Abstract
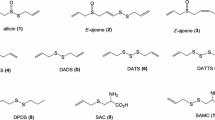
Allium species, belonging to Alliaceae family, are among the oldest cultivated vegetables used as food. Garlic, onions, leeks and chives, which belong to this family, have been reported to have medicinal properties. The Allium species constituents have been shown to have antibacterial and antioxidant activities, and, in addition, other biological properties. These activities are related to their rich organosulfur compounds. These organosulfur compounds are believed to prevent the development of cancer, cardiovascular, neurological, diabetes, liver diseases as well as allergy and arthritis. There have also been reports on toxicities of these compounds. The major active compounds of Allium species includes, diallyl disulfide, diallyl trisulfide, diallyl sulfide, dipropyl disulfide, dipropyl trisulfide, 1-propenylpropyl disulfide, allyl methyl disulfide and dimethyl disulfide. The aim of this review is to focus on a variety of experimental and clinical reports on the effectiveness, toxicities and possible mechanisms of actions of the active compounds of garlic, onions, leek and chives.



Similar content being viewed by others
References
Tepe B, Sokmen M, Akpulat H, Sokman A (2005) In vitro antioxidant activities of the methanol extracts of five species from Turkey. Food Chem 92:89–92
Benkeblia N (2004) Antimicrobial activity of essential oil extracts of various onions (Allium cepa) and garlic (Allium sativum). LWT-Food Sci Technol 37(2):263–268
Augusti KT, Mathew PT (1974) lipid lowering effect of allicin (diallyl disulfide oxide) on long-term feeding in normal rats. Experentia 30:468–470
Lanzotti V (2006) The analysis of onion and garlic. J Chromatogr A 1112:3–22
Corzo-Martinez M, Corzo N, Villamiel M (2007) Biological properties of onions and garlic. Trends Food Sci Technol 18:609–625
Yin M, Cheng W (1998) Antioxidant activity of several Allium members. J Agric Food Chem 46:4097–4101
Yin M, Cheng W (2003) Antioxidant and antimicrobial effects of four garlic-derived organosulfur compounds in ground beef. Meat Sci 63:23–28
Iranshahi M (2012) A review of volatile sulfur-containing compounds from terrestrial plants: biosynthesis, distribution and analytical methods. J Essen oil Res 24(4):393–434. https://doi.org/10.1080/10412905.2012.692918
Milner JA (1996) Garlic: Its carcinogenic and antitumor properties. Nutr Rev 54:S82–S86. https://doi.org/10.1111/j.1753-4887.1996.tb03823.x
Block E (1985) The chemistry of garlic and onion. Sci Amer 252:114–119
Ye CL, Dai DH, Hu WL (2013) Antimicrobial and antioxidant activities of the essential oil from onion (Allium cepa). Food Control 30:48–53
Rattanachaikunsopon P, Phumkhachorn P (2008) Diallyl sulfide content and antimicrobial activity against food-borne pathogenis bacteria of chives (Allium Schoenoprasum). Bionsci Biotechnol Biochem 72:2987–2991
Stajner D, Milic-DeMarino M, Canadanovic- Brunet J (2003) Screening for antioxidant properties of leeks, Allium sphaerocephalon L. J Herbs Spices Med Plants 10:75–82
Fista G, Bloukas J, Siomos A (2004) Effect of leek and onion on processing and quality characteristics of Greek traditional sausages. Meat Sci 68:163–172
Amin M, Kapadnis BP (2005) Heat stable antimicrobial activity of Allium ascalonicum against bacteria and fungi. Ind J Exp Biol 43:751–754
Leelarungrayub N, Rattanapone V, Chanarat N, Gebicki JM (2006) Quantitative evaluation of the antioxidant properties of garlic and shallot preparations. Nutrition 22:266–274
Yabuki Y, Mukaida Y, Saito Y, Oshima K, Takashima T, Muroi E, Hashimoto K, Uda Y (2010) Characterisation of volatile sulfur-containing compounds generated in crushed leaves of Chinese chive (Allium tuberosum Rottler). Food Chem 120:343–348
Bernaert N, de Paepe D, Bouten C, de Clercq H, Steward D, van Bockstaele E, de Loose M, van Droogenbroeck B (2012) Antioxidant capacity, total phenolic and ascorbate content as a function of the genetic diversity of leek (Allium ampeloprasum var. porrum). Food Chem 134:669–677
Rattanachaikunsopon P, Phumkhachorn P (2009) Shallot (Allium ascalonicum L.) oil: diallyl sulfide content and antimicrobial activity against food-borne pathogenic bacteria. Afr J Microbiol Res 3:747–750. https://doi.org/10.5897/AJMR.9000657
Fulder S, Blackwood J (2000) Garlic: nature’s original remedy. Healing Arts Press, US
Bianchini F, Vainio H (2001) Allium vegetables and organosulfur compounds; do they help prevent cancer? Environ Health Perspect 109:893–902
Świderski F, Dabrowska M, Rusaczonek A, Waszkiewicz-Robak B (2007) Bioactive substances of garlic and their role in dietoprophylaxis and dietotherapy. Roczn PZH 58(1):41–46
Lutomski J (2001) Garlic fascination—yesterday and today. Post Fitoter 1, pp 7–14 (Polish). http://www.czytelniamedyczna.pl/2459,fascynacja-czosnkiem-wczoraj-i-dzis.html
Barnes J, Anderson LA, Phillipson JD (1996) Herbal medicines: a guide for health-care professionals, vol 9. Pharmaceutical Press, London, p 296
Mnayar D, Fabiono-Tixier AS, Petitcolas E, Hamieh T, Nehme N, Ferrant C, Fernandez X, Chemat F (2014) Chemical composition, antibacterial and antioxidant activities of six essential oils from the Alliaceae family. Molecules 19:20034–20053. https://doi.org/10.3390/molecules191220034
Virtanen AL, Matikkala EJ (1976) The isolation of S-methyl-L-cysteine and S-n-propyl-L-cysteine sulphoxide from onion (Allium cepa) and the antibiotic activity of crushed onion. Acta Chem Scand 13:1898–1900
Casella S, Leonardi M, Melai B, Fratini F, Pistelli L (2012) The role of diallyl sulfides and dipropyl sulfides in the In vitro antimicrobial activity of the essential oil of garlic, Allium sativum L., and Allium porrum L. Phytother Res 27:380–383
As W, Pensky J (1958) Tumor inhibition by a sulfhydryl-blocking agent related to an active principle of garlic (Allium sativum). Cancer Res 18:1301–1308
Gilbert HF (1990) Molecular and cellular aspects of thiol-disulfide exchange. Adv Enzymol Relat Areas Mol Biol 63:69–172
Keusgen M (2011) Volatile compounds of the genus Allium L (onions). In: Qian M (ed) Volatile sulfur compounds in food. American Chemical Society, Washington, p 183
Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils—a review. Food Chem Toxicol 46:446–475
Seow YX, Yeo CR, Chung HL, Yuk HG (2014) Plant essential oils as active antimicrobial agents. Crit Rev Food Sci 54:625–644
Amagase H (2006) Clarifying the real bioactive constituents of garlic. J Nutr 136:716S-725S
Milner JA (2006) Preclinical perspective on garlic and cancer. J Nutr 136:827S-831S
Borek C (2001) Antioxidant health effects of aged garlic extract. J Nutr 131:1010–1015
Mei X, Wang MC, Xu HX, Pan XP, Gao CY, Han N, Fu MY (1982) Garlic and gastric cancer—the effect of garlic on nitrite and nitrate in gastric juice. Acta Nutr Sin 4:53–58
Takezaki T, Gao CM, Ding JH, Liu TK, Li MS, Tajima K (1999) Comparative studies of lifestyles of residents of high and low risk areas for gastric cancer in Jiangsu Province, China: with special reference to Allium vegetables. J Epidemiol 9:297–305
Haenszet W, Kurhara M, Segi M, Lee RK (1972) Stomach cancer among Japanese in Hawaii. J Natl Cancer Inst 49:969–988
Trichopoulos D, Quranos G, Day NE, Tzonou A, Manousos O, Papadimitriou C, Trichopolos A (1985) Diet and cancer of the stomach: a case–control study in Greece. Int J Cancer 36:291–297
Tajima K, Tominaga S (1985) Dietary habits and gastro-intestinal cancers: a comparative case-control study of stomach and large intestinal cancers in Nagaya, Japan. Jpn J Cancer Res 76:705–716
Haenszel W, Locke FB, Segi M (1980) A case-control study of large bowel cancer in Japan. J Natl Cancer Inst 64:17–22
Tuyns AJ, Kaaks R, Haelterman M (1988) Colorectal cancer and the consumption of foods: a case-control study in Belgium. Nutr Cancer 11:189–204
Levi F, Pasche C, La Vecchia C, Lucchini F, Franceschi S (1999) Food groups and colorectal cancer risk. Br J Cancer 79:1283–1287
Levi V, La Vecchia C, Gulle C, Negri E (1993) Dietary factors and breast cancer risk in Vausi, Switzerland. Nutr Cancer 19:327–335
Challier B, Peranau JM, Viel JF (1993) Garlic, onion and cereal fiber as protective factors for breast cancer: a French case-control study. Eur J Epidemiol 14:737–747
Katsouyanni K, Trichopolos D, Boyle P, Xirouchaki E, Trichopolou A, Lisseos B, Vasilaros S, MacMahon B (1986) Diet and breast cancer: a case-control study in Greece. Int J Cancer 38:815–820
Dorant E, van den Brand PA, Oldbohm RA (1995) Allium vegetable consumption, garlic supplement intake, and female breast carcinoma incidence. Breast Cancer Res Treat 33:163–170
Sankaranarayanan R, Varghese C, Duffy SW, Padmakumary G, Day NE, Nair MK (1994) A case-control study of diet and lung cancer in Kerala, South India. Int J Cancer 58:644–649
Hu J, La Vecchia C, Nefri E, Chalenoud L, Boselli C, Jia X, Liu R, Huang G, Bi D, Wang C (1999) Diet and brain cancer in adults: a case-control study in northeast China. Int J Cancer . https://doi.org/10.1002/(sici)1097-0215(19990331)81:1%3c20::aid-ijc4%3e3.0.co;2-2
Zheng W, Blot WJ, Shu XO, Gao YT, Ji BT, Ziegher RG, Fraumeni J (1992) Diet and other risk factors for laryngeal cancer in Shanghai, China. Am J Epidemiol 136:178–191
Key TJ, Silcocks PB, Davey GK, Appleby PN, Bishop DT (1997) A case-control study of diet and prostate cancer. Br J Cancer 76:678–687
Shu XO, Zheng W, Polischman N, Brinton LA, Hatch MC, Gao YT, Fraumeni JF Jr (1993) A population based case-control of dietary factors and endometrial cancer in Shanghai, People’s Republic of China. Am J Epidemiol 137:155–156
Sparnins VL, Barany G, Wattenberg LW (1988) Effects of organosulfur compounds from garlic and onions on benzo[a]pyrene-induced neoplasia and glutathione S- transferase activity in the mouse. Carcinogenesis 9:131–134
Sumiyoshi H, Wargovich MJ (1990) Chemoprevention of 1,2-dimethylhydrazine-induced colon cancer in mice by naturally occurring organosulfur compounds. Cancer Res 50(16):5084–5087
Hong YS, Ham YA, Choi JH, Kim J (2000) Effects of allyl sulfur compounds and garlic extract on the expression of Bcl-2, Bax, and p53 in non-small cell lung cancer cell lines. Exp Mol Med 32:127–134
Takahashi S, Hakoi K, Yada H, Hirose M, Ito N, Fukushima S (1992) Enhancing effects of diallyl sulfide on hepatocarcinogenesis and inhibitory actions of the related diallyl disulfide on colon and renal carcinogenesis in rats. Carcinogenesis 13:1513–1518
Wargovich MJ, Woods C, Eng WW, Stephens LC, Gray K (1988) Chemoprevention of N-nitrosomethylbenzylamine-induced esophageal cancer in rats by naturally occurring thioether, diallyl sulfide. Cancer Res 48:6872–6875
Ip C, Lisk DJ, Stoewsand GS (1992) Mammary cancer prevention by regular garlic and selenium-enriched garlic. Nutr Cancer 17:279–286
Liu J, Lin RI, Milner JA (1992) Inhibition of 7, 12-dimethylbenz[a]anthracene-induced mammary tumors and DNA adducts by garlic powder. Carcinogenesis 13:1847–1851
Schaffer EM, Liu JZ, Green J, Dangler CA, Miler JA (1996) Garlic and associated allyl sulfur components inhibit N-methyl-N-nitrosourea-induced rat mammary carcinogenesis. Cancer Lett 102:199–204
Cohen LA, Zhao Z, Pittman B, Lubel R (1999) S-allylcysteine, garlic constituent, fails to inhibit N-methylnitrosourea-induced rat mammary tumorigenesis. Nutr Cancer 35:58–63
Wattenberg LW, Sparnins VL, Barany G (1989) Inhibition of N-nitrosodiethylamine carcinogenesis in mice by naturally occurring organosulfur compounds and monoterpenes. Cancer Res 49:2689–2692
Sadhana AS, Rao AR, Kucheria K, Bijani V (1988) Inhibitory action of garlic oil on the initiation of benzo[a]pyrene-induced skin carcinogenesis in mice. Cancer Lett 40(2):193–197. https://doi.org/10.1016/0304-3835(88)90010-9
Singh A, Shukla Y (1998) Antitumor activity of diallyl sulfide on polycyclic hydrocarbon-induced mouse skin carcinogenesis. Cancer Lett 131:209–214
Cope KA, Milner JA, Seifried R, Seifried HE, Harrison EH (2006) Effects of garlic ingestion in humans on the formation of urinary nitrosoproline. FASEB J 20:A152–A152. https://doi.org/10.1096/fasebj.20.4.A152-a
Amagase H, Schaffer EM, Milner JA (1996) Dietary components modify the ability of garlic to suppress 7,12-dimethylbenz[a]anthracene-induced mammary DNA adducts. J Nutr 126:817–824
Pinto JT, Qiago C, Xing J, Suffoletto BP, Schubert KB, Rivlin RS, Huryk RF, Bacich DJ, Heston WD (2000) Alteration of prostate biomarker expression and testosterone utilization in human LBCaP prostate carcinoma cells by garlic derived S-allylmercaptocysteine. Prostate 45:304–314
Soni KB, Lahiri M, Chackradeo P, Bhide SV, Kuttan R (1997) protective effect of food additives on aflatoxin-induced mutagenicity and hepatocarcinogenicity. Cancer Lett 115:129–133
Zhang YS, Chen XR, Yu YN (1989) Antimutagenic effects of garlic (Allium sativum L.) on 4NQO-induced mutagenesis in Escherichia coli WP20. Mutat Res 227:215–219
Knasmuller S, de Martin R, Domjan G, Szakmary A (1989) Studies on the antimutagenic activities of garlic extract. Environ Mol Mutagen 13:357–385
Wargovich MJ (1987) Diallyl sulfide, a flavour component of garlic (Allium sativum), inhibits dimethylhydrazine-induced colon cancer. Carcinogenesis 8:487–489
Sparnins VL, Mott AW, Barany G, Wattenberg LW (1986) Effects of allyl methyl trisulfide on glutathione S-transferase activity and BP-induced neoplasia in the mouse. Nutr Cancer 8:211–215
Srivastava SK, Hu X, Xia H, Zaren HA, Chatterjee MI, Agarwal R, Singh SV (1997) Mechanism of differential efficacy of garlic organosulfides in preventing benzo[a]pyrene-induced cancer in mice. Cancer Lett 118:61–67
Munday R, Munday CM (1988) Low doses of diallyl disulfide, a compound derived from garlic, increase tissue activities of quinone reductase and glutathione transferase in the gastrointestinal tract of the rat. Nutr Cancer 34:42–48
Brady JF, Li DC, Ishizaki H, Yang CS (1988) Effect of diallyl sulfide on rat liver microsomal nitrosamine metabolism and other monooxygenase activities. Cancer Res 48:5937–5940
Brady JF, Wang MH, Hong JY, Xiao F, Li Y, Yoo JS, Ning SM, Lee MJ, Fukuto JM, Gapac JM (1991) Modulation of hepatic microsomal monooxygenase enzymes and cytotoxicity by diallyl sulfide. Toxicol Appl Pharmacol 108:342–354
Chung JG, Chen GW, Wu LT, Chang HL, Lin JG, Yeh CC, Wang TF (1998) Effects of garlic compounds diallyl sulfide and diallyl disulfide on arylamine N-Acetyltransferase activity in strains of helicobacter pylori form peptic ulcer patients. Am J Chin Med 26:353–364
Chung JG (1999) Effects of garlic components diallyl sulfide and diallyl disulfide on arylamine N-acetyltransferase activity in human bladder tumor cells. Drug Chem Toxicol 22:343–358. https://doi.org/10.3109/01480549909017839
Chen L, Hong JY, So E, Hussin AH, Cheng WF, Yang CS (1999) Decrease of hepatic catalase levels by treatment with diallyl sulfide and garlic homogenates in rats and mice. J Biochem Mol Toxicol 13:127–134
Arranz N, Haza AI, Garcia A, Moller L, Rafter J, Morales P (2007) Protective effects of organosulfur compounds towards N-nitrosamine-induced DNA damage in the single-cell gel electrophoresis (SCGE)/HepG2 assay. Food Chem Toxicol 45:1662–1669
Belloir C, Singh V, Daurat C, Siess MH, Le Bon AM (2006) Protective effects of garlic sulfur compounds against DNA damage induced by direct- and indirect-acting genotoxic agents in HepG2 cells. Food Chem Toxicol 44:827–834
Garcia A, Haza AI, Arranz N, Deigado ME, Rafter J, Morales P (2008) Organosulfur compounds alone or in combination with vitamin C protects towards N-nitrosopiperidine and N-nitrosodibutylamine-induced oxidative DNA damage in HepG2 cells. Chem Biol Interact 173:9–18
Amagase H, Milner JA (1993) Impact of various sources of garlic and their constituents on 7,12-dimethylbenz[a]anthracene binding to mammary cell DNA. Carcinogenesis 14:1627–1631
Hageman GJ, van Herwijnen MH, Schilderman PA, Rhijnsburger EH, Moonen EJ, Kleinjans JC (1997) Reducing effects of garlic constituents on DNA adduct formation in human lymphocytes in vitro. Nutr Cancer 27:177–185
Klebanoff SJ (2005) Myeloperoxidase: friend and foe. J Leukoc Biol 77:598–625
Tüzün A, Erdil A, Inal V, Aydin A, Bağci S, Yeşilova Z, Sayal A, Karaeren N, Dağalp K (2002) Oxidative stress and antioxidant capacity in patients with inflammatory bowel disease. Clin Biochem 35:569–572
Chaudhary G, Mahajan UB, Goyal SN, Ojha S, Patil CR, Subramanya SB (2017) Protective effect of Lagerstroemia speciosa against dextran sulfate sodium induced ulcerative colitis in C57BL/6 mice. Am J Transl Res 9:1792–1800
Bastaki SM, Adeghate E, Amir N, Ojha S, Oz M (2018) Menthol inhibits oxidative stress and inflammation in acetic acid-induced colitis in rat colonic mucosa. Am J Transl Res 10:4210–4222
Sheen LY, Chen HW, Kung YL, Liu CT, Lii CK (1999) Effects of garlic oil and its organosulfur compounds on the activities of hepatic drug-metabolizing and antioxidant enzymes in rats fed high- and low-fat diets. Nutr Cancer 35:160–166
Gudi VA, Singh SV (1991) Effect of diallyl sulfide, a naturally occurring anticarcinogen, on glutathione-dependent detoxifying enzyme of female CD-1 mouse tissues. Biochem Pharmacol 42:1261–1265
Chen L, Hong JY, So E, Hussin AH, Chen WF, Yang CS (1999) Decrease of hepatic catalase level by treatment with diallyl sulfide and garlic homogenates in rats and mice. J Biochem Mol Toxicol 13:127–134
Imal J, Ide N, Nagae S, Moriguchi T, Matsuura H, Itakura Y (1994) Antioxidant and radical scavenging effects of aged garlic extracts and its constituents. Planta Med 60:417–420
Fanelli SL, Castro GD, de Toranzo EG, Castro JA (1998) Mechanisms of the preventive properties of some garlic components in the carbon tetrachloride-promoted oxidative stress. Diallyl sulfide, diallyl disulfide, allyl mercaptan and allyl methyl sulfide. Res Comm Mol Pathol Pharmacol 102:163–174
Ye C, Dai DH, Hu WI (2013) Antimicrobial and antioxidant activities of the essential oil from onion. (Allium cepa L.). Food Control 30:48–53
Corzo-Martinez M, Corzo N, Villamiel M (2007) Biological properties of onions and garlic. Trends in Food Sci Technol 18:609–625
Takahashi M, Shibamoto T (2008) Chemical compositions and antioxidant/antiinflammatory activities of steam distillate from freeze-dried onion. (Allium cepa.L.) sprout. J Agricultural Food Chem 56:10462–10467
Garcia A, Morales P, Arranz N, Delgado ME, Rafter J, Haza AL (2009) Antiapoptotic effects of dietary antioxidants towards N-nitrosopiperidine and N-nitrosodibutylamine-induced apoptosis in HL-60 and HepG2 cells. J Appl Toxicol 29:403–413
Sundaran SG, Milner JA (1993) Impact of organosulfur compounds in garlic on canine mammary tumor cells in culture. Cancer Lett 74:85–90
Sundaran SG, Milner JA (1996) Diallyl disulfide induces apoptosis of human colon tumor cells. Carcinogenesis 17:669–673
Sakamoto K, Lawson LD, Milner JA (1997) Allyl sulfides from garlic suppress the in vitro proliferation of humanA549 lung tumor cells. Nutr Cancer 29:152–156
Welch C, Wuarin L, Sidell N (1992) antiproliferative effect of the garlic compound S-allyl cysteine on human neuroblastoma cells in vitro. Cancer Lett 63:211–219
Takeyama H, Hoon DS, Saxton RE, Morton DL, Irie RF (1993) Growth inhibition and modulation of cell markers of melanoma by S-allyl cysteine. Oncology 50:63–69
Pinto JT, Qiao C, Xing J, Rivlin RS, Protomastro ML, Weissler ML, Tao Y, Thaler H, Heston WD (1997) Effects of garlic thioallyl derivatives on growth, glutathione concentration and polyamine formation of human prostate carcinoma cells in culture. Am J Clin 66:398–405
Perchellet JP, Perchellet EM, Abney NL, Zirnstein JA, Belman S (1986) Effects of garlic and onion oils on glutathione peroxidase activity, the ratio of reduced/oxidized glutathione and ornithine decarboxylase induction in isolated mouse epidermal cells treated with tumor promotors. Cancer Biochem Biophys 8:299–312
Baer AR, Wargovich MJ (1989) Role of ornithine decarboxylase in diallyl sulfide inhibition of colonic radiation injury in the mouse. Cancer Res 49:5073–5076
Takada N, Matsuda T, Otoshi T, Yano Y, Otani S, Hasegava T, Nakae D, Konishi Y, Fukushima S (1994) Enhancement by organosulfur compounds from garlic and onion of diethylnitrosamine-induced glutathione S-transferase positive foci in the rat liver. Cancer Res 54:2895–2899
Kaufman SH, Gores GJ (2000) Apoptosis in cancer: cause and cure. BioEssays 22:1007–1017
Ghobriel IM, Witzig TE, Adjei AA (2005) Targeting apoptosis pathways in cancer therapy. CA Cancer J Clin 55:178–194
Dirsch VM, Gerbes AL, Vollmar AM (1998) Ajoene, a compound of garlic, induces apoptosis in human promyeloleukemia cells, accompanied by generation of reactive oxygen species and activation of nuclear factor kB. Mol Pharmacol 53:402–407
Kim J, Kim Y, Kyung K (2004) Inhibitory activity of essential oils of garlic and onion against bacteria and yeast. J Food Prot 67:499–504
Kim JW, Huh JE, Kyung SH, Kyung KH (2004) Antimicrobial activity of alk(en)yl sulfides found in essential oils of garlic and onion. Food Sci Biothechnol 13:235–239
Tsao S, Yin M (2001) In vitro activity of four diallyl sulfides occurring naturally in garlic and Chinese leek oils. J Med Microbiol 50:646–649
Appendini P, Hotchkiss PT (2002) Review of antimicrobial food packaging. Innov Food Sci Emerg Technol 3:113–126
Mellado-Garcia P, Maisanaba S, Puerto M, Liana-Luiz-Cabello M, Prieto AI, Marcos R, Pichardo S, Camean AM (2015) Genotoxicity assessment of propyl thiosulfinate oxide, an oranosulfur compound from Allium extract, intended to food packaging. Food Chem Toxicol 86:365–373
Cutter CN (2000) Antimicrobial effect of herb extracts against Escherichia coli O157:H7, Listeria monocytogenes, and Salmonella typhimurium associated with beef. J Food Prot 63:601–607
Nissen H, Alveseike O, Bredholt S, Holck A, Nesbakken T (2000) Comparison between the growth of Yersinia enterocolitica, Listeria monocytogenes, Escherichia coli O157:H7 and Salmonella Spp. in ground beef packed by three commercially used packaging techniques. Int J Food Microbiol 59:211–220
Dwivedi C, Abu-Ghazaleh A, Guenther J (1996) Effects of diallyl sulfide and diallyl disulfide on cisplatin-induced changes in glutathione-s-transferase activity. Anticancer Drugs 7:792–794
Yeh YY, Liu L (2001) Cholesterol-lowering effect of garlic extracts and organosulfur compounds: human and animal studies. J Nutr 131:989S-993S
Tsao SM, Yin MC (2001) In-vitro activity of garlic oil and four diallyl sulfides against antibiotic-resistant Pseudomonas aeroginosa and Klebsiella pneumloniae. J Antimicrob Chemother 47:665–670
Ross ZM, O’Gara EA, Hill DJ, Sleightholme HV, Maslin DJ (2001) Antimicrobial properties of garlic oil against human enteric bacteria: evaluation of methodologies and camparisons with garlic oil sulfides and garlic powder. App Environ Microbiol 67:475–480
Faustman C, Cassens RG (1990) The biochemical basis for discoloration of fresh meat; a review. J Muscle Foods 1(3):217–243. https://doi.org/10.1111/j.1745-4573.1990.tb00366.x
Mitsumoto M, Arnold RN, Schaefer DM, Cassens RG (1993) Dietary versus postmortem supplementation of vitamin E on pigment and lipid stability in ground beef. J Anim Sci 71:1812–1816
Zhao T, Doyle MP, Berg DE (2001) Fate of Campylobacter jejuni in butter. J Food Prot 63:120–122
Snoussi M, Trabelsi N, Dehmeni A, Benzekri R, Bouslama L, Hajlaoui B, Papetti A (2016) Phytochemical analysis, antimicrobial and antioxidant activities of Allium roseum var. odoratissimum (Desf.) Coss extracts. Ind Crops Products 89:533–542
Marchese A, Berbieri R, Sanchez-Silva A, Dagila M, Nabavi SF, Jafari NJ, Izadi M, Ajami A, Nabawi SM (2016) Antifungal and antibacterial activities of allicin: a review. Trends Food Sci Tech 52:49–56
Sharma D, Rani R, Chaturvedi M, Rohilla P, Yadav J (2019) In silico and in vitro approach of Allium cepa and isolated quercetin against MDR bacterial strains and Mycobacterium smegmatis. S Afr J Bot 124:29–35. https://doi.org/10.1016/j.sajb.2019.04.019
Beceiro A, Tomás M, Bou G (2013) Antimicrobial resistance and virulence: a successful or deleterious association in the bacterial world? Clin Microbiol Rev 26:185–230. https://doi.org/10.1128/CMR.00059-12
Al-Yousef HM, Ahmed AF, Al-Shabib NA, Laeeq S, Khan RA, Rehman MT, Alsalme A, Al-Ajmi MF, Khan MS, Husain FM (2017) Onion peel ethylacetate fraction and its derived constituent Quercetin 4′-O-β-D Glucopyranoside attenuates quorum sensing regulated virulence and biofilm formation. Front Microbiol 8:1675. https://doi.org/10.3389/fmicb.2017.01675
Quecan BXV, Santos JT, Rivera ML, Hassimotto NM, Almeida FA, Pinto UM (2019) Effect of quercetin rich onion extracts on bacterial quorum sensing. Front Microbiol 10:867. https://doi.org/10.3389/fmicb.2019.00867
Burt S (2004) Essential oils: their antibacterial properties and potential application in foods—a review. Int J Food Microbiol 94:223–253
Amagase H, Petesch BL, Matsuura H, Kasuga S, Itakura Y (2001) Intake of garlic and its active components. J Nutr 131(3):955–962
Zeng T, Guo FF, Zhang CL, Song FY, Zhao XL, Xie KQ (2012) A meta-analysis of randomized, double-blind, placebo-controlled trials for the effects of garlic on serum lipid profiles. J Sci Food Agri 92(9):1892–1902
Liu L, Yeh YY (2001) Water-soluble organosulfur compounds of garlic inhibit fatty acid and triglyceride syntheses in cultured rat hepatocytes. Lipids 36(4):395–400
Yeh YY, Liu L (2001) Cholesterol-lowering effects of garlic extracts and organosulfur compounds: human and animal studies. J Nutr 131(3):989S-993S
Lau BHS (2001) Suppresion of LDL oxidation by garlic. J Nutr 131:985–988
Ried K, Frank OR, Stocks NP (2008) Aged garlic extract lowers blood pressure in patients with treated but uncontrolled hypertension: a randomized controlled trial. Maturitas 67(2):144–150
Qidwai W, Qureshi R, Hasan SN, Azam SI (2000) Effects of dietary garlic (Allium sativum) on the blood pressure in humans—a pilot study. J Pak Med Assoc 50(6):204–207
Sobenin IA, Pryanishkov VV, Kunnova LM, Rabinovich YA, Martirosyan DM, Orekhov AN (2010) The effects of time-released garlic powder tablets on multifunctional cardiovascular risks in patients with coronary heart disease. Lipids Health Dis 9:119
Kim-park S, Ku DD (2000) Garlic elicits a nitric oxide-dependend relaxation and inhibits hypoxic pulmonary vasoconstriction in rats. Clin Exp Pharmacol Physiol 27(10):780–786
Morihara N, Sumioka I, Moriguchi T, Uda N, Kyo E (2002) Aged garlic extract enhances production of nitric oxide. Life Sci 71(5):509–517
Gouvea SA, Moyses MR, Bissoli NS, Pires JG, Cabral AM, Abreu GR (2003) Oral administration of L-arginine decreases blood pressure and increases renal excretion of sodium and water in renovascular hypertensive rats. Braz J Med Biol Res 36(7):943–949
Zhao W, Zhang J, Lu Y, Wang R (2001) The vasorelaxant effect of H(2)S as a novel endogenous gaseous K(ATP) channel opener. EMBO J 20(21):6008–6016
Shivarajah A, McDonald MC, Thiemermann C (2006) The production of hydrogen sulfide limits myocardial ischemia and reperfusion injury and contributes to the cardioprotective effects of preconditioning with endotoxin, but not ischemia in the rat. Shock 26(2):154–161
Koenitzer JR, Isbell TS, Patel HD, Benavides GA, Dickinson DA, Patel RP et al (2007) Hydrogen sulfide, mediates vasoactivity in an O2-dependent manner. Am J Physiol Heart Circ Physiol 292(4):H1953–H1960
Calvert JW, Coetzee WA, Lefer DJ (2010) Novel insights into hydrogen sulfide-mediated cytoprotection. Antioxid Redox Signal 12(10):1203–1217
Benavides GA, Squadrito GL, Mills RW, Patel HD, Isbell TS (2007) Hydrogen sulfide mediates the vasoactivity of garlic. Proc Natl Acad Sci USA 104(46):17977–17982
Pierre S, Crosbie L, Duttaroy AK (2005) Inhibitory effect of aqueous extracts of some herbs on human platelet aggregation in vitro. Platelets 16(8):469–473
Qi R, Liano F, Inoue K, Yatomi Y, Sato K, Yzaki Y (2000) Inhibition by diallyl trisulfide, a garlic component, of intracellular Ca (2+) mobilization without affecting inositol-1,4,5-triphosphate (IP(3)) formation in activated platelets. Biochem Pharmacol 60(10):1475–1483
Chang HS, Yamato O, Yamasaki M, Maede Y (2005) Modulatary influence of sodium 2-propenyl thiosullfate from garlic on cylcooxygenase activity in canine platelets: possible mechanism for the anti-aggregatory effects. Prostagladins Leuko Essent Fatty Acids 72(5):351–355
Bergmann C, Sano M (2006) Cardiac risk factors and potential treatments in Alzheimer’s disease. Neurol Res 28(6):595–604
Stampfer MJ (2006) Cardiovascular disease and Alzheimer’s disease: common links. Intern Med 260(3):211–223
Rosendorff C, Beeri MS, Silverman JM (2007) Cardiovascular risk factors for Alzheimer’s disease. Am J Geriatr Cardiol 16(3):143–149
Launer LJ (2002) Demonstrating the case that AD is a vascular disease: epidemiological evidence. Aging Res Rev 1:6–77
Chauhan NB (2006) Effect of aged garlic extract on APP processing and tau phosphorylation in Alzheimer’s disease transgeneic model Tg2576. J Ethnopharmacol 108(3):385–394
Ray B, Chauhan NB, Lahiri DK (2011) The “aged garlic extract” (AGE) and one of its active ingredients S-allyl-L-cysteine(SAC) as potential preventive and therapeutic agents for Alzheimer’s disease (AD). Curr Med Chem 18(22):33063313
Ray B, Lahiri CNB, Lahiri DK (2011) Oxidative insults to neurons and synapse are prevented by aged garlic extract S-allyl-L-cysteine treatment in the neuronal culture and APP-Tg mouse model. J Neurochem 117(3):388–402
Kim JM, Chang N, Kim WK, Chun HS (2006) Dietary S-allyl-L-cysteine reduces mortality with decrease incidence of stroke and behavioral changes in stroke-prone spontaneously hypertensive rats. Biosci Biotech Biochem 70(8):1969–1971
Atif F, Yousuf S, Agrawal SK (2009) S-allyl-L-cysteine diminishes cerebral ischemia-induced mitochondrial dysfunction in hippocampus. Brain Res 1265:128–137
Garcia E, Villeda-Hernandez J, Pedraza-Chaverri J, Maldonado PD, Santamaria A (2010) S-allylcysteine reduces MPTP-induced striatal cell damage via inhibition of pro-inflammatory cytokine tumor necrosis factor alpha and inducible nitric oxide synthase expression in mice. Phytomedicine 18(1):65–73
Numagami Y, Sato S, Ohnishi ST (1996) Attenuation of rat ischemic brain damage by aged garlic extract: a possible protecting mechanism as antioxidants. Neurochem Int 29(2):135–143
Perez-Severiano F, Salvatierra-Sanchez R, Rodriguez-Perez M, Cuevas-Martinez EY, Guevara J, Limon D, Maldonado PD et al (2004) S-allylcysteine prevents amyloid-beta peptide-induced oxidative stress in rat hippocampus and ameliorates learning deficits. Eur J Pharmacol 489(3):197–202
Peng Q, Buzzard AR, Lau BH (2002) Neuroprotective effects of garlic compounds in amyloid-beta peptide induced apoptosis in vitro. Med Sci Monit 8:328–337
Jackson R, McNeil B, Taylor C, Holl G, Ruff D, Gwebu ET (2002) Effect of aged garlic extract on caspase-3-activity in vitro. Nutr Neuroscience 5:287–290
Koh SH, Kwon H, Park KH, Ko JK, Kim JH, Hwang MS et al (2005) Protective effect of diallyl disulfide on oxidative stress injured neuronally differentiated PC 12 cells. Brain Res 133(2):176–186. https://doi.org/10.1016/j.molbrainres.2004.10.006
Coyle JT, Puttfarcken P (1993) Oxidative stress, glutamate and neurodegeneration disorders. Science 262:689–695
Saleem S, Ahmed M, Ahmed AS, Yousuf S, Ansari MA, Khan MB, Ishrat T, Islam F (2006) Behavioral and histologic neuroprotection of aqueous garlic extracts after reversible local cerebral ischemia. J Med food 9(4):537–544
Gupta R, Singh M, Sharma A (2003) Neuroprotective effect of antioxidants on ischemia reperfusion-induced cerebral injury. Pharmacol Res 48(2):209–215
Zeng T, Cuo FF, Zhang CL, Zhao S, Dou DD, Gao XC et al (2008) The anti-fatty liver effects of garlic oil on cute ethanol-exposed mice. Chem Biol Interact 176(2–3):234–242
Nencini C, Franchi GG, Cavallo F, Micheli L (2010) Protective effect of Allium neopolitanum Cyr. Versus Allium Sativum L on acute ethanol-induced oxidative stress in rat liver. J Med Food 13(2):329–335
Kishimoto R, Ueda M, Yoshinaga H, Goda K, Park SS (1999) Combined effects of ethanol and garlic on hepatic ethanol metabolism in mice. J Nutr Sci Vitaminol 45(3):275–286
Wu CC, Sheen NY, Chen HW, Tsai SJ, Lii CK (2001) Effects of organosulfur compounds from garlic oil on the antioxidant system in the rat liver and red blood cells. Food Chem Toxicol 39(6):563–569
Khosla P, Karam RS, Bhargava VK (2004) Effect of garlic oil on chemical induced gastric ulcers in rats. Phytother Res 18(1):87–91
Park EY, Ki SH, Ko MS, Kim CW, Lee MH, Lee YS et al (2005) Garlic oil and DDB, comprised in a pharmaceutical composition and treatment of patients with viral hepatitis, prevents acute liver injuries potentiated by glutathione deficiency in rats. Chem Biol Interact 155(1–2):82–96
Gedik N, Kabasaki L, Sehirli O, Ercan F, Sirvanci S, Keyer-uysar M (2005) Long-term administration of aqueous garlic extract (AGE) alleviates liver fibrosis and oxidative damage induced by biliary obstruction in rats. Life Sci 76(22):2593–2606
Chung LY (2006) The antioxidant properties of garlic compounds: allyl cysteine, allinin, allicin and allyl disulfide. J Med Food 9(2):205–213
Xiao J, Ching YP, Liong EC, Nanaji AA, Fung ML, Tipoe JL (2013) Garlic-derived S-allylmercaptocysteine is a hepato-protective agent in non-alcoholic fatty liver disease in vivo animal model. Eur J Nutr 52(1):179–191
Raso GM, Meli R, Di Carlo G, Pacilio M, Di Carlo R (2001) Inhibition of inducible nitric oxide synthase and cyclooxygenase-2-expression by flavonoids in macrophage J774A. Life Sci 68(8):921–931
Gu G, Du Y, Hu H, Jin C (2003) Synthesis of 2-chloro-4-nitrophenylalpha-L-fucopyranoside: a substrates for alpha-L-fucosidase (AFU). Carbohydr Res 338(15):1603–1607
Chang HP, Chen YH (2005) Differential effects of organosulfur compounds from garlic oil on nitric oxide and prostaglandin E2 in stimulated macrophages. Nutrition 21(4):530–536
Kyo E, Uda N, Kasuga S, Itakura Y (2001) Immunomodulatory effect of aged garlic extract. J Nutr 131(3s):1075S-1079S
Zare A, Farzaneh P, Pourpak Z, Zahedi F, Moin M, Shahabi S (2008) Purified aged garlic extract modulates allergic airway inflammation in BALB/c mice. Iran J Allergy Asthma Immunol 7(3):133–141
Jeong YY, Park HJ, Cho YW, Kim EJ, Kim CT, Mun YJ et al (2010) Aged red garlic extract reduces cigarette smoke-extract-induced cell death in human bronchial smooth muscle cells by increasing intercellular glutathione levels. Phytother Res 26(1):18–25
William FM, Skimmer J, Spector TD, Cassidy A, Clarke IM, Davidson RM et al (2010) Dietary garlic and hip osteoarthritis; evidence of a protective effect and putative mechanism of action. BMC Musculoskelet Disord 11:280
Lee HS, Lee CH, Tsai HC, Salters DM (2009) Inhibition of cyclooxygenase 2 expression by diallyl sulfide on joint inflammation induced by urate crystal and IL1β. Osteoarthr Cartil 17(1):91–99
Amir N, Al Dhaheri A, Al Jaberi N, Al Marzouki F, Bastaki SMA (2011) Comparative effect of garlic (Allium sativum), onion (Allium cepa) and black seed (Nigella sativa) on gastric acid secretion and gastric ulcer. Res Report Med Chem 1:1–7
Vasudevan K, Vember S, Veeraghavan K, Haranath PS (2000) Influence of intragastric perfusion of aqueous spice extract on acid secretion in anesthetized albino rats. Indian J Gastroenterol 19(2):53–56
Rafsanjani FN, Shahrani M, Vahedian J (2006) Garlic effects on gastric acid and pepsin secretion in rat. Pak Med Sci 22(3):265–288
Bastaki SMS, Chandranath SI, Singh J (2002) Comparison of antisecretory and antiulcer activity of epidermal growth factor, urogastrone and transferring growth factor alpha and its derivatives in rodents in vivo. Mol Cell Biochem 236:83–94
Bastaki SMA, Hasan MY, Chandranath SI, Schmassmann A, Garner A (2003) PD-136450: a CCK 2 (gastrin) receptor antagonist with antisecretory, anxiolytic and antiulcer activity. Mol Cell Biochem 252:83–90
Khosla P, Karan RS, Bhargava VK (2004) Effect of garlic oil on ethanol induced gastric ulcer in rats. Phytother Res 18:87–91
Goldman IL, Kopelberg M, Debaene JEP, Schwartz BS (1996) Antiplatelet activity of onion (Allium cepa) is sulfur dependent. Thromb Haemost 54:183–186
Esplugues JV, Whittle BJR (1990) Close-arterial administration of the thromboxane mimetic U-46619 induces damage to the rat gastric mucosa. Prostaglandins 35:137–144
Konturek SJ, Brzozowski T, Piatucki I, Radecki T, Dembińska-Kieć A (1983) Role of prostaglandin and thromboxane biosynthesis in gastric necrosis produced by taurocholate and ethanol. Dig Dis Sci 28:154–160
Sumiyoshi H, Wargovich MJ (1989) Garlic (Allium sativum): a review of its relationship to cancer. Asia Pacific J Pharmacol 4:133–140
Shen TY (1977) Prostaglandin synthetase inhibitors. In: Berti F, Samuelsson B, Velo GP (eds) Prostaglandins and thromboxanes, vol 13. Springer, Boston
Bastaki SMA, Amir N (2018) Effect of allylsulfide, allyldisulfide and quercetin on indomethacin- and alcohol-induced gastric ulcer induced in the rat in vivo. US Patent number 10.039, 738, 7 Aug 2018
Rizvi SI, Mishra N (2013) Traditional Indian medicines used for the management of diabetes mellitus. J Diabetes Res. https://doi.org/10.1155/2013/712092
Akash MSH, Shen Q, Rehman K, Chen S (2012) Interleukin-1 receptor antagonist: a new therapy for type 2 diabetes mellitus. J Pharm Sci 101:147–1658
Rosenstock J (2000) Treatment strategies and new therapeutic advances for type 2 diabetes mellitus. Diabetes Edu 26:14–18
Cuny T, Guerci B, Cariou B (2012) New avenues for the pharmacological management of type 2 diabetes mellitus: an update. Ann Endocrinol 73:459–468
Akash MSH, Rehman K, Sun H, Chen S (2013) Interleukin-1 receptor antagonist improves normoglycemia and insulin sensitivity in diabetic in Goto-kakizaki-rats. Eur J Pharmacol 701:87–95
Rajan M, Kumar KY, Kumar PS, Ramaniyam RT, Kumar NS (2010) Antidiabetic activity of ethanolic extract on Albizia odoratissima (Lf) in alloxan-induced diabetic rats. Ind J Pharm Sci 2:786–791
Romila Y, Mazumdar PR, Choudury MD (2010) A review on antidiabetic used by the people of Manipur characterized by hypoglycemic activity. Assam Uni J Sci Tech Bioenviron Sci 6(1):167–175
Assaduzammam M, Akhtar MF, Islam MA, Khan MRI, Anisuzammam ASM, Ahmed M (2010) Evaluation of antidiabetic, antihyperlipidimic and hepatoprotective effects of Allium sativum (Linn) in alloxan induce diabetic rat. Bangladesh Pharmaceutical Journal 13(1):28–33
Rehman K, Akash MSH, Azhar S, Khan SA, Abid R, Waseem A et al (2012) A Biochemical and histopathologic study showing protection and treatment of gentamycin-induced nephrotoxicity in rabbits using vitamin C. Afr J Tradit Complement Altern Med 9:360–365
Parasd SK, Kulshreshtha A, Qureshi TN (2009) Antidiabetic activity of some herbal plants in streptozotocin-induced diabetes. Pak J Nutr 8:551–557
Patil R, Patil R, Ahirwar B, Ahirwar D (2011) Current status of Indian medicinal plants with antidiabetic potential: a review. Asian Pac J Trop Biomed. https://doi.org/10.1016/S2221-1691(11)60175-5
Kumari K, Augusti KT (2002) Antidiabetic and antioxidant effects of S-methyl cysteine sulfoxide isolated from onion (Allium cepa Linn.) as compared to standard drugs in alloxan diabetic rats. Ind J Exp Biol 40:1005–1009
El-Demerdash FM, Yousef MI, El-Naga NIA (2005) Biochemical study on the hypoglycemic effects of onion and garlic in alloxan-induced diabetic rats. Food Chem Toxicol 43:57–63. https://doi.org/10.1016/j.fct.2004.08.012
Ogunmodede CS, Sallu IC, Ogunlade B, Akunna CG, Oyewopo AO (2012) An evaluation of the hypoglycemic antioxidant and hepatoprotective potential of onion (Allium cepa L.) on alloxan-induced diabetic rabbits. Int J Pharmacol 8:21–29
Babu PS, Srinivasan K (1997) Influence of dietary capsaicin and onion on the metabolic abnormalities associated with streptozotocin-induced diabetes mellitus. Mol Cell Biochem 175:49–57
Harrikesh D, Anamika S, Angad Mohanrao P, Chandrashekar Ramdas T, Babu Vinayak G (2012) Allicin, a SUR2 opener: possible mechanism for the treatment of diabetic hypertension in rats. Braz J Pharmacogn 22(5):1053–1105
Campbell JD, Sansom MS, Ashcroft FM (2003) Potassium channel regulation. EMBO Rep 4(11):1038–1042
Nagae S, Ushijima M, Hatono S, Imal J, Kasuga S, Matsuura H, Itakura Y, Higashi Y (1994) Pharmacokinetics of garlic compound S-allyl cysteine. Planta Med 60:214–217
Song G, Sumit B, Guangyi Y, Arijita D, Ming H (2013) Oral bioavailability challenges of natural products used in cancer chemoprevention. Prog Chem 25(9):1553–1574
Amano H, Kazamori D, Itoh K (2016) Pharmacokinetics and N-acetylation metabolism of S-methyl-L-cysteine and trans-S-1-propenyl-L-cysteine in rats and dogs. Xenobiotica 46:1017–1025
Amano H, Kazamori D, Itoh K, Kodera Y (2015) Metabolism, excretion, and pharmacokinetics of S-allyl-L-cysteine in rats and dogs. Drug Metabol Dispos 43:749–755
Kodera Y, Suzuki A, Imado O, Kasuga S, Sumioka I, Kanezawa A, Taru N, Fujikawa M et al (2002) Physical, chemical, and biological properties of s-allylcysteine, an amino acid derived from garlic. J Agri Food Chem 50:622–632
Lachmann G, Lorenz D, Radeck W, Siegers C-P (1994) The pharmacokinetics of the S35 labeled garlic constituents alliin, allicin and vinyldithiine. Arzneimittelforsschung 44:734–743
Pushpendran CK, Devasagayam TP, Chintalwar GJ, Banerji A, Eapen J (1980) The metabolic fate of (355)-diallyl disulfide in mice. Experentia 36:1000–1001
Lawson L, Hughes B (1992) Characterization of the formation of allicin and other thiosulfates from garlic. Planta Med 58(04):345–350
Berthold HD, Sudhop T, Bergman KV (1998) Effect of garlic oil preparation on serum lipoproteins and cholesterol metabolism. J Am Med Assoc 279:1900–1902
McCrindle BW, Helden E, Conner WT (1998) Garlic extract therapy in children with hypercholesterolemia. Arch Pediatr Adolesc Med 152:1089–1094
Rahman MS (2007) Allicin and other functional active components in garlic: health benefits and bioavailability. Int J Food Prop 10(2):245–268
Egen-Schwind C, Eckard R, Kemper FH (1992) Metabolism of garlic constituents in the isolated perfused rat liver. Planta Med 58:301–305. https://doi.org/10.1055/s-2006-961471
Omar SH, Al-Wabel NA (2010) Organosulfur compounds and possible mechanism of garlic in cancer. Saudi Pharm J 18(1):51–58. https://doi.org/10.1016/j.jsps.2009.12.007
Chope GA, Cools K, Terry LA (2011) Alliums (onion, garlic, leek and shallot). Health-Promot Prop Fruits Veg 1:5–28
Shankar S, Kumar D, Srivastava RK (2013) Epigenetic modification by dietary phytochemicals: implications for personalized nutrition. Pharmacol Therap 138(1):1–17. https://doi.org/10.1016/j.pharmthera.2012.11.002
Poojary MM, Putnik P, Bursać Kovaĉević D, Barba FJ, Lorenzo JM, Dias DA, Shepigilman A (2017) Stability and extraction of bioactive sulfur compounds from Allium genus processed by traditional and innovative technologies. H Food Comp Analysis 61:28–39. https://doi.org/10.1016/jfca.2017.04.007
Ramirez DA, Locatelli DA, Gonzalez RE, Cavagnaro PF, Camargo AB (2017) Analytical methods for bioactive sulfur compounds in Allium: an integrated review and future directions. J Food Comp Analysis 61:4–19. https://doi.org/10.1016/jfca.2016.09.012
Block E (2010) Garlic and other Alliums. The lore and the science. Royal Society of Chemistry, UK
Banergee SK, Mukherjee PK, Maulik SK (2003) Garlic as an antioxidant: the good, the bad and the ugly. Phytother Res 17:97–106
Wilson EA, Demmig-Adams B (2007) Antioxidant anti-inflammatory and antimicrobial properties of garlic and onions. Nutr Food Sci 37(3):178–183
Veitch NC, Smith M, Barnes J, Anderson LA, Phillipson JD (2013) Herbal Medicines, 4th edn. Pharmaceutical Press, London
Jappe U, Bonnekoh B, Hausen BM, Gollnick H (1999) Garlic-related dermatoses: a case report and review of the literature. Am J Contract Derm 10:37–39
Dixit VP, Joshi S (1982) Effects of chronic administration of garlic (Allium sativum Linn) on testicular function. Ind J Exp Biol 20(7):534–536
Kodera Y (2004) Dietary tolerance/absorption/metabolism of garlic. In: Lanchance P (ed) Nutraceuticals: designer foods. III garlic, soy and licorice. CT: Food & Nutrition Press, Trumbell, pp 95–105
Nagakawa S, Masamoto K, Sumiyoshi H, Harada H (1984) Acute toxicity test of garlic extract. J Toxicol Sci 9(1):57–60
Sumiyoshi H, Kanezawa A, Masamoto K, Harada H, Nakagami S, Yokota A (1984) Chronic toxicity test of garlic extracts in rats. J Toxicol Sci 9(1):61–75
Yoshida S, Hiran Y, Nakagawa S (1984) Mutagenicity and cytotoxicity tests of garlic. J Toxicol Sci 9(1):77–86
Nagakawa S, Masamoto K, Sumiyoshi H, Kunihiro K, Fuwa T (1980) Effect of raw and extract-aged garlic juice on growth of young rats and their organs after oral administration of (Author’s translation). J Toxicol Sci 5(1):91–112
Assayed ME, Khalaf AA, Salem HA (2010) Protective effects of garlic extract and vitamin C against in vivo cypermethrin-induced teratogenic effects in rat offspring. Food Chem Toxicol 48(11):3153–3158
Stevenson C, Pittler MH, Ernst E (2000) Garlic for treating hypercholesterolemia. A meta-analysis of randomized clinical trials. Ann Internal Med 133(6):420–429. https://doi.org/10.7326/0003-4819-133-6-200009190-00009
Steiner M, Khan AH, Holbert D, Lin RI (1996) A double-blind crossover study in moderately hypercholesterolemic men that compare the effect of aged garlic extract and placebo administration on blood lipids. Am J Clin Nutr 64(6):866–870
Musk SR, Clapham P, Johnson IT (1997) Cytotoxicity and genotoxicity of diallyl sulfide and diallyl disulfide towards Chinese hamster ovary cells. Food Chem Toxicol 35(3–4):379–385. https://doi.org/10.1016/s0278-6915(97)00120-8
Musk SRR, Clapham P, Johnson IT (1997) Cytotoxicity and genotoxicity of diallyl sulfide and diallyl disulfide towards Chinese hamster ovary cells. Food Chem Toxicol 35:379–385
Macan H, Uykimpang R, Alconcel M, Takasu J, Razon R, Amagase H et al (2006) Aged garlic extract may be safe for patients on warfarin therapy. J Nutr 136(3):793S-795S
Benkehlia N (2004) Antimicrobial activity of essential oil extracts of various onion (Allium cepa) and garlic (Allium sativum). LWT-Food Sci Technol 37(2):263–268
Lanzotti V (2006) The analysis of onion and garlic. J Chromatography A 1112:3–22
Xiao D, Choi S, Johnson DE, Vogel WC, Johnson CS, Trump DL et al (2004) Diallyl trisulfide –induced apoptosis in human prostate cancer cells involves C-Jun N-terminal kinase and extracellular-signal regulated kinase-phosphorylation Bcl-2. Oncogene 23:5594–5606
Shin HA, Cha YY, Park MS, Kim JM, Lim YC (2010) Diallyl sulfide induces growth inhibition and apoptosis of anaplastic thyroid cancer cells by mitochondrial signaling pathway. Oral Oncol 46:e15–e18
Sundaram SC, Milner JA (1996) Diallyl disulfide suppresses the growth of human colon cancer cell xenographs in athymic nude mice. J Nutr 126:1355–1361
Llana-Ruiz-Cabello M, Maisanaba S, Gutierrez-Praena D, Prieto AI, Pichardo S, Jos A, Moreno FJ, Camean A (2015) Cytotoxic and mutagenic in vitro assessment of two organosulfur compounds derived from onion to be used in the food industry. Food Chem 166:423–431
Acknowledgements
SMAB, SO and EA all contributed equally and did the literature survey. SMAB wrote the initial article, and SO and EA edited and made further contribution; HK created the chemical structures and edited the article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bastaki, S.M.A., Ojha, S., Kalasz, H. et al. Chemical constituents and medicinal properties of Allium species. Mol Cell Biochem 476, 4301–4321 (2021). https://doi.org/10.1007/s11010-021-04213-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-021-04213-2