497–503. https://doi.org/10.1007/s00592-014-0676-x
Awasthi D, Nagarkoti S, Kumar A, Dubey M, Singh AK, Pathak P et al (2016) Oxidized LDL induced extracellular trap formation in human neutrophils via TLR-PKC-IRAK-MAPK and NADPH-oxidase activation. Free Radic Biol Med 93:190–203. https://doi.org/10.1016/j.freeradbiomed.2016.01.004
Han H, Liu C, Li M, Wang J, Liu YS, Zhou Y et al (2022) Increased intracellular Cl(-) concentration mediates neutrophil extracellular traps formation in atherosclerotic cardiovascular diseases. Acta Pharmacol Sin 43:2848–2861. https://doi.org/10.1038/s41401-022-00911-9
Arita K, Shimizu T, Hashimoto H, Hidaka Y, Yamada M, Sato M (2006) Structural basis for histone N-terminal recognition by human peptidylarginine deiminase 4. Proc Natl Acad Sci USA 103:5291–5296. https://doi.org/10.1073/pnas.0509639103
Boeltz S, Amini P, Anders HJ, Andrade F, Bilyy R, Chatfield S et al (2019) To NET or not to NET:current opinions and state of the science regarding the formation of neutrophil extracellular traps. Cell Death Differ 26:395–408. https://doi.org/10.1038/s41418-018-0261-x
Osaka M, Deushi M, Aoyama J, Funakoshi T, Ishigami A, Yoshida M (2021) High-fat diet enhances neutrophil adhesion in LDLR-Null mice via hypercitrullination of histone H3. JACC Basic Transl Sci 6:507–523. https://doi.org/10.1016/j.jacbts.2021.04.002
Thiam HR, Wong SL, Qiu R, Kittisopikul M, Vahabikashi A, Goldman AE et al (2020) NETosis proceeds by cytoskeleton and endomembrane disassembly and PAD4-mediated chromatin decondensation and nuclear envelope rupture. Proc Natl Acad Sci USA 117:7326–7337. https://doi.org/10.1073/pnas.1909546117
Gupta AK, Giaglis S, Hasler P, Hahn S (2014) Efficient neutrophil extracellular trap induction requires mobilization of both intracellular and extracellular calcium pools and is modulated by cyclosporine A. PLoS one 9:e97088. https://doi.org/10.1371/journal.pone.0097088
Sprenkeler E, Tool A, Henriet S, van Bruggen R, Kuijpers TW (2022) Formation of neutrophil extracellular traps requires actin cytoskeleton rearrangements. Blood 139:3166–3180. https://doi.org/10.1182/blood.2021013565
Sun S, Zou X, Wang D, Liu Y, Zhang Z, Guo J et al (2023) IRGM/Irgm1 deficiency inhibits neutrophil-platelet interactions and thrombosis in experimental atherosclerosis and arterial injury. Biomed Pharmacother 158:114152. https://doi.org/10.1016/j.biopha.2022.114152
Papayannopoulos V, Metzler KD, Hakkim A, Zychlinsky A (2010) Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J Cell Biol 191:677–691. https://doi.org/10.1083/jcb.201006052
Shimonaga K, Matsushige T, Takahashi H, Hashimoto Y, Yoshiyama M, Ono C et al (2021) Peptidylarginine deiminase 4 as a Possible biomarker of plaque instability in carotid artery stenosis. J Stroke Cerebrovasc Dis 30:105816. https://doi.org/10.1016/j.jstrokecerebrovasdis.2021.105816
Hofbauer TM, Mangold A, Scherz T, Seidl V, Panzenböck A, Ondracek AS et al (2019) Neutrophil extracellular traps and fibrocytes in ST-segment elevation myocardial infarction. Basic Res Cardiol 114:33. https://doi.org/10.1007/s00395-019-0740-3
Liu J, Yang D, Wang X, Zhu Z, Wang T, Ma A et al (2019) Neutrophil extracellular traps and dsDNA predict outcomes among patients with ST-elevation myocardial infarction. Sci Rep 9:11599. https://doi.org/10.1038/s41598-019-47853-7
Mangold A, Alias S, Scherz T, Hofbauer M, Jakowitsch J, Panzenböck A et al (2015) Coronary neutrophil extracellular trap burden and deoxyribonuclease activity in ST-elevation acute coronary syndrome are predictors of ST-segment resolution and infarct size. Circ Res 116:1182–1192. https://doi.org/10.1161/CIRCRESAHA.116.304944
de Vries JJ, Autar A, van Dam-Nolen D, Donkel SJ, Kassem M, van der Kolk AG et al (2022) Association between plaque vulnerability and neutrophil extracellular traps (NETs) levels: the plaque At RISK study. PLoS one 17:e0269805. https://doi.org/10.1371/journal.pone.0269805
Ferré-Vallverdú M, Latorre AM, Fuset MP, Sánchez E, Madrid I, Ten F et al (2022) Neutrophil extracellular traps (NETs) in patients with STEMI. Association with percutaneous coronary intervention and antithrombotic treatments. Thromb Res 213:78–83. https://doi.org/10.1016/j.thromres.2022.03.002
Helseth R, Shetelig C, Andersen GØ, Langseth MS, Limalanathan S, Opstad TB et al (2019) Neutrophil extracellular trap components associate with infarct size, ventricular function, and clinical outcome in STEMI. Mediators Inflamm 2019:7816491. https://doi.org/10.1155/2019/7816491
Borissoff JI, Joosen IA, Versteylen MO, Brill A, Fuchs TA, Savchenko AS et al (2013) Elevated levels of circulating DNA and chromatin are independently associated with severe coronary atherosclerosis and a prothrombotic state. Arterioscler Thromb Vasc Biol 33:2032–2040. https://doi.org/10.1161/ATVBAHA.113.301627
Langseth MS, Helseth R, Ritschel V, Hansen CH, Andersen GØ, Eritsland J et al (2020) Double-stranded DNA and NETs components in relation to clinical outcome after ST-elevation myocardial infarction. Sci Rep 10:5007. https://doi.org/10.1038/s41598-020-61971-7
Langseth MS, Opstad TB, Bratseth V, Solheim S, Arnesen H, Pettersen AÅ et al (2018) Markers of neutrophil extracellular traps are associated with adverse clinical outcome in stable coronary artery disease. Eur J Prev Cardiol 25:762–769. https://doi.org/10.1177/2047487318760618
Kluge KE, Langseth MS, Opstad TB, Pettersen AÅ, Arnesen H, Tønnessen T et al (2020) Complement activation in association with markers of neutrophil extracellular traps and acute myocardial infarction in stable coronary artery disease. Mediators Inflamm 2020:5080743. https://doi.org/10.1155/2020/5080743
Lordan R, Tsoupras A, Zabetakis I (2021) Platelet activation and prothrombotic mediators at the nexus of inflammation and atherosclerosis: potential role of antiplatelet agents. Blood Rev 45:100694. https://doi.org/10.1016/j.blre.2020.100694
Constantinescu-Bercu A, Grassi L, Frontini M, Salles-Crawley II, Woollard K, Crawley JT (2020) Activated αIIbβ3 on platelets mediates flow-dependent NETosis via SLC44A2. Elife. https://doi.org/10.7554/eLife.53353
Pircher J, Engelmann B, Massberg S, Schulz C (2019) Platelet-neutrophil crosstalk in atherothrombosis. Thromb Haemost 119:1274–1282. https://doi.org/10.1055/s-0039-1692983
Wienkamp AK, Erpenbeck L, Rossaint J (2022) Platelets in the NETworks interweaving inflammation and thrombosis. Front Immunol 13:953129. https://doi.org/10.3389/fimmu.2022.953129
Etulain J, Martinod K, Wong SL, Cifuni SM, Schattner M, Wagner DD (2015) P-selectin promotes neutrophil extracellular trap formation in mice. Blood 126:242–246. https://doi.org/10.1182/blood-2015-01-624023
Sandoval-Pérez A, Berger R, Garaizar A, Farr SE, Brehm MA, König G et al (2020) DNA binds to a specific site of the adhesive blood-protein von Willebrand factor guided by electrostatic interactions. Nucleic Acids Res 48:7333–7344. https://doi.org/10.1093/nar/gkaa466
Alkarithi G, Duval C, Shi Y, Macrae FL, Ariëns R (2021) Thrombus structural composition in cardiovascular disease. Arterioscler Thromb Vasc Biol 41:2370–2383. https://doi.org/10.1161/ATVBAHA.120.315754
Joshi A, Schmidt LE, Burnap SA, Lu R, Chan MV, Armstrong PC et al (2022) Neutrophil-derived protein S100A8/A9 alters the platelet proteome in acute myocardial infarction and is associated with changes in platelet reactivity. Arterioscler Thromb Vasc Biol 42:49–62. https://doi.org/10.1161/ATVBAHA.121.317113
Dou H, Kotini A, Liu W, Fidler T, Endo-Umeda K, Sun X et al (2021) Oxidized phospholipids promote NETosis and arterial thrombosis in LNK(SH2B3) deficiency. Circulation 144:1940–1954. https://doi.org/10.1161/CIRCULATIONAHA.121.056414
Oggero S, de Gaetano M, Marcone S, Fitzsimons S, Pinto AL, Ikramova D et al (2021) Extracellular vesicles from monocyte/platelet aggregates modulate human atherosclerotic plaque reactivity. J Extracell Vesicles 10:12084. https://doi.org/10.1002/jev2.12084
Momi S, Falcinelli E, Petito E, Ciarrocca TG, Ossoli A, Gresele P (2022) Matrix metalloproteinase-2 on activated platelets triggers endothelial PAR-1 initiating atherosclerosis. Eur Heart J 43:504–514. https://doi.org/10.1093/eurheartj/ehab631
Koelwyn GJ, Corr EM, Erbay E, Moore KJ (2018) Regulation of macrophage immunometabolism in atherosclerosis. Nat Immunol 19:526–537. https://doi.org/10.1038/s41590-018-0113-3
Kuznetsova T, Prange K, Glass CK, de Winther M (2020) Transcriptional and epigenetic regulation of macrophages in atherosclerosis. Nat Rev Cardiol 17:216–228. https://doi.org/10.1038/s41569-019-0265-3
Schumski A, Ortega-Gómez A, Wichapong K, Winter C, Lemnitzer P, Viola JR et al (2021) Endotoxinemia accelerates atherosclerosis through electrostatic charge-mediated monocyte adhesion. Circulation 143:254–266. https://doi.org/10.1161/CIRCULATIONAHA.120.046677
Tucker B, Ephraums J, King TW, Abburi K, Rye KA, Cochran BJ (2023) Impact of impaired cholesterol homeostasis on neutrophils in atherosclerosis. Arterioscler Thromb Vasc Biol 43:618–627. https://doi.org/10.1161/ATVBAHA.123.316246
Lazzaretto B, Fadeel B (2019) Intra- and extracellular degradation of neutrophil extracellular traps by macrophages and dendritic cells. J Immunol 203:2276–2290. https://doi.org/10.4049/jimmunol.1800159
Tsourouktsoglou TD, Warnatsch A, Ioannou M, Hoving D, Wang Q, Papayannopoulos V (2020) Histones, DNA, and citrullination promote neutrophil extracellular trap inflammation by regulating the localization and activation of TLR4. Cell Rep 31:107602. https://doi.org/10.1016/j.celrep.2020.107602
Warnatsch A, Ioannou M, Wang Q, Papayannopoulos V (2015) Inflammation. neutrophil extracellular traps license macrophages for cytokine production in atherosclerosis. Science 349:316–320. https://doi.org/10.1126/science.aaa8064
Yalcinkaya M, Fotakis P, Liu W, Endo-Umeda K, Dou H, Abramowicz S et al (2023) Cholesterol accumulation in macrophages drives NETosis in atherosclerotic plaques via IL-1β secretion. Cardiovasc Res 119:969–981. https://doi.org/10.1093/cvr/cvac189
Josefs T, Barrett TJ, Brown EJ, Quezada A, Wu X, Voisin M et al (2020) Neutrophil extracellular traps promote macrophage inflammation and impair atherosclerosis resolution in diabetic mice. JCI Insight. https://doi.org/10.1172/jci.insight.134796
Conforti A, Wahlers T, Paunel-Görgülü A (2021) Neutrophil extracellular traps modulate inflammatory markers and uptake of oxidized LDL by human and murine macrophages. PLoS one 16:e0259894. https://doi.org/10.1371/journal.pone.0259894
Liu Q, Pan J, Bao L, Xu C, Qi Y, Jiang B et al (2022) Major vault protein prevents atherosclerotic plaque destabilization by suppressing macrophage ASK1-JNK signaling. Arterioscler Thromb Vasc Biol 42:580–596. https://doi.org/10.1161/ATVBAHA.121.316662
Jeon S, Kim TK, Jeong SJ, Jung IH, Kim N, Lee MN et al (2020) Anti-inflammatory actions of soluble ninjurin-1 ameliorate atherosclerosis. Circulation 142:1736–1751. https://doi.org/10.1161/CIRCULATIONAHA.120.046907
Sano M, Maejima Y, Nakagama S, Shiheido-Watanabe Y, Tamura N, Hirao K et al (2022) Neutrophil extracellular traps-mediated Beclin-1 suppression aggravates atherosclerosis by inhibiting macrophage autophagy. Front Cell Dev Biol 10:876147. https://doi.org/10.3389/fcell.2022.876147
Guo C, Davies MJ, Hawkins CL (2020) Role of thiocyanate in the modulation of myeloperoxidase-derived oxidant induced damage to macrophages. Redox Biol 36:101666. https://doi.org/10.1016/j.redox.2020.101666
Tabas I, Bornfeldt KE (2016) Macrophage phenotype and function in different stages of atherosclerosis. Circ Res 118:653–667. https://doi.org/10.1161/CIRCRESAHA.115.306256
Groh L, Keating ST, Joosten L, Netea MG, Riksen NP (2018) Monocyte and macrophage immunometabolism in atherosclerosis. Semin Immunopathol 40:203–214. https://doi.org/10.1007/s00281-017-0656-7
Gimbrone MJ, García-Cardeña G (2016) Endothelial cell dysfunction and the pathobiology of atherosclerosis. Circ Res 118:620–636. https://doi.org/10.1161/CIRCRESAHA.115.306301
Chatterjee V, Yang X, Ma Y, Cha B, Meegan JE, Wu M et al (2020) Endothelial microvesicles carrying Src-rich cargo impair adherens junction integrity and cytoskeleton homeostasis. Cardiovasc Res 116:1525–1538. https://doi.org/10.1093/cvr/cvz238
Winkler MJ, Müller P, Sharifi AM, Wobst J, Winter H, Mokry M et al (2020) Functional investigation of the coronary artery disease gene SVEP1. Basic Res Cardiol 115:67. https://doi.org/10.1007/s00395-020-00828-6
Karshovska E, Mohibullah R, Zhu M, Zahedi F, Thomas D, Magkrioti C et al (2022) Endothelial ENPP2 (Ectonucleotide Pyrophosphatase/Phosphodiesterase 2) increases atherosclerosis in female and male mice. Arterioscler Thromb Vasc Biol 42:1023–1036. https://doi.org/10.1161/ATVBAHA.122.317682
Aoyama J, Osaka M, Deushi M, Hosoya S, Ishigami A, Maehara T et al (2022) CXCL1-triggered PAD4 cytoplasmic translocation enhances neutrophil adhesion through citrullination of PDIA1. J Atheroscler Thromb 29:1307–1318. https://doi.org/10.5551/jat.63237
Gao H, Wang X, Lin C, An Z, Yu J, Cao H et al (2020) Exosomal MALAT1 derived from ox-LDL-treated endothelial cells induce neutrophil extracellular traps to aggravate atherosclerosis. Biol Chem 401:367–376. https://doi.org/10.1515/hsz-2019-0219
Chen L, Hu L, Li Q, Ma J, Li H (2019) Exosome-encapsulated miR-505 from ox-LDL-treated vascular endothelial cells aggravates atherosclerosis by inducing NET formation. Acta Biochim Biophys Sin (Shanghai) 51:1233–1241. https://doi.org/10.1093/abbs/gmz123
Quillard T, Araújo HA, Franck G, Shvartz E, Sukhova G, Libby P (2015) TLR2 and neutrophils potentiate endothelial stress, apoptosis and detachment: implications for superficial erosion. Eur Heart J 36:1394–1404. https://doi.org/10.1093/eurheartj/ehv044
Franck G, Mawson TL, Folco EJ, Molinaro R, Ruvkun V, Engelbertsen D et al (2018) Roles of PAD4 and NETosis in experimental atherosclerosis and arterial injury: implications for superficial erosion. Circ Res 123:33–42. https://doi.org/10.1161/CIRCRESAHA.117.312494
Chen X, Duong MN, Nicholls SJ, Bursill C (2018) Myeloperoxidase modification of high-density lipoprotein suppresses human endothelial cell proliferation and migration via inhibition of ERK1/2 and Akt activation. Atherosclerosis 273:75–83. https://doi.org/10.1016/j.atherosclerosis.2018.04.006
Huang Y, DiDonato JA, Levison BS, Schmitt D, Li L, Wu Y et al (2014) An abundant dysfunctional apolipoprotein A1 in human atheroma. Nat Med 20:193–203. https://doi.org/10.1038/nm.3459
Nybo T, Dieterich S, Gamon LF, Chuang CY, Hammer A, Hoefler G et al (2019) Chlorination and oxidation of the extracellular matrix protein laminin and basement membrane extracts by hypochlorous acid and myeloperoxidase. Redox Biol 20:496–513. https://doi.org/10.1016/j.redox.2018.10.022
Abdo AI, Rayner BS, van Reyk DM, Hawkins CL (2017) Low-density lipoprotein modified by myeloperoxidase oxidants induces endothelial dysfunction. Redox Biol 13:623–632. https://doi.org/10.1016/j.redox.2017.08.004
Folco EJ, Mawson TL, Vromman A, Bernardes-Souza B, Franck G, Persson O et al (2018) Neutrophil extracellular traps induce endothelial cell activation and tissue factor production through interleukin-1α and cathepsin G. Arterioscler Thromb Vasc Biol 38:1901–1912. https://doi.org/10.1161/ATVBAHA.118.311150
Miano JM, Fisher EA, Majesky MW (2021) Fate and State of vascular smooth muscle cells in atherosclerosis. Circulation 143:2110–2116. https://doi.org/10.1161/CIRCULATIONAHA.120.049922
Basatemur GL, Jørgensen HF, Clarke M, Bennett MR, Mallat Z (2019) Vascular smooth muscle cells in atherosclerosis. Nat Rev Cardiol 16:727–744. https://doi.org/10.1038/s41569-019-0227-9
Grootaert M, Bennett MR (2021) Vascular smooth muscle cells in atherosclerosis: time for a re-assessment. Cardiovasc Res 117:2326–2339. https://doi.org/10.1093/cvr/cvab046
Silvestre-Roig C, Braster Q, Ortega-Gomez A, Soehnlein O (2020) Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol 17:327–340. https://doi.org/10.1038/s41569-019-0326-7
Oesterle A, Bowman MA (2015) S100A12 and the S100/calgranulins: emerging biomarkers for atherosclerosis and possibly therapeutic targets. Arterioscler Thromb Vasc Biol 35:2496–2507. https://doi.org/10.1161/ATVBAHA.115.302072
Faria A, Persaud SJ (2017) Cardiac oxidative stress in diabetes: Mechanisms and therapeutic potential. Pharmacol Ther 172:50–62. https://doi.org/10.1016/j.pharmthera.2016.11.013
Chellan B, Sutton NR, Hofmann BM (2018) S100/RAGE-mediated inflammation and modified cholesterol lipoproteins as mediators of osteoblastic differentiation of vascular smooth muscle cells. Front Cardiovasc Med 5:163. https://doi.org/10.3389/fcvm.2018.00163
Silvestre-Roig C, Braster Q, Wichapong K, Lee EY, Teulon JM, Berrebeh N et al (2019) Externalized histone H4 orchestrates chronic inflammation by inducing lytic cell death. Nature 569:236–240. https://doi.org/10.1038/s41586-019-1167-6
Cai H, Chuang CY, Vanichkitrungruang S, Hawkins CL, Davies MJ (2019) Hypochlorous acid-modified extracellular matrix contributes to the behavioral switching of human coronary artery smooth muscle cells. Free Radic Biol Med 134:516–526. https://doi.org/10.1016/j.freeradbiomed.2019.01.044
Vasilyev N, Williams T, Brennan ML, Unzek S, Zhou X, Heinecke JW et al (2005) Myeloperoxidase-generated oxidants modulate left ventricular remodeling but not infarct size after myocardial infarction. Circulation 112:2812–2820. https://doi.org/10.1161/CIRCULATIONAHA.105.542340
Cheng D, Talib J, Stanley CP, Rashid I, Michaëlsson E, Lindstedt EL et al (2019) Inhibition of MPO (Myeloperoxidase) attenuates endothelial dysfunction in mouse models of vascular inflammation and atherosclerosis. Arterioscler Thromb Vasc Biol 39:1448–1457. https://doi.org/10.1161/ATVBAHA.119.312725
Yang M, Chen Q, Mei L, Wen G, An W, Zhou X et al (2021) Neutrophil elastase promotes neointimal hyperplasia by targeting toll-like receptor 4 (TLR4)-NF-κB signalling. Br J Pharmacol 178:4048–4068. https://doi.org/10.1111/bph.15583
Yang S, Chen L, Wang Z, Chen J, Ni Q, Guo X et al (2023) Neutrophil extracellular traps induce abdominal aortic aneurysm formation by promoting the synthetic and proinflammatory smooth muscle cell phenotype via Hippo-YAP pathway. Transl Res 255:85–96. https://doi.org/10.1016/j.trsl.2022.11.010
Zhai M, Gong S, Luan P, Shi Y, Kou W, Zeng Y et al (2022) Extracellular traps from activated vascular smooth muscle cells drive the progression of atherosclerosis. Nat Commun 13:7500. https://doi.org/10.1038/s41467-022-35330-1
Imazio M, Nidorf M (2021) Colchicine and the heart. Eur Heart J 42:2745–2760. https://doi.org/10.1093/eurheartj/ehab221
Knight JS, Luo W, O’Dell AA, Yalavarthi S, Zhao W, Subramanian V et al (2014) Peptidylarginine deiminase inhibition reduces vascular damage and modulates innate immune responses in murine models of atherosclerosis. Circ Res 114:947–956. https://doi.org/10.1161/CIRCRESAHA.114.303312
Molinaro R, Yu M, Sausen G, Bichsel CA, Corbo C, Folco EJ et al (2021) Targeted delivery of protein arginine deiminase-4 inhibitors to limit arterial intimal NETosis and preserve endothelial integrity. Cardiovasc Res 117:2652–2663. https://doi.org/10.1093/cvr/cvab074
Van Avondt K, Maegdefessel L, Soehnlein O (2019) Therapeutic targeting of neutrophil extracellular traps in atherogenic inflammation. Thromb Haemost 119:542–552. https://doi.org/10.1055/s-0039-1678664
Chen J, Du R, Wang W, An F, Ye L, Chen H et al (2020) The anti-inflammatory effects of Pseudorlaric acid D on atherosclerosis. Biomed Pharmacother 125:109993. https://doi.org/10.1016/j.biopha.2020.109993
Liberale L, Holy EW, Akhmedov A, Bonetti NR, Nietlispach F, Matter CM et al (2019) Interleukin-1β mediates arterial thrombus formation via NET-Associated tissue factor. J Clin Med. https://doi.org/10.3390/jcm8122072
Tong W, Hui H, Shang W, Zhang Y, Tian F, Ma Q et al (2021) Highly sensitive magnetic particle imaging of vulnerable atherosclerotic plaque with active myeloperoxidase-targeted nanoparticles. Theranostics 11:506–521. https://doi.org/10.7150/thno.49812
Chen W, Tumanov S, Kong S, Cheng D, Michaëlsson E, Bongers A et al (2022) Therapeutic inhibition of MPO stabilizes pre-existing high risk atherosclerotic plaque. Redox Biol 58:102532. https://doi.org/10.1016/j.redox.2022.102532
Rashid I, Maghzal GJ, Chen YC, Cheng D, Talib J, Newington D et al (2018) Myeloperoxidase is a potential molecular imaging and therapeutic target for the identification and stabilization of high-risk atherosclerotic plaque. Eur Heart J 39:3301–3310. https://doi.org/10.1093/eurheartj/ehy419
Chirivi R, van Rosmalen J, van der Linden M, Euler M, Schmets G, Bogatkevich G et al (2021) Therapeutic ACPA inhibits NET formation: a potential therapy for neutrophil-mediated inflammatory diseases. Cell Mol Immunol 18:1528–1544. https://doi.org/10.1038/s41423-020-0381-3
Hallberg L, Barlous K, Hawkins CL (2023) Antioxidant strategies to modulate NETosis and the release of neutrophil extracellular traps during chronic inflammation. Antioxidants (Basel). https://doi.org/10.3390/antiox12020478
Chu Z, Huang Q, Ma K, Liu X, Zhang W, Cui S et al (2023) Novel neutrophil extracellular trap-related mechanisms in diabetic wounds inspire a promising treatment strategy with hypoxia-challenged small extracellular vesicles. Bioact Mater 27:257–270. https://doi.org/10.1016/j.bioactmat.2023.04.007
Acknowledgements
This work was supported by Jiangsu Provincial Health Commission Fund Project and Jiangsu Provincial Medical Youth Talent (Grant numbers [H2018004] and [QNRC2016837]). Figures were created in BioRender.com and Pymol 2.4.1 software.
Funding
This work was supported by Jiangsu Provincial Health Commission Fund Project and Jiangsu Provincial Medical Youth Talent (Grant Numbers [H2018004] and [QNRC2016837]).
Author information
Authors and Affiliations
Contributions
YW contributed toward manuscript writing & editing, JL contributed toward manuscript writing & Editing, and CW contributed toward editing and final approval of manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Wang, C. & Li, J. Neutrophil extracellular traps: a catalyst for atherosclerosis. Mol Cell Biochem (2024). https://doi.org/10.1007/s11010-024-04931-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11010-024-04931-3